Abstract
Introduction
There have been no reports describing successful drug resistance reversal in tumors in the clinic until now. Conversion of drug resistance reversal studies to drug resistance prevention studies may assist in the development of more efficient anti-tumor strategies. The present study demonstrates the prevention of doxorubicin-induced drug resistance by tetrandrine on leukemia cells.
Materials and methods
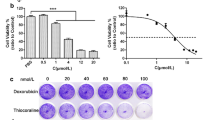
K562 cells were either solely treated with doxorubicin (0.6 mg/ml) or pretreated with tetrandrine of different concentrations (0.5, 1.0 and 2.0 μg/ml) for 24 h followed by combined-treatment with doxorubicin (0.6 μg/ml).
Results
The results showed that doxorubicin treatment only resulted in elevated levels of mdr1 mRNA/P-gp expression. Doxorubicin also induced up-regulation of P-gp functional activities, as intracellular retention of rhodamine was decreased; however, 2.0 μg/ml tetrandrine significantly inhibited the overexpression of doxorubicin-induced mdr1 mRNA/P-gp. Consistently, the functional activity of P-gp was also inhibited, which led to increased intracellular drug retention and the recovery of cell sensitivity to chemotherapeutic drugs in combined treatment groups. Both mRNA and protein levels of NF-κB were up-regulated in the cells treated with doxorubicin only. Results from an electrophoretic mobility shift assay and a chromatin immunoprecipitation assay demonstrated the enhanced binding to the promoter region of mdr1 gene compared to the control group. However, tetrandrine could markedly inhibit the doxorubicin-induced expression of NF-κB mRNA and protein. In addition, it also attenuated the NF-κB DNA-binding activity.
Conclusion
In summary, tetrandrine can prevent doxorubicin-induced mdr1 mRNA/P-gp expression and P-gp functions in a dose-dependent manner through a mechanism that may involve inhibition of doxorubicin-induced NF-κB mRNA expression and protein activity.




Similar content being viewed by others
References
Amiri KI, Richmond A (2005a) Role of nuclear factor-kappa B in melanoma. Cancer Metastasis Rev 24(2):301–313
Amiri KI, Richmond A (2005b) Role of nuclear factor-kappa B in melanoma. Cancer Metastasis Rev 24(2):301–313
Ao ZF, Xia W (1995) Reversal of daunorubicin resistance by tetrandrine in leukemic cells. Zhonghua Xue Ye Xue Za Zhi 16(5):235–238
Bentires-Alj M, Barbu V, Fillet M et al (2003) NF-kappaB transcription factor induces drug resistance through MDR1 expression in cancer cells. Oncogene 22(1):90–97
Dai CL, Xiong HY, Tang LF et al (2007) Tetrandrine achieved plasma concentrations capable of reversing MDR in vitro and had no apparent effect on doxorubicin pharmacokinetics in mice. Cancer Chemother Pharmacol 60(5):741–750
Friedenberg WR, Rue M, Blood EA et al (2006) Phase III study of PSC-833 (valspodar) in combination with vincristine, doxorubicin, and dexamethasone (valspodar/VAD) versus VAD alone in patients with recurring or refractory multiple myeloma (E1A95): a trial of the Eastern Cooperative Oncology Group. Cancer 106(4):830–838
Garcia MG, Alaniz L, Lopes EC (2005) et al. Inhibition of NF-kappaB activity by BAY 11-7082 increases apoptosis in multidrug resistant leukemic T-cell lines. Leuk Res 29(12):1425–1434
Gottesman MM, Fojo T, Bates SE (2002) Multidrug resistance in cancer: role of ATP-dependent transporters. Nature Rev Cancer 2:48–58
Ho LJ, Juan TY, Chao P et al (2004a) Plant alkaloid tetrandrine downregulates IkappaBalpha kinases-IkappaBalpha-NF-kappaB signaling pathway in human peripheral blood T cell. Br J Pharmacol 143(7):919–927
Ho LJ, Juan TY, Chao P et al (2004b) Plant alkaloid tetrandrine downregulates IκBα kinases-IκBα-NF-κB signaling pathway in human peripheral blood T cell. Bri J Pharmacol 143(7):919–927
Kothan S, Dechsupa S, Leger G et al (2004) Spontaneous mitochondrial membrane potential change during apoptotic induction by quercetin in K562 and K562/adr cells. Can J Physiol Pharmacol 82(12):1084–1090
List AF, Kopecky KJ, Willman CL et al (2001) Benefit of cyclosporine modulation of drug resistance in patients with poor-risk acute myeloid leukemia: a Southwest Oncology Group study. Blood 98:3212–3220
Lu XJ, Xu WL, Luo WJ et al (2008) Effect of tetrandrine on the doxorubicin-induced expression of mdr1 gene in K562 cells. Zhonghua Xue Ye Xue Za Zhi 29(7):468–471
Perez-Tomas R (2006) Multidrug resistance: retrospect and prospects in anti-cancer drug treatment. Curr Med Chem 13(16):1859–1876
Ramkissoon SH, Patel PS, Taborga M et al (2007) Nuclear factor-kappaB is central to the expression of truncated neurokinin-1 receptor in breast cancer: implication for breast cancer cell quiescence within bone marrow stroma. Cancer Res 67(4):1653–1659
Shinoda C, Maruyama M, Fujishita T et al (2005) Doxorubicin induces expression of multidrug resistance-associated protein 1 in human small cell lung cancer cell lines by the c-jun N-terminal kinase pathway. Int J Cancer 117(1):21–31
Xiu F, Alison S, Dominic M et al (1996) Cyclosporin A and PSC 833 prevent up-regulation of MDR1 expression by Anthracyclines in a human multidrug-resistant cell line. Clin Cancer Res 2:713–720
Xu JY, Zhou Q, Shen P et al (1999) Reversal effect of TTD on human multidrug resistant KBV200 cell line. J Exp Clin Cancer Res 18(4):549–552
Xu WL, Shen HL, Ao ZF et al (2006) Combination of tetrandrine as a potential-reversing agent with daunorubicin, etoposide and cytarabine for the treatment of refractory and relapsed acute myelogenous leukemia. Leuk Res 30(4):407–413
Zhang H, Li YY, Wu XZ et al (2006) Effect of Tetrandrine on LPS-induced NF-κB activation in isolated pancreatic acinar cells of rat. World J Gastroenterol 12(26):4232–4236
Zhu X, Sui M, Fan W (2005) In vitro and in vivo characterizations of tetrandrine on the reversal of P-glycoprotein-mediated drug resistance to paclitaxel. Anticancer Res 25(3B):1953–1962
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, H., Xu, W., Chen, Q. et al. Tetrandrine prevents acquired drug resistance of K562 cells through inhibition of mdr1 gene transcription. J Cancer Res Clin Oncol 136, 659–665 (2010). https://doi.org/10.1007/s00432-009-0704-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-009-0704-3