Abstract
Background
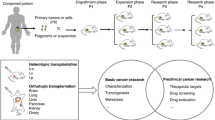
Preclinical trials of cancer therapeutics require both in vitro and in vivo evaluations. Recently, a patient-derived xenograft model in immunodeficient mice has been reported as a valuable in vivo evaluation system. In our current study, we aimed to establish a more efficient and accurate system for preclinical trials by generating primary cancer cells from patients and performing xenograft transfers of these cells into mice.
Methods
Human lung cancer specimens (n = 4) obtained from chemo-naive patients were cultured in bronchiolar epithelial basal medium supplemented with growth factors, followed by inoculation into non-obese diabetic/severe combined immunodeficient mice. The generated tumors in the mice were validated phenotypically and genetically using the original specimen and primary cancer cells.
Results
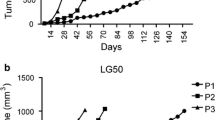
Immunohistochemical analysis of marker proteins, including cytokeratin 7, cytokeratin 20, epidermal growth factor receptor, thyroid transcription factor-1, CD56, chromogranin, and synaptophysin, demonstrated that the xenograft tumors were originated from the patient tumors. Moreover, mutation profiling using the OncoMap System, which analyzes mutations at 440 sites in 41 tumor-related genes, showed the same patterns in both the patient and xenograft tumors.
Conclusions
These results indicate that our animal system is suitable for the amplification of patient tumors and will therefore be beneficial for both in vivo and in vitro assessments and preclinical trials of chemotherapeutics. This has the potential to provide a very effective tool for future personalized therapy and for conducting translational lung cancer research.




Similar content being viewed by others
References
Anguiano A, Nevins JR, Potti A (2008) Toward the individualization of lung cancer therapy. Cancer 113:1760–1767
Azzoli CG, Park BJ, Pao W, Zakowski M, Kris MG (2008) Molecularly tailored adjuvant chemotherapy for resected non-small cell lung cancer: a time for excitement and equipoise. J Thorac Oncol 3:84–93
Bepler G, Begum M, Simon GR (2008) Molecular analysis-based treatment strategies for non-small cell lung cancer. Cancer Control 15:130–139
Bertolini G, Roz L, Perego P, Tortoreto M, Fontanella E, Gatti L, Pratesi G, Fabbri A, Andriani F, Tinelli S, Roz E, Caserini R, Lo Vullo S, Camerini T, Mariani L, Delia D, Calabro E, Pastorino U, Sozzi G (2009) Highly tumorigenic lung cancer CD133+ cells display stem-like features and are spared by cisplatin treatment. Proc Natl Acad Sci USA 106:16281–16286
Cree IA, Glaysher S, Harvey AL (2010) Efficacy of anti-cancer agents in cell lines versus human primary tumour tissue. Curr Opin Pharmacol 10:375–379
Daniel VC, Marchionni L, Hierman JS, Rhodes JT, Devereux WL, Rudin CM, Yung R, Parmigiani G, Dorsch M, Peacock CD, Watkins DN (2009) A primary xenograft model of small-cell lung cancer reveals irreversible changes in gene expression imposed by culture in vitro. Cancer Res 69:3364–3373
De Witt Hamer PC, Van Tilborg AA, Eijk PP, Sminia P, Troost D, Van Noorden CJ, Ylstra B, Leenstra S (2008) The genomic profile of human malignant glioma is altered early in primary cell culture and preserved in spheroids. Oncogene 27:2091–2096
Dong X, Guan J, English JC, Flint J, Yee J, Evans K, Murray N, Macaulay C, Ng RT, Gout PW, Lam WL, Laskin J, Ling V, Lam S, Wang Y (2010) Patient-derived first generation xenografts of non-small cell lung cancers: promising tools for predicting drug responses for personalized chemotherapy. Clin Cancer Res 16:1442–1451
Gedaly R, Angulo P, Chen C, Creasy KT, Spear BT, Hundley J, Daily MF, Shah M, Evers BM (2012) The role of PI3 K/mTOR inhibition in combination with sorafenib in hepatocellular carcinoma treatment. Anticancer Res 32:2531–2536
Herbst RS, Heymach JV, Lippman SM (2008) Lung cancer. N Engl J Med 359:1367–1380
Hodkinson PS, Mackinnon AC, Sethi T (2007) Extracellular matrix regulation of drug resistance in small-cell lung cancer. Int J Radiat Biol 83:733–741
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ (2009) Cancer statistics, 2009 CA: a cancer journal for clinicians 59: 225–249
John T, Kohler D, Pintilie M, Yanagawa N, Pham NA, Li M, Panchal D, Hui F, Meng F, Shepherd FA, Tsao MS (2011) The ability to form primary tumor xenografts is predictive of increased risk of disease recurrence in early-stage non-small cell lung cancer. Clin Cancer Res 17:134–141
John T, Yanagawa N, Kohler D, Craddock KJ, Bandarchi-Chamkhaleh B, Pintilie M, Sykes J, To C, Li M, Panchal D, Chen W, Shepherd FA, Tsao MS (2012) Characterization of lymphomas developing in immunodeficient mice implanted with primary human non-small cell lung cancer. J Thorac Oncol 7:1101–1108
Julien S, Merino-Trigo A, Lacroix L, Pocard M, Goere D, Mariani P, Landron S, Bigot L, Nemati F, Cuilliere-Dartigues P, Weiswald LB, Lantuas D, Morgand L, Pham E, Gonin P, Dangles-Marie V, Job B, Dessen P, Bruno A, Pierre A, De The H, Soliman H, Nunes M, Lardier G, Calvet L, Demers B, Prevost G, Vrignaud P, Roman-Roman S, Duchamp O, Berthet C (2012) Characterization of a large panel of patient-derived tumor xenografts representing the clinical heterogeneity of human colorectal cancer. Clin Cancer Res : an official journal of the American Association for Cancer Research
Kerbel RS (2003) Human tumor xenografts as predictive preclinical models for anticancer drug activity in humans: better than commonly perceived-but they can be improved. Cancer Biol Ther 2:S134–S139
MacConaill LE, Campbell CD, Kehoe SM, Bass AJ, Hatton C, Niu L, Davis M, Yao K, Hanna M, Mondal C, Luongo L, Emery CM, Baker AC, Philips J, Goff DJ, Fiorentino M, Rubin MA, Polyak K, Chan J, Wang Y, Fletcher JA, Santagata S, Corso G, Roviello F, Shivdasani R, Kieran MW, Ligon KL, Stiles CD, Hahn WC, Meyerson ML, Garraway LA (2009) Profiling critical cancer gene mutations in clinical tumor samples. PLoS One 4:e7887
Marangoni E, Vincent-Salomon A, Auger N, Degeorges A, Assayag F, de Cremoux P, de Plater L, Guyader C, De Pinieux G, Judde JG, Rebucci M, Tran-Perennou C, Sastre-Garau X, Sigal-Zafrani B, Delattre O, Dieras V, Poupon MF (2007) A new model of patient tumor-derived breast cancer xenografts for preclinical assays. Clin Cancer Res 13:3989–3998
Merk J, Rolff J, Becker M, Leschber G, Fichtner I (2009) Patient-derived xenografts of non-small-cell lung cancer: a pre-clinical model to evaluate adjuvant chemotherapy? Eur J Cardiothorac Surg 36:454–459
Moro M, Bertolini G, Tortoreto M, Pastorino U, Sozzi G, Roz L (2012) Patient-derived xenografts of non small cell lung cancer: resurgence of an old model for investigation of modern concepts of tailored therapy and cancer stem cells. J Biomed Biotechnol 2012:568567
Pandita A, Aldape KD, Zadeh G, Guha A, James CD (2004) Contrasting in vivo and in vitro fates of glioblastoma cell subpopulations with amplified EGFR. Genes Chromosomes Cancer 39:29–36
Rubio-Viqueira B, Hidalgo M (2009) Direct in vivo xenograft tumor model for predicting chemotherapeutic drug response in cancer patients. Clin Pharmacol Ther 85:217–221
Spira A, Ettinger DS (2004) Multidisciplinary management of lung cancer. N Engl J Med 350:379–392
Wehenkel M, Ban JO, Ho YK, Carmony KC, Hong JT, Kim KB (2012) A selective inhibitor of the immunoproteasome subunit LMP2 induces apoptosis in PC-3 cells and suppresses tumour growth in nude mice. Br J Cancer 107:53–62
Yoshimasu T, Ohta F, Oura S, Tamaki T, Shimizu Y, Naito K, Kiyoi M, Hirai Y, Kawago M, Okamura Y (2009) Histoculture drug response assay for gefitinib in non-small-cell lung cancer. Gen Thorac Cardiovasc Surg 57:138–143
Acknowledgments
This study was supported by a grant of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A062254 and HI10C2014), and by the Leading Foreign Research Institute Recruitment Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (MSIP) (2011-0030105).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hyang Sook Seol and Young-Ah Suh contributed equally to this work.
Rights and permissions
About this article
Cite this article
Seol, H.S., Suh, YA., Ryu, Yj. et al. A patient-derived xenograft mouse model generated from primary cultured cells recapitulates patient tumors phenotypically and genetically. J Cancer Res Clin Oncol 139, 1471–1480 (2013). https://doi.org/10.1007/s00432-013-1449-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-013-1449-6