Abstract
Purpose
The purpose of this study was to characterize changes in the expression of copper–zinc superoxide dismutase (Cu/Zn-SOD) and manganese SOD (Mn-SOD) in oral squamous-cell carcinoma (OSCC).
Methods
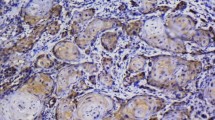
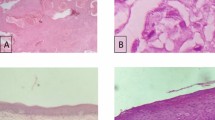
Real-time quantitative reverse transcriptase-polymerase chain reaction analysis of Cu/Zn-SOD and Mn-SOD mRNA expression was carried out in 50 pairs of OSCC tissue specimens and corresponding normal tissues. Mn-SOD protein expression was evaluated further in 65 OSCC tissue samples and 33 oral premalignant lesions (OPLs) using immunohistochemistry.
Results
Significant (P < 0.001) upregulation of Mn-SOD mRNA expression was observed in OSCC tissues compared with the normal tissue counterparts, whereas no significant difference was detected in Cu/Zn-SOD expression. Significant increases in Mn-SOD protein expression were seen in both OPLs (P < 0.001) and OSCC tissue (P < 0.001) together with a high incidence of lymph node metastasis (P = 0.04).
Conclusions
Our findings suggested that Mn-SOD overexpression is a frequent and early event during oral carcinogenesis and could contribute to aggressive OSCC.




Similar content being viewed by others
References
Bilalovic N, Sandstad B, Golouh R, Nesland JM, Selak I, Torlakovic EE (2004) CD10 protein expression in tumor and stromal cells of malignant melanoma is associated with tumor progression. Mod Pathol 17:1251–1258. doi:10.1038/modpathol.3800174
Chung-man Ho J, Zheng S, Comhair SA, Farver C, Erzurum SC (2001) Differential expression of manganese superoxide dismutase and catalase in lung cancer. Cancer Res 61:8578–8585
Connor KM, Hempel N, Nelson KK, Dabiri G, Gamarra A, Belarmino J, Van De Water L, Mian BM, Melendez JA (2007) Manganese superoxide dismutase enhances the invasive and migratory activity of tumor cells. Cancer Res 67:10260–10267. doi:10.1158/0008-5472.CAN-07-1204
Czesnikiewicz-Guzik M, Lorkowska B, Zapala J, Czajka M, Szuta M, Loster B, Guzik TJ, Korbut R (2008) NADPH oxidase and uncoupled nitric oxide synthase are major sources of reactive oxygen species in oral squamous cell carcinoma: potential implications for immune regulation in high oxidative stress conditions. J Physiol Pharmacol 59:139–152
Fisher CJ, Goswami PC (2008) Mitochondria-targeted antioxidant enzyme activity regulates radioresistance in human pancreatic cancer cells. Cancer Biol Ther 7:1271–1279
Guo G, Yan-Sanders Y, Lyn-Cook BD, Wang T, Tamae D, Ogi J, Khaletskiy A, Li Z, Weydert C, Longmate JA, Huang TT, Spitz DR, Oberley LW, Li JJ (2003) Manganese superoxide dismutase-mediated gene expression in radiation-induced adaptive responses. Mol Cell Biol 23:2362–2378. doi:10.1128/MCB.23.7.2362-2378.2003
Guzik TJ, Harrison DG (2006) Vascular NADPH oxidases as drug targets for novel antioxidant strategies. Drug Discov Today 11:524–533. doi:10.1016/j.drudis.2006.04.003
Hileman EA, Achanta G, Huang P (2001) Superoxide dismutase: an emerging target for cancer therapeutics. Expert Opin Ther Targets 5:697–710. doi:10.1517/14728222.5.6.697
Hwang TS, Choi HK, Han HS (2006) Differential expression of manganese superoxide dismutase, copper/zinc superoxide dismutase, and catalase in gastric adenocarcinoma and normal gastric mucosa. Eur J Surg Oncol 33:474–479. doi:10.1016/j.ejso.2006.10.024
Iwase K, Nagasaka A, Kato K, Itoh A, Jimbo S, Hibi Y, Kobayashi N, Yamamoto H, Seko T, Miura K (2006) Cu/Zn- and Mn-superoxide dismutase distribution and concentration in adrenal tumors. J Surg Res 135:150–155. doi:10.1016/j.jss.2006.03.027
Janssen AM, Bosman CB, Sier CF, Griffioen G, Kubben FJ, Lamers CB, van Krieken JH, van de Velde CJ, Verspaget HW (1998) Superoxide dismutases in relation to the overall survival of colorectal cancer patients. Br J Cancer 78:1051–1057
Janssen AM, Bosman CB, van Duijn W, Oostendorp-van de Ruit MM, Kubben FJ, Griffioen G, Lamers CB, van Krieken JH, van de Velde CJ, Verspaget HW (2000) Superoxide dismutases in gastric and esophageal cancer and the prognostic impact in gastric cancer. Clin Cancer Res 6:3183–3192
Johnson F, Giulivi C (2005) Superoxide dismutases and their impact upon human health. Mol Aspects Med 26:340–352. doi:10.1016/j.mam.2005.07.006
Kalen AL, Sarsour EH, Venkataraman S, Goswami PC (2006) Mn-superoxide dismutase overexpression enhances G2 accumulation and radioresistance in human oral squamous carcinoma cells. Antioxid Redox Signal 8:1273–1281. doi:10.1089/ars.2006.8.1273
Kim JJ, Chae SW, Hur GC, Cho SJ, Kim MK, Choi J, Nam SY, Kim WH, Yang HK, Lee BL (2002–2003) Manganese superoxide dismutase expression correlates with a poor prognosis in gastric cancer. Pathobiology 70:353–360. doi:10.1159/000071276
Kinnula VL, Crapo JD (2004) Superoxide dismutases in malignant cells and human tumors. Free Radic Biol Med 36:718–744. doi:10.1016/j.freeradbiomed.2003.12.010
Klaunig JE, Kamendulis LM (2004) The role of oxidative stress in carcinogenesis. Annu Rev Pharmacol Toxicol 44:239–267. doi:10.1146/annurev.pharmtox.44.101802.121851
Kwee JK, Mitidieri E, Affonso OR (1991) Lowered superoxide dismutase in highly metastatic B16 melanoma cells. Cancer Lett 57:199–202. doi:10.1016/0304-3835(91)90157-D
La Vecchia C, Lucchini F, Negri E, Levi F (2004) Trends in oral cancer mortality in Europe. Oral Oncol 40:433–439. doi:10.1016/j.oraloncology.2003.09.013
Landriscina M, Remiddi F, Ria F, Palazzotti B, De Leo ME, Iacoangeli M, Rosselli R, Scerrati M, Galeotti T (1996) The level of MnSOD is directly correlated with grade of brain tumours of neuroepithelial origin. Br J Cancer 74:1877–1885
Li JJ, Oberley LW, St Clair DK, Ridnour LA, Oberley TD (1995) Phenotypic changes induced in human breast cancer cells by overexpression of manganese-containing superoxide dismutase. Oncogene 10:1989–2000
Li N, Oberley TD, Oberley LW, Zhong W (1998) Overexpression of manganese superoxide dismutase in DU145 human prostate carcinoma cells has multiple effects on cell phenotype. Prostate 35:221–233. doi:10.1002/(SICI)1097-0045(19980515)35:3<221::AID-PROS8>3.0.CO;2-J
Liao CT, Chang JT, Wang HM, Ng SH, Hsueh C, Lee LY, Lin CH, Chen IH, Huang SF, Cheng AJ, Yen TC (2007) Survival in squamous cell carcinoma of the oral cavity: differences between pT4 N0 and other stage IVA categories. Cancer 110:564–571. doi:10.1002/cncr.22814
Lo WY, Tsai MH, Tsai Y, Hua CH, Tsai FJ, Huang SY, Tsai CH, Lai CC (2006) Identification of over-expressed proteins in oral squamous cell carcinoma (OSCC) patients by clinical proteomic analysis. Clin Chim Acta 376:101–107. doi:10.1016/j.cca.2006.06.030
McCarty KS Jr, Szabo E, Flowers JL, Cox EB, Leight GS, Miller L, Konrath J, Soper JT, Budwit DA, Creasman WT, Seigler HF, McCarty KS Sr (1986) Use of monoclonal anti-estrogen receptor antibody in the immunohistochemical evaluation of human tumors. Cancer Res 46:4244–4248
Morandini R, Boeynaems JM, Duhant X, Jacquemotte F, Kinnaert E, Ghanem G (1999) SODs are involved in the regulation of ICAM-1 expression in human melanoma and endothelial cells. Cell Mol Biol 45:1053–1063
Muramatsu H, Kogawa K, Tanaka M, Okumura K, Nishihori Y, Koike K, Kuga T, Niitsu Y (1995) Superoxide dismutase in SAS human tongue carcinoma cell line is a factor defining invasiveness and cell motility. Cancer Res 55:6210–6214
Nakano T, Oka K, Taniguchi N (1996) Manganese superoxide dismutase expression correlates with p53 status and local recurrence of cervical carcinoma treated with radiation therapy. Cancer Res 56:2771–2775
Nelson KK, Ranganathan AC, Mansouri J, Rodriguez AM, Providence KM, Rutter JL, Pumiglia K, Bennett JA, Melendez JA (2003) Elevated sod2 activity augments matrix metalloproteinase expression: evidence for the involvement of endogenous hydrogen peroxide in regulating metastasis. Clin Cancer Res 9:424–432
Petros JA, Baumann AK, Ruiz-Pesini E, Amin MB, Sun CQ, Hall J, Lim S, Issa MM, Flanders WD, Hosseini SH, Marshall FF, Wallace DC (2005) mtDNA mutations increase tumorigenicity in prostate cancer. Proc Natl Acad Sci USA 102:719–724. doi:10.1073/pnas.0408894102
Prior SL, Griffiths AP, Baxter JM, Baxter PW, Hodder SC, Silvester KC, Lewis PD (2006) Mitochondrial DNA mutations in oral squamous cell carcinoma. Carcinogenesis 27:945–950. doi:10.1093/carcin/bgi326
Ria F, Landriscina M, Remiddi F, Rosselli R, Iacoangeli M, Scerrati M, Pani G, Borrello S, Galeotti T (2001) The level of manganese superoxide dismutase content is an independent prognostic factor for glioblastoma: biological mechanisms and clinical implications. Br J Cancer 84:529–534. doi:10.1054/bjoc.2000.1594
Salzman R, Kankov K, Pcal L, Tomandl J, Horkov Z, Kostrica R (2007) Increased activity of superoxide dismutase in advanced stages of head and neck squamous cell carcinoma with locoregional metastases. Neoplasma 54:321–325
St Clair DK, Wan XS, Oberley TD, Muse KE, St Clair WH (1992) Suppression of radiation-induced neoplastic transformation by overexpression of mitochondrial superoxide dismutase. Mol Carcinog 6:238–242. doi:10.1002/mc.2940060404
Sudbo J, Reith A (2005) The evolution of predictive oncology and molecular-based therapy for oral cancer prevention. Int J Cancer 115:339–345. doi:10.1002/ijc.20896
Tanaka M, Kogawa K, Nishihori Y, Kuribayashi K, Nakamura K, Muramatsu H, Koike K, Sakamaki S, Niitsu Y (1997) Suppression of intracellular Cu-Zn SOD results in enhanced motility and metastasis of Meth A sarcoma cells. Int J Cancer 73:187–192. doi:10.1002/(SICI)1097-0215(19971009)73:2<187::AID-IJC4>3.0.CO;2-S
Valko M, Izakovic M, Mazur M, Rhodes CJ, Telser J (2004) Role of oxygen radicals in DNA damage and cancer incidence. Mol Cell Biochem 266:37–56. doi:10.1023/B:MCBI.0000049134.69131.89
Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M (2006) Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact 160:1–40. doi:10.1016/j.cbi.2005.12.009
Wenk J, Brenneisen P, Wlaschek M, Poswig A, Briviba K, Oberley TD, Scharffetter-Kochanek K (1999) Stable overexpression of manganese superoxide dismutase in mitochondria identifies hydrogen peroxide as a major oxidant in the AP-1-mediated induction of matrix-degrading metalloprotease-1. J Biol Chem 274:25869–25876. doi:10.1074/jbc.274.36.25869
Yan T, Oberley LW, Zhong W, St Clair DK (1996) Manganese-containing superoxide dismutase overexpression causes phenotypic reversion in SV40-transformed human lung fibroblasts. Cancer Res 56:2864–2871
Ye H, Wang A, Lee BS, Yu T, Sheng S, Peng T, Hu S, Crowe DL, Zhou X (2008) Proteomic based identification of manganese superoxide dismutase 2 (SOD2) as a metastasis marker for oral squamous cell carcinoma. Cancer Genomics Proteomics 5:85–94
Yeung BH, Wong KY, Lin MC, Wong CK, Mashima T, Tsuruo T, Wong AS (2008) Chemosensitisation by manganese superoxide dismutase inhibition is caspase-9 dependent and involves extracellular signal-regulated kinase 1/2. Br J Cancer 99:283–293. doi:10.1038/sj.bjc.6604477
Zelko IN, Mariani TJ, Folz RJ (2002) Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic Biol Med 33:337–349. doi:10.1016/S0891-5849(02)00905-X
Zhou A, Scoggin S, Gaynor RB, Williams NS (2003) Identification of NF-kappa B-regulated genes induced by TNFalpha utilizing expression profiling and RNA interference. Oncogene 22:2054–2064. doi:10.1038/sj.onc.1206262
Zhou S, Kachhap S, Sun W, Wu G, Chuang A, Poeta L, Grumbine L, Mithani SK, Chatterjee A, Koch W, Westra WH, Maitra A, Glazer C, Carducci M, Sidransky D, McFate T, Verma A, Califano JA (2007) Frequency and phenotypic implications of mitochondrial DNA mutations in human squamous cell cancers of the head and neck. Proc Natl Acad Sci USA 104:7540–7545. doi:10.1073/pnas.0610818104
Acknowledgments
We thank Lynda C. Charters for editing this manuscript. This study was partly supported by a Grant-in-Aid Scientific Research (No. 20791492) from the Ministry of Education, Culture, Sports, Science and Technology of Japan and Global COE Program (Global Center for Education and Research in Immune System Regulation and Treatment), MEXT, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
H. Yokoe, H. Nomura contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yokoe, H., Nomura, H., Yamano, Y. et al. Characterization of intracellular superoxide dismutase alterations in premalignant and malignant lesions of the oral cavity: correlation with lymph node metastasis. J Cancer Res Clin Oncol 135, 1625–1633 (2009). https://doi.org/10.1007/s00432-009-0610-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-009-0610-8