Abstract
Purpose
p33ING1b, as a candidate tumour suppressor gene, has been found to be expressed a proportion of oral squamous cell carcinomas (OSCCs), however, its clinicopathological significance is not studied yet. Our aim was to investigate association of p33ING1b expression with clinicopathological variables and particularly interesting new cysteine–histidine rich protein (PINCH) in OSCCs.
Methods
p33ING1b expression was immumohistochemically examined in 20 normal oral mucosa specimens and 49 OSCCs.
Results
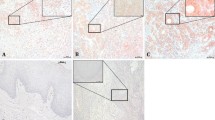
Normal squamous cells showed only p33ING1b nuclear expression (no cytoplasmic expression), with a rate of 90% positive cases. While 24% of OSCCs appeared cytoplasmic expression (11 of them with weak nuclear staining) and the rest tumours (76%) were negative for p33ING1b. Furthermore, the cases having lymph node metastasis showed a higher frequency of positive cytoplasmic expression than those without metastasis (P = 0.03). The p33ING1b cytoplasmic expression was positively related to PINCH expression (P = 0.04), the cases positive for both proteins had a high rate of the metastasis (P = 0.03).
Conclusions
The transfer of p33ING1b protein from the nucleus to the cytoplasm may result in loss of normal cellular function of the protein, which might play a role in the tumourigenesis and metastasis of OSCCs.



Similar content being viewed by others
Abbreviations
- OSCC:
-
Oral squamous cell carcinoma
- PINCH:
-
Particularly interesting new cysteine–histidine rich protein
References
Bosari S, Viale G, Bossi P, Maggioni M, Coggi G, Murray J, Lee AK (1994) Cytoplasmic accumulation of p53 protein: an independent prognostic indicator in colorectal adenocarcinomas. J Natl Cancer Inst 86:681–687
Chen L, Lu W, Agrawal S, Zhou W, Zhang R, Chen J (1999) Ubiquitous induction of p53 in tumor cells by antisense inhibition of MDM2 expression. Mol Med 5:21–34
Flamini G, Curigliano G, Ratto C, Astone A, Ferretti G, Nucera P, Sofo L, Sgambato A, Boninsegna A, Crucitti F, Cittadini A (1996) Prognostic significance of cytoplasmic p53 overexpression in colorectal cancer. An immunohistochemical analysis. Eur J Cancer 32:802–806
Gao J, Arbman G, Reader A, Sun XF (2004) Expression of PINCH protein is an independent factor in colorectal cancer patients. Neoplasia 6:796–801
Garkavtsev I, Kazarov A, Gudkov A, Riabowol K (1996) Suppression of the novel growth inhibitor p33ING1 promotes neoplastic transformation. Nat Genet 14:415–420
Garkavtsev I, Grigorian IA, Ossovskaya VS, Chernov M V, Chumakov P M, Gudkov A V (1998) The candidate tumour suppressor p33ING1 cooperates with p53 in cell growth control. Nature 391:295–298
Gunduz M, Ouchida M, Fukushima K, Hanafusa H, Etani T, Nishioka S, Nishizaki K, Shimizu K (2000) Genomic structure of the human ING1 gene and tumor-specific mutations detected in head and neck squamous cell carcinomas. Cancer Res 60:3143–3146
Gunduz M, Nagatsuka H, Demircan K, Gunduz E, Cengiz B, Ouchida M, Tsujigiwa H, Yamachika E, Fukushima K, Beder L, Hirohata S, Ninomiya Y, Nishizaki K, Shimizu K, Nagai N (2005) Frequent deletion and down-regulation of ING4, a candidate tumor suppressor gene at 12p13, in head and neck squamous cell carcinomas. Gene 356:109–117
Hoque MO, Kawamata H, Nakashiro K, Omotehara F, Hino S, Uchida D, Harada K, Begum NM, Yoshida H, Sato M, Fujimori T (2002) Dysfunction of the p53 tumor suppressor pathway in head and neck cancer. Int J Oncol 21:119–126
Klotzsche O, Etzrodt D, Hohenberg H, Bohn W, Deppert W (1998) Cytoplasmic retention of mutant tsp53 is dependent on an intermediate filament protein (vimentin) scaffold. Oncogene 16:3423–3434
Krishnamurthy J, Kannan K, Feng J, Mohanprasad BK, Tsuchida N, Shanmugam G (2001) Mutational analysis of the candidate tumor suppressor gene ING1 in Indian oral squamous cell carcinoma. Oral Oncol 37:222–224
Nouman GS, Anderson JJ, Lunec J, Angus B (2003) The role of the tumour suppressor p33 ING1b in human neoplasia. J Clin Pathol 56:491–496
Nouman GS, Anderson JJ, Mathers ME, Leonard N, Crosier S, Lunec J, Angus B (2002a) Nuclear to cytoplasmic compartment shift of the p33ING1b tumour suppressor protein is associated with malignancy in melanocytic lesions. Histopathology 40:360–366
Nouman GS, Anderson JJ, Wood KM, Lunec J, Hall AG, Reid MM, Angus B (2002b) Loss of nuclear expression of the inhibitor of growth p33ING1b in childhood acute lymphoblastic leukaemia. J Clin Pathol 55:596–601
Ohgi T, Masaki T, Nakai S, Morishita A, Yukimasa S, Nagai M, Miyauchi Y, Funaki T, Kurokohchi K, Watanabe S, Kuriyama S (2002) Expression of p33(ING1) in hepatocellular carcinoma: relationships to tumour differentiation and cyclin E kinase activity. Scand J Gastroenterol 37:1440–1448
Oki E, Maehara Y, Tokunaga E, Kakeji Y, Sugimachi K (1999) Reduced expression of p33(ING1) and the relationship with p53 expression in human gastric cancer. Cancer Lett 147:157–162
Rearden A (1994) A new LIM protein containing an autopitope homologous to “senescent cell antigen”. Biochem Biophys Res Commun 201:1124–1134
Scott M, Bonnefin P, Vieyra D, Boisvert FM, Young D, Bazett-Jones DP, Riabowol K (2001) UV-induced binding of ING1 to PCNA regulates the induction of apoptosis. J Cell Sci 114:3455–3462
Sun XF, Carstensen JM, Zhang H, Stal O, Wingren S, Hatschek T, Nordenskjold B (1992) Prognostic significance of cytoplasmic p53 oncoprotein in colorectal adenocarcinoma. Lancet 340:1369–1373
Sun XF, Zhang H, Carstensen J, Jansson A, Nordenskjold B (1997) Heat shock protein 72/73 in relation to cytoplasmic p53 expression and prognosis in colorectal adenocarcinomas. Int J Cancer 74:600–604
Tachibana M, Shinagawa Y, Kawamata H, Omotehara F, Horiuchi H, Ohkura Y, Kubota K, Imai Y, Fujibayashi T, Fujimori T (2002) RT-PCR amplification of RNA extracted from formalin-fixed, paraffin-embedded oral cancer sections: analysis of p53 pathway. Anticancer Res 23:2891–2896
Tao W, Levine AJ (1999) Nucleocytoplasmic shuttling of oncoprotein Hdm-2 is required for Hdm2-mediated degradation of p53. Proc Natl Acad Sci USA 96:3077–3080
Toyama T, Iwase H, Watson P, Muzik H, Saettler E, Magliocco A, DiFrancesco L, Forsyth P, Garkavtsev I, Kobayashi S, Riabowol K (1999) Suppression of ING1 expression in sporadic breast cancer. Oncogene 8:5187–5193
Tu Y, Li F,Giocoechea S, Wu C (1999) The LIM-only protein PINCH directly interacts with intergrin-linked kinase and is recruited to inegrin-rich sites in spreading cells. Mol Cell Biol 19:2425–2434
Vieyra D, Senger DL, Toyama T, Muzik H, Brasher PM, Johnston RN, Riabowol K, Forsyth PA (2003) Altered subcellular localization and low frequency of mutations of ING1 in human brain tumors. Clin Cancer Res 9:5952–5961
Wang-Rodriguez J, Dreilinger AD, Alsharabi GM, Rearden A (2002) The signaling adapter protein is up-regulated in he stroma of common cancess, notably at invasive edges. Cancer 95:1387–1395
Zhang JT, Li QX, Wang DW, Zhu ZL, Yang YH, Cui DS, Wang MW, Sun XF (2005) Up regulation of PINCH in the stroma of oral squamous cell carcinoma predicts nodal metastasis. Oncol Rep 14:1519–1522
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, JT., Wang, DW., Li, QX. et al. Nuclear to cytoplasmic shift of p33ING1b protein from normal oral mucosa to oral squamous cell carcinoma in relation to clinicopathological variables. J Cancer Res Clin Oncol 134, 421–426 (2008). https://doi.org/10.1007/s00432-007-0305-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-007-0305-y