Abstract
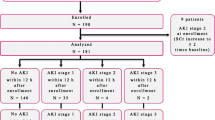
Early identification of patients with acute kidney injury (AKI) being at high risk for adverse outcome can influence medical treatment. This study compares urinary calprotectin, kidney injury molecule-1 (KIM-1), and neutrophil gelatinase-associated lipocalin (NGAL) for their performance in predicting mortality and need for renal replacement therapy (RRT) in pediatric AKI patients. Urinary biomarker concentrations were assessed prospectively in 141 subjects aged 0–18 years including 55 patients with established AKI according to pediatric Risk, Injury, Failure, Loss, and End-stage kidney disease (pRIFLE) criteria, 27 patients without AKI, and 59 healthy children. Within the AKI group, receiver operating characteristic (ROC) curve analysis revealed moderate to poor performance of calprotectin and KIM-1 in the prediction of 30-day mortality (calprotectin area under the curve (AUC) 0.55; KIM-1 AUC 0.55) and 3-month mortality (calprotectin AUC 0.61; KIM-1 AUC 0.60) and fair performance in the prediction of RRT requirement (calprotectin AUC 0.72; KIM-1 AUC 0.71). Urinary NGAL showed good performance in predicting 30-day (AUC 0.79) and 3-month (AUC 0.81) mortality and moderate performance in predicting RRT (AUC 0.61).
Conclusions: Whereas urinary calprotectin and KIM-1 can be useful for the prediction of RRT, urinary NGAL has a good diagnostic performance in predicting mortality in pediatric patients with AKI of heterogeneous etiology.
What is known: • There is increasing evidence that urinary biomarkers like neutrophil gelatinase-associated lipocalin (NGAL) and kidney injury molecule-1 (KIM-1) are valuable for the prediction of adverse outcome in adult acute kidney injury (AKI), whereas data on pediatric AKI is scarce. |
What is new: • Urinary calprotectin and KIM-1 do not predict mortality in our heterogeneous pediatric AKI cohort, but they show moderate performance in the prediction of dialysis. • Urinary NGAL is a good predictor of mortality performing better than pRIFLE stage, eGFR, or creatinine, but it shows moderate performance in the prediction of dialysis. |





Similar content being viewed by others
Abbreviations
- AKI:
-
acute kidney injury
- AUC:
-
area under the curve
- eCCl:
-
estimated creatinine clearance
- HUS:
-
hemolytic uremic syndrome
- KDIGO:
-
Kidney Disease: Improving Global Outcomes
- KIM-1:
-
kidney injury molecule-1
- NGAL:
-
neutrophil gelatinase-associated lipocalin
- pRIFLE:
-
pediatric Risk, Injury, Failure, Loss, and End-stage kidney disease
- ROC:
-
receiver operating characteristic
- RRT:
-
renal replacement therapy
- SCr:
-
serum creatinine
References
Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL (2007) Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int 71:1028–1035
Alge JL, Arthur JM (2015) Biomarkers of AKI: a review of mechanistic relevance and potential therapeutic implications. Clin J Am Soc Nephrol 10:147–155
Bojan M, Vicca S, Lopez-Lopez V, Mogenet A, Pouard P, Falissard B, Journois D (2014) Predictive performance of urine neutrophil gelatinase-associated lipocalin for dialysis requirement and death following cardiac surgery in neonates and infants. Clin J Am Soc Nephrol 9:285–294
Chang CH, Yang CH, Yang HY, Chen TH, Lin CY, Chang SW, Chen YT, Hung CC, Fang JT, Yang CW, Chen YC (2015) Urinary biomarkers improve the diagnosis of intrinsic acute kidney injury in coronary care units. Medicine (Baltimore) 94:e1703
Cruz DN, Bagshaw SM, Maisel A, Lewington A, Thadhani R, Chakravarthi R, Murray PT, Mehta RL, Chawla LS (2013) Use of biomarkers to assess prognosis and guide management of patients with acute kidney injury. Contrib Nephrol 182:45–64
Fan H, Zhao Y, Zhu JH, Song FC (2014) Urine neutrophil gelatinase-associated lipocalin in septic patients with and without acute kidney injury. Ren Fail 36:1399–1403
Fujiu K, Manabe I, Nagai R (2011) Renal collecting duct epithelial cells regulate inflammation in tubulointerstitial damage in mice. J Clin Invest 121:3425–3441
Gluba A, Banach M, Hannam S, Mikhailidis DP, Sakowicz A, Rysz J (2010) The role of Toll-like receptors in renal diseases. Nat Rev Nephrol 6:224–235
Heller F, Frischmann S, Grunbaum M, Zidek W, Westhoff TH (2011) Urinary calprotectin and the distinction between prerenal and intrinsic acute kidney injury. Clin J Am Soc Nephrol 6:2347–2355
Hjortrup PB, Haase N, Treschow F, Moller MH, Perner A (2015) Predictive value of NGAL for use of renal replacement therapy in patients with severe sepsis. Acta Anaesthesiol Scand 59:25–34
Jetton JG, Askenazi DJ (2012) Update on acute kidney injury in the neonate. Curr Opin Pediatr 24:191–196
Kellum JA, Lameire N, Group KAGW (2013) Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (part 1). Crit Care 17:204
Liangos O, Perianayagam MC, Vaidya VS, Han WK, Wald R, Tighiouart H, MacKinnon RW, Li L, Balakrishnan VS, Pereira BJ, Bonventre JV, Jaber BL (2007) Urinary N-acetyl-beta-(D)-glucosaminidase activity and kidney injury molecule-1 level are associated with adverse outcomes in acute renal failure. J Am Soc Nephrol 18:904–912
Macedo E, Mehta RL (2014) Targeting recovery from acute kidney injury: incidence and prevalence of recovery. Nephron Clin Pract 127:4–9
Mishra OP, Rai AK, Srivastava P, Pandey K, Abhinay A, Prasad R, Mishra RN, Schaefer F (2017) Predictive ability of urinary biomarkers for outcome in children with acute kidney injury. Pediatr Nephrol 32:521-527
Murray PT, Mehta RL, Shaw A, Ronco C, Endre Z, Kellum JA, Chawla LS, Cruz D, Ince C, Okusa MD, Workgroup A (2014) Potential use of biomarkers in acute kidney injury: report and summary of recommendations from the 10th Acute Dialysis Quality Initiative consensus conference. Kidney Int 85:513–521
Nickolas TL, Schmidt-Ott KM, Canetta P, Forster C, Singer E, Sise M, Elger A, Maarouf O, Sola-Del Valle DA, O'Rourke M, Sherman E, Lee P, Geara A, Imus P, Guddati A, Polland A, Rahman W, Elitok S, Malik N, Giglio J, El-Sayegh S, Devarajan P, Hebbar S, Saggi SJ, Hahn B, Kettritz R, Luft FC, Barasch J (2012) Diagnostic and prognostic stratification in the emergency department using urinary biomarkers of nephron damage: a multicenter prospective cohort study. J Am Coll Cardiol 59:246–255
Palazzuoli A, Ruocco G, Pellegrini M, De Gori C, Del Castillo G, Franci B, Nuti R, Ronco C (2015) Comparison of neutrophil gelatinase-associated lipocalin versus B-type natriuretic peptide and cystatin C to predict early acute kidney injury and outcome in patients with acute heart failure. Am J Cardiol 116:104–111
Ronco C (2014) Biomarkers for acute kidney injury: is NGAL ready for clinical use? Crit Care 18:680
Schmidt-Ott KM, Mori K, Li JY, Kalandadze A, Cohen DJ, Devarajan P, Barasch J (2007) Dual action of neutrophil gelatinase-associated lipocalin. J Am Soc Nephrol 18:407–413
Schrezenmeier EV, Barasch J, Budde K, Westhoff T, Schmidt-Ott KM (2017) Biomarkers in acute kidney injury—pathophysiological basis and clinical performance. Acta Physiol (Oxf) 219:554–572
Schwartz GJ, Munoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, Furth SL (2009) New equations to estimate GFR in children with CKD. J Am Soc Nephrol 20:629–637
Selewski DT, Charlton JR, Jetton JG, Guillet R, Mhanna MJ, Askenazi DJ, Kent AL (2015) Neonatal acute kidney injury. Pediatrics 136:e463–e473
Vogl T, Tenbrock K, Ludwig S, Leukert N, Ehrhardt C, van Zoelen MA, Nacken W, Foell D, van der Poll T, Sorg C, Roth J (2007) Mrp8 and Mrp14 are endogenous activators of Toll-like receptor 4, promoting lethal, endotoxin-induced shock. Nat Med 13:1042–1049
Waikar SS, Sabbisetti VS, Bonventre JV (2010) Normalization of urinary biomarkers to creatinine during changes in glomerular filtration rate. Kidney Int 78:486–494
Westhoff JH, Fichtner A, Waldherr S, Pagonas N, Seibert FS, Babel N, Tonshoff B, Bauer F, Westhoff TH (2016) Urinary biomarkers for the differentiation of prerenal and intrinsic pediatric acute kidney injury. Pediatr Nephrol 31:2353–2363
Yang HN, Boo CS, Kim MG, Jo SK, Cho WY, Kim HK (2010) Urine neutrophil gelatinase-associated lipocalin: an independent predictor of adverse outcomes in acute kidney injury. Am J Nephrol 31:501–509
Acknowledgements
We thank Simone Voigt for her indefatigable efforts in this study. We are indebted to all children and their parents who participated in the study.
Author information
Authors and Affiliations
Contributions
JHW, AF, and THW conceived and designed the experiments. JHW, SW and AF performed the experiments. JHW, AF, THW, BT, FSS, and FB analyzed the data or interpreted the results. JHW, AF, BT, and THW wrote the draft of the article. All other authors commented on the manuscript.
Corresponding author
Ethics declarations
Funding
The study was funded by the German Research Foundation (Research Unit FOR1368).
Conflict of interest
T.H.W. declares that patent “Assay method for intrinsic acute kidney injury” is granted. T.H.W. received speakers’ honoraria and/or research grants from Astellas, Novartis, Bristol-Myers Squibb, Pfizer, and Sanofi. All authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained by legal guardians of each patient with assent from the patient when appropriate for all individual participants included in the study.
Additional information
Communicated by Mario Bianchetti
Electronic supplementary material
Online Resource Table 1
Hosmer–Lemeshow goodness-of-fit test for biomarker calibration. (PDF 172 kb)
Online Resource Table 2
Urinary calprotectin, KIM-1, and NGAL in neonates and children stratified for pRIFLE stage. (PDF 42 kb)
Online Resource Table 3
Prognostic accuracy of urinary biomarker/urinary creatinine ratios in predicting mortality and need for RRT in pediatric AKI. (PDF 214 kb)
Online Resource Table 4
Prognostic accuracy of urinary calprotectin, KIM-1, and NGAL in predicting mortality and RRT in neonates and children. (PDF 46 kb)
Online Resource Table 5
Urinary calprotectin, KIM-1, and NGAL in the prediction of adverse outcomes in neonates classified by the neonatal-modified KDIGO definition of AKI. (PDF 39 kb)
Rights and permissions
About this article
Cite this article
Westhoff, J.H., Seibert, F.S., Waldherr, S. et al. Urinary calprotectin, kidney injury molecule-1, and neutrophil gelatinase-associated lipocalin for the prediction of adverse outcome in pediatric acute kidney injury. Eur J Pediatr 176, 745–755 (2017). https://doi.org/10.1007/s00431-017-2907-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-017-2907-y