Abstract
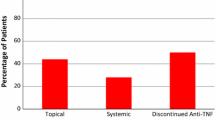
Studies showing a substantial frequency of dermatologic complications in paediatric Crohn’s disease (CD) patients on anti-tumour necrosis factor (TNF) therapy preferentially include patients treated with infliximab. We aimed to identify risk factors for the cumulative incidence of skin complications in a paediatric cohort receiving either adalimumab or infliximab and found an association between current skin complications and the patient’s current clinical condition. This study retrospectively evaluated dermatologic complications in an inception cohort of 100 paediatric CD patients receiving the first anti-TNF (Motol PIBD cohort). Patient data were collected every 3 months. The lesions were classified as psoriatic, atopic dermatitis, or others. We used Cox regression to evaluate the association between predefined variables and the time to complication and a generalised linear mixed model to assess the association between the patient’s current condition and the occurrence of complications. Among the 89 included children, 35 (39%) presented with dermatologic lesions. The only predictor associated with any complication was infliximab (versus adalimumab) therapy (hazard ratio [HR]: 2.07; 95% confidence interval [CI]: 1.03–4.17; p = 0.04). Infliximab therapy (HR: 5.5; 95%CI: 1.59–19.06; p = 0.01) and a family history of atopy (HR: 3.4; 95%CI 1.35–8.57, p = 0.002) were associated with early manifestation of atopic dermatitis. Lower C-reactive protein levels (odds ratio [OR], 0.947; 95% CI, − 0.898 to 0.998; p = 0.046) and infliximab (versus adalimumab) were associated with the occurrence of any dermatologic complications (OR, 5.93; 95% CI, 1.59–22.07; p = 0.008).
Conclusion: The frequency of skin complications seems high in paediatric CD patients treated with anti-TNF and is even higher in those treated with infliximab.
What is Known: •The dermatologic complications occur during treatment with anti-tumour necrosis factor. •The frequency of skin complications in paediatric patients with Crohn’s disease is high. | |
What is New: •Infliximab (vs. adalimumab) was identified as a strong risk factor for the cumulative incidence of skin complications. •Lower C-reactive protein levels were associated with the current occurrence of dermatologic complications. |



Similar content being viewed by others
Data availability
N/A
Code availability
N/A
Abbreviations
- ADA:
-
Adalimumab
- Anti-TNF:
-
Anti-tumour necrosis factor
- CI:
-
Confidence interval
- CD:
-
Crohn’s disease
- CRP:
-
C-reactive protein
- f-CPT:
-
F-calprotectin
- HR:
-
Hazards ratio
- IFX:
-
Infliximab
- IQR:
-
Interquartile range
- OR:
-
Odds ratio
- wPCDAI:
-
Crohn’s disease activity index
References
Hyams J, Crandall W, Kugathasan S, Griffiths A, Olson A, Johanns J, Liu G, Travers S, Heuschkel R, Markowitz J, Cohen S, Winter H, Veereman-Wauters G, Ferry G, Baldassano R (2007) Induction and maintenance infliximab therapy for the treatment of moderate-to-severe Crohn’s disease in children. Gastroenterology 132(3):863–873; quiz 1165-1166. https://doi.org/10.1053/j.gastro.2006.12.003
Hyams JS, Griffiths A, Markowitz J, Baldassano RN, Faubion WA Jr, Colletti RB, Dubinsky M, Kierkus J, Rosh J, Wang Y, Huang B, Bittle B, Marshall M, Lazar A (2012) Safety and efficacy of adalimumab for moderate to severe Crohn’s disease in children. Gastroenterology 143(2):365–374 e362. https://doi.org/10.1053/j.gastro.2012.04.046
Jongsma M, Aardoom M, Cozijnsen M, van Pieterson M, de Meij T, Norbruis O, Groeneweg M, Wolters V, van Wering H, Hojsak I, Kolho KL, Hummel T, Stapelbroek J, van der Feen C, Rheenen P, van Wijk M, Teklenburg-Roord S, Escher J, Samsom J, Ridder L (2020) OP38 Top-down infliximab superior to step-up in children with Moderate-to-Severe Crohn’s disease: a multicentre randomised controlled trial. J Crohn's Colitis 14(Supplement_1):S039–S039. https://doi.org/10.1093/ecco-jcc/jjz203.037
van Rheenen PF, Aloi M, Assa A, Bronsky J, Escher JC, Fagerberg UL, Gasparetto M, Gerasimidis K, Griffiths A, Henderson P, Koletzko S, Kolho KL, Levine A, van Limbergen J, Martin de Carpi FJ, Navas-Lopez VM, Oliva S, de Ridder L, Russell RK, Shouval D, Spinelli A, Turner D, Wilson D, Wine E, Ruemmele FM (2020) The medical management of paediatric Crohn’s disease: an ECCO-ESPGHAN Guideline Update. J Crohns Colitis 15:171–194. https://doi.org/10.1093/ecco-jcc/jjaa161
Kirchgesner J, Lemaitre M, Carrat F, Zureik M, Carbonnel F, Dray-Spira R (2018) Risk of serious and opportunistic infections associated with treatment of inflammatory bowel diseases. Gastroenterology 155(2):337–346 e310. https://doi.org/10.1053/j.gastro.2018.04.012
Lichtenstein GR, Feagan BG, Cohen RD, Salzberg BA, Diamond RH, Price S, Langholff W, Londhe A, Sandborn WJ (2012) Serious infection and mortality in patients with Crohn’s disease: more than 5 years of follow-up in the TREAT registry. Am J Gastroenterol 107(9):1409–1422. https://doi.org/10.1038/ajg.2012.218
Freling E, Baumann C, Cuny JF, Bigard MA, Schmutz JL, Barbaud A, Peyrin-Biroulet L (2015) Cumulative incidence of, risk factors for, and outcome of dermatological complications of anti-TNF therapy in inflammatory bowel disease: a 14-year experience. Am J Gastroenterol 110(8):1186–1196. https://doi.org/10.1038/ajg.2015.205
Cleynen I, Van Moerkercke W, Billiet T, Vandecandelaere P, Vande Casteele N, Breynaert C, Ballet V, Ferrante M, Noman M, Assche GV, Rutgeerts P, van den Oord JJ, Gils A, Segaert S, Vermeire S (2016) Characteristics of skin lesions associated with anti-tumor necrosis factor therapy in patients with inflammatory bowel disease: a cohort study. Ann Intern Med 164(1):10–22. https://doi.org/10.7326/M15-0729
Sridhar S, Maltz RM, Boyle B, Kim SC (2018) Dermatological manifestations in pediatric patients with inflammatory bowel diseases on anti-TNF therapy. Inflamm Bowel Dis 24:2086–2092. https://doi.org/10.1093/ibd/izy112
Malkonen T, Wikstrom A, Heiskanen K, Merras-Salmio L, Mustonen H, Sipponen T, Kolho KL (2014) Skin reactions during anti-TNFalpha therapy for pediatric inflammatory bowel disease: a 2-year prospective study. Inflamm Bowel Dis 20(8):1309–1315. https://doi.org/10.1097/MIB.0000000000000088
Courbette O, Aupiais C, Viala J, Hugot JP, Louveau B, Chatenoud L, Bourrat E, Martinez-Vinson C (2019) Infliximab paradoxical psoriasis in a cohort of children with inflammatory bowel disease. J Pediatr Gastroenterol Nutr 69:189–193. https://doi.org/10.1097/MPG.0000000000002349
Sherlock ME, Walters T, Tabbers MM, Frost K, Zachos M, Muise A, Pope E, Griffiths AM (2013) Infliximab-induced psoriasis and psoriasiform skin lesions in pediatric Crohn disease and a potential association with IL-23 receptor polymorphisms. J Pediatr Gastroenterol Nutr 56(5):512–518. https://doi.org/10.1097/MPG.0b013e31828390ba
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O'Neal L, McLeod L, Delacqua G, Delacqua F, Kirby J, Duda SN, Consortium RE (2019) The REDCap consortium: Building an international community of software platform partners. J Biomed Inform 95:103208. https://doi.org/10.1016/j.jbi.2019.103208
Team RC (2020) R: A language and environment for statistical computing R Foundation for Statistical Computing
Tillack C, Ehmann LM, Friedrich M, Laubender RP, Papay P, Vogelsang H, Stallhofer J, Beigel F, Bedynek A, Wetzke M, Maier H, Koburger M, Wagner J, Glas J, Diegelmann J, Koglin S, Dombrowski Y, Schauber J, Wollenberg A, Brand S (2014) Anti-TNF antibody-induced psoriasiform skin lesions in patients with inflammatory bowel disease are characterised by interferon-gamma-expressing Th1 cells and IL-17A/IL-22-expressing Th17 cells and respond to anti-IL-12/IL-23 antibody treatment. Gut 63(4):567–577. https://doi.org/10.1136/gutjnl-2012-302853
Nigam GB, Bhandare AP, Antoniou GA, Limdi JK (2020) Systematic review and meta-analysis of dermatological reactions in patients with inflammatory bowel disease treated with anti-tumour necrosis factor therapy. Eur J Gastroenterol Hepatol Publish Ahead of Print. https://doi.org/10.1097/MEG.0000000000001917
Eyerich S, Onken AT, Weidinger S, Franke A, Nasorri F, Pennino D, Grosber M, Pfab F, Schmidt-Weber CB, Mempel M, Hein R, Ring J, Cavani A, Eyerich K (2011) Mutual antagonism of T cells causing psoriasis and atopic eczema. N Engl J Med 365(3):231–238. https://doi.org/10.1056/NEJMoa1104200
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Funding
The project was supported by a grant from the Working Group of Pediatric Gastroenterology and Nutrition Associated with the Czech Pediatric Society of the Czech Medical Association of J. E. Purkyně, and by the Ministry of Health, Czech Republic, for the conceptual development of research organizations (00064203, University Hospital Motol, Prague, Czech Republic).
Author information
Authors and Affiliations
Contributions
All authors have made substantial contributions to the published work. OH: study design and data analysis, writing up of the first draft of the paper. DK: review of the literature, patient recruitment, data collection. IC: patient recruitment, data collection. KM: patient recruitment, data collection. TL: patient recruitment, data collection. KP: patient recruitment, data collection. MS: patient recruitment, data collection. KZ: patient recruitment, data collection. JB: responsible for leading the project team and revised the work critically for important intellectual content.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the local ethics committee.
Consent to participate
Informed consent was obtained from the parents of all patients.
Consent for publication
N/A
Conflict of interest
Ondrej Hradsky: Lectures/congress fees/consultancy (outside the submitted work)—MSD, AbbVie, Nutricia, Nestlé, Ferring, and Falk
Denis Kazeka: no conflicts of interest.
Ivana Copova: Lectures/congress fees/consultancy (outside the submitted work)—MSD, Nutricia, Nestlé
Katarina Mitrova: Lectures/congress fees/consultancy (outside the submitted work)—AbbVie, Takeda
Tereza Lerchova: Lectures/congress fees/consultancy (outside the submitted work)—Ferring, Nutricia, Biocodex
Kristyna Pospisilova: Lectures/congress fees/consultancy (outside the submitted work)—MSD, Nutricia, Nestlé a Mead Johnsons
Miroslava Sulovcova: no conflicts of interest
Kristyna Zarubova: Lectures/congress fees/consultancy (outside the submitted work)—Nestlé
Jiri Bronsky: Lectures/congress fees/consultancy (outside the submitted work)—MSD, AbbVie, Nutricia, Nestlé, Ferring, Biocodex, and Walmark
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hradsky, O., Kazeka, D., Copova, I. et al. Risk factors for dermatological complications of anti-TNF therapy in a cohort of children with Crohn’s disease. Eur J Pediatr 180, 3001–3008 (2021). https://doi.org/10.1007/s00431-021-04077-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-021-04077-0