Abstract
Purpose
A high lactate content in malignant head and neck cancer (Head and neck squamous cell carcinomas, HNSCC) is associated with a higher risk of metastatic spread and lower overall patient survival. However, until present, the underlying mechanisms are not clearly understood. Here, a systematic comparison of glucose metabolism in HNSCC and homologous normal tissue is presented for the first time.
Methods
The concentrations of glucose, lactate and ATP were measured in cryobiopsies of 29 human HNSCC and of 9 normal mucosa using bioluminescence imaging. The protein expression of lactate dehydrogenase (LDH) was analyzed by Western blotting.
Results
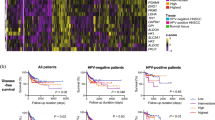
Tumors own a higher content of lactate and LDH in comparison with normal tissues. However, within the tumor group, the grade of LDH expression shows substantially strong variation and overlap with normal values. Furthermore, LDH expression was not correlated with tumor lactate content. Investigating a small subpopulation, patients with a short-term survival had significantly higher tumor lactate levels compared to patients with long-term survival.
Conclusions
The data provide clear evidence of an enhanced glycolysis in tumors compared to normal tissue. This may partially but not completely attributable to an elevated expression of LDH. High tumor lactate levels may be predictive for restricted patient survival. In conclusion, lactate measurements, for example non-invasively with MRT, should be advanced for use in clinical routine as a supportive tool for tumor diagnosis and prognosis.





Similar content being viewed by others
References
Albers MJ, Bok R, Chen AP, Cunningham CH, Zierhut ML, Zhang VY, Kohler SJ, Tropp J, Hurd RE, Yen YF, Nelson SJ, Vigneron DB, Kurhanewicz J (2008) Hyperpolarized 13C lactate, pyruvate, and alanine: noninvasive biomarkers for prostate cancer detection and grading. Cancer Res 68:8607–8615
Bier J (1981) Definitionen zum radikalchirurgischen Vorgehen bei Plattenepithelkarzinomen der Mundhöhle. Dtsch Z Mund Kiefer Gesichts Chir 6:369–372
Brindle K (2008) New approaches for imaging tumour responses to treatment. Nat Rev Cancer 8:94–107
Brizel DM, Schroeder T, Scher RL, Walenta S, Clough RW, Dewhirst MW, Mueller-Klieser W (2001) Elevated tumor lactate concentrations predict for an increased risk of metastases in head-and-neck cancer. Int J Radiat Oncol Biol Phys 51:349–353
Chen J, Zhao S, Nakada K, Kuge Y, Tamaki N, Okada F, Wang J, Shindo M, Higashino F, Takeda K, Asaka M, Katoh H, Sugiyama T, Hosokawa M, Kobayashi M (2003) Dominant-negative hypoxia-inducible factor-1 alpha reduces tumorigenicity of pancreatic cancer cells through the suppression of glucose metabolism. Am J Pathol 162:1283–1291
DeBerardinis RJ, Cheng T (2009) Q’s next: the diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene 29:313–314
Gatenby RA, Gillies RJ (2004) Why do cancers have high aerobic glycolysis? Nat Rev Cancer 4:891–899
Gottlieb E, Tomlinson IP (2005) Mitochondrial tumour suppressors: a genetic and biochemical update. Nat Rev Cancer 5:857–866
Isaacs JS, Jung YJ, Mole DR, Lee S, Torres-Cabala C, Chung YL, Merino M, Trepel J, Zbar B, Toro J, Ratcliffe PJ, Linehan WM, Neckers L (2005) HIF overexpression correlates with biallelic loss of fumarate hydratase in renal cancer: novel role of fumarate in regulation of HIF stability. Cancer Cell 8:143–153
Koukourakis MI, Giatromanolaki A, Sivridis E, Bougioukas G, Didilis V, Gatter KC, Harris AL (2003) Lactate dehydrogenase-5 (LDH-5) overexpression in non-small-cell lung cancer tissues is linked to tumour hypoxia, angiogenic factor production and poor prognosis. Br J Cancer 89:877–885
Krishnamachary B, Berg-Dixon S, Kelly B, Agani F, Feldser D, Ferreira G, Iyer N, LaRusch J, Pak B, Taghavi P, Semenza GL (2003) Regulation of colon carcinoma cell invasion by hypoxia-inducible factor 1. Cancer Res 63:1138–1143
Kroemer G, Pouyssegur J (2008) Tumor cell metabolism: cancer’s Achilles’ heel. Cancer Cell 13:472–482
Lu H, Forbes RA, Verma A (2002) Hypoxia-inducible factor 1 activation by aerobic glycolysis implicates the Warburg effect in carcinogenesis. J Biol Chem 277:23111–23115
Matoba S, Kang JG, Patino WD, Wragg A, Boehm M, Gavrilova O, Hurley PJ, Bunz F, Hwang PM (2006) p53 regulates mitochondrial respiration. Science 312:1650–1653
Matoba M, Tonami H, Kondou T, Yokota H, Higashi K, Toga H, Sakuma T (2007) Lung carcinoma: diffusion-weighted MR imaging—preliminary evaluation with apparent diffusion coefficient. Radiology 243:570–577
Mueller-Klieser W, Walenta S (1993) Geographical mapping of metabolites in biological tissue with quantitative bioluminescence and single photon imaging. Histochem J 25:407–420
Mueller-Klieser W, Schaefer C, Walenta S, Rofstad EK, Fenton BM, Sutherland RM (1990) Assessment of tumor energy and oxygenation status by bioluminescence, nuclear magnetic resonance spectroscopy, and cryospectrophotometry. Cancer Res 50:1681–1685
Pore N, Jiang Z, Gupta A, Cerniglia G, Kao GD, Maity A (2006) EGFR tyrosine kinase inhibitors decrease VEGF expression by both hypoxia-inducible factor (HIF)-1-independent and HIF-1-dependent mechanisms. Cancer Res 66:3197–3204
Quennet V, Yaromina A, Zips D, Rosner A, Walenta S, Baumann M, Mueller-Klieser W (2006) Tumor lactate content predicts for response to fractionated irradiation of human squamous cell carcinomas in nude mice. Radiother Oncol 81:130–135
Rofstad EK (2000) Microenvironment-induced cancer metastasis. Int J Radiat Biol 76:589–605
Sattler UG, Hirschhaeuser F, Mueller-Klieser WF (2010) Manipulation of glycolysis in malignant tumors: fantasy or therapy? Curr Med Chem 17:96–108
Schwickert G, Walenta S, Sundfor K, Rofstad EK, Mueller-Klieser W (1995) Correlation of high lactate levels in human cervical cancer with incidence of metastasis. Cancer Res 55:4757–4759
Selak MA, Armour SM, MacKenzie ED, Boulahbel H, Watson DG, Mansfield KD, Pan Y, Simon MC, Thompson CB, Gottlieb E (2005) Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-alpha prolyl hydroxylase. Cancer Cell 7:77–85
Semenza GL (2002) HIF-1 and tumor progression: pathophysiology and therapeutics. Trends Mol Med 8:S62–S67
Vousden KH, Ryan KM (2009) p53 and metabolism. Nat Rev Cancer 9:691–700
Walenta S, Mueller-Klieser WF (2004) Lactate: mirror and motor of tumor malignancy. Semin Radiat Oncol 14:267–274
Walenta S, Dotsch J, Mueller-Klieser W (1990) ATP concentrations in multicellular tumor spheroids assessed by single photon imaging and quantitative bioluminescence. Eur J Cell Biol 52:389–393
Walenta S, Dellian M, Goetz AE, Kuhnle GE, Mueller-Klieser W (1992) Pixel-to-pixel correlation between images of absolute ATP concentrations and blood flow in tumours. Br J Cancer 66:1099–1102
Walenta S, Salameh A, Lyng H, Evensen JF, Mitze M, Rofstad EK, Mueller-Klieser W (1997) Correlation of high lactate levels in head and neck tumors with incidence of metastasis. Am J Pathol 150:409–415
Walenta S, Wetterling M, Lehrke M, Schwickert G, Sundfor K, Rofstad EK, Mueller-Klieser W (2000) High lactate levels predict likelihood of metastases, tumor recurrence, and restricted patient survival in human cervical cancers. Cancer Res 60:916–921
Walenta S, Schroeder T, Mueller-Klieser W (2002) Metabolic mapping with bioluminescence: basic and clinical relevance. Biomol Eng 18:249–262
Walenta S, Chau TV, Schroeder T, Lehr HA, Kunz-Schughart LA, Fuerst A, Mueller-Klieser W (2003) Metabolic classification of human rectal adenocarcinomas: a novel guideline for clinical oncologists? J Cancer Res Clin Oncol 129:321–326
Walenta S, Schroeder T, Mueller-Klieser W (2004) Lactate in solid malignant tumors: potential basis of a metabolic classification in clinical oncology. Curr Med Chem 11:2195–2204
Yaromina A, Quennet V, Zips D, Meyer S, Shakirin G, Walenta S, Mueller-Klieser W, Baumann M (2009) Co-localisation of hypoxia and perfusion markers with parameters of glucose metabolism in human squamous cell carcinoma (hSCC) xenografts. Int J Radiat Biol 85:972–980
Zhou S, Kachhap S, Singh KK (2003) Mitochondrial impairment in p53-deficient human cancer cells. Mutagenesis 18:287–292
Acknowledgments
This work was supported in part by the Deutsche Forschungsgemeinschaft (Mu 576/14-1) and the Stiftung fuer Innovation Rheinland-Pfalz (15202-38 62 61/606).
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ziebart, T., Walenta, S., Kunkel, M. et al. Metabolic and proteomic differentials in head and neck squamous cell carcinomas and normal gingival tissue. J Cancer Res Clin Oncol 137, 193–199 (2011). https://doi.org/10.1007/s00432-010-0875-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-010-0875-y