Abstract
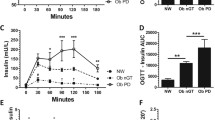
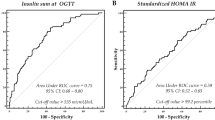
Available data on metabolically healthy obese (MHO) phenotype in children suggest that gender, puberty, waist circumference, insulin sensitivity, and other laboratory predictors have a role in distinguishing these children from metabolically unhealthy obese (MUO) youth. The goal of this study was to identify predictors of MHO phenotype and to analyze glucose and insulin metabolism during oral glucose tolerance test (OGTT) in MHO children. OGTT was performed in 244 obese children and adolescents aged 4.6–18.9 years. Subjects were classified as MHO in case of no fulfilled criterion of metabolic syndrome except anthropometry or as MUO (≥2 fulfilled criteria). Among the subjects, 21.7 % had MHO phenotype, and they were more likely to be female, younger, and in earlier stages of pubertal development, with lower degree of abdominal obesity. Insulin resistance was the only independent laboratory predictor of MUO phenotype (OR 1.59, CI 1.13–2.25), with 82 % sensitivity and 60 % specificity for diagnosing MUO using HOMA-IR cutoff point of ≥2.85. Although no significant differences were observed in glucose regulation, MUO children had higher insulin demand throughout OGTT, with 1.53 times higher total insulin secretion.
Conclusion: Further research is needed to investigate the possibility of targeted treatment of insulin resistance to minimize pubertal cross-over to MUO in obese children.
What is Known: • Substantial proportion of the obese youth (21–68 %) displays a metabolically healthy (MHO) phenotype. • Gender, puberty, waist circumference, insulin sensitivity, and lower levels of uric acid and transaminases have a possible role in distinguishing MHO from metabolically unhealthy obese (MUO) children. |
What is New: • Insulin resistance was found to be the only significant laboratory predictor of MUO when adjusted for gender, puberty, and the degree of abdominal obesity. • Besides basal insulin resistance, MUO children were found to have a significantly higher insulin secretion throughout OGTT in order to maintain glucose homeostasis. |

Similar content being viewed by others
Abbreviations
- BMI:
-
Body mass index
- GlucoseAUC :
-
Area under the glucose curve
- HOMA-IR:
-
Homeostatic model assessment of insulin resistance index
- IDF:
-
International Diabetes Federation
- InsulinAUC :
-
Area under the insulin curve
- ISI:
-
Matsuda insulin sensitivity index
- MHO:
-
Metabolically healthy obese
- MUO:
-
Metabolically unhealthy obese
- OGTT:
-
Oral glucose tolerance test
- SDS:
-
Standard deviation score
- WC:
-
Waist circumference
- WHO:
-
World Health Organization
References
Alberti K (1999) Deffinition, diagnosis and classification of diabetes mellitus and its complications. Report of a WHO consultation. Diabet Med 15:S39–S53
Black JA, White B, Viner RM, Simmons RK (2013) Bariatric surgery for obese children and adolescents: a systematic review and meta-analysis. Obes Rev 14:634–644
Bluher M (2010) The distinction of metabolically ‘healthy’ from ‘unhealthy’ obese individuals. Curr Opin Lipidol 21:38–43
Bluher S, Schwarz P (2014) Metabolically healthy obesity from childhood to adulthood—does weight status alone matter? Metabolism 63:1084–1092
Bokor S, Frelut ML, Vania A, Hadjiathanasiou CG, Anastasakou M, Malecka-Tendera E, Matusik P, Molnar D (2008) Prevalence of metabolic syndrome in European obese children. Int J Pediatr Obes 3(Suppl 2):3–8
Cali AM, Caprio S (2008) Obesity in children and adolescents. J Clin Endocrinol Metab 93:S31–36
Camhi SM, Waring ME, Sisson SB, Hayman LL, Must A (2013) Physical activity and screen time in metabolically healthy obese phenotypes in adolescents and adults. J Obes 2013:984613
de Onis M, Onyango AW, Borghi E, Siyam A, Nishida C, Siekmann J (2007) Development of a WHO growth reference for school-aged children and adolescents. Bull World Health Organ 85:660–667
Diagnosis and classification of diabetes mellitus (2010). Diabetes Care 33 Suppl 1:S62-69
Fernandez JR, Redden DT, Pietrobelli A, Allison DB (2004) Waist circumference percentiles in nationally representative samples of African-American, European-American, and Mexican-American children and adolescents. J Pediatr 145:439–444
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Hobkirk JP, King RF, Gately P, Pemberton P, Smith A, Barth JH, Harman N, Davies I, Carroll S (2013) The predictive ability of triglycerides and waist (hypertriglyceridemic waist) in assessing metabolic triad change in obese children and adolescents. Metab Syndr Relat Disord 11:336–342
Lemieux I, Poirier P, Bergeron J, Almeras N, Lamarche B, Cantin B, Dagenais GR, Despres JP (2007) Hypertriglyceridemic waist: a useful screening phenotype in preventive cardiology? Can J Cardiol 23(Suppl B):23B–31B
Levy-Marchal C, Arslanian S, Cutfield W, Sinaiko A, Druet C, Marcovecchio ML, Chiarelli F (2010) Insulin resistance in children: consensus, perspective, and future directions. J Clin Endocrinol Metab 95:5189–5198
Lobstein T, Baur L, Uauy R (2004) Obesity in children and young people: a crisis in public health. Obes Rev 5(Suppl 1):4–104
Mangge H, Zelzer S, Puerstner P, Schnedl WJ, Reeves G, Postolache TT, Weghuber D (2013) Uric acid best predicts metabolically unhealthy obesity with increased cardiovascular risk in youth and adults. Obesity (Silver Spring) 21:E71–77
Marshall WA, Tanner JM (1969) Variations in pattern of pubertal changes in girls. Arch Dis Child 44:291–303
Marshall WA, Tanner JM (1970) Variations in the pattern of pubertal changes in boys. Arch Dis Child 45:13–23
Matsuda M, DeFronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22:1462–1470
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419
Olshansky SJ, Passaro DJ, Hershow RC, Layden J, Carnes BA, Brody J, Hayflick L, Butler RN, Allison DB, Ludwig DS (2005) A potential decline in life expectancy in the United States in the 21st century. N Engl J Med 352:1138–1145
Primeau V, Coderre L, Karelis AD, Brochu M, Lavoie ME, Messier V, Sladek R, Rabasa-Lhoret R (2011) Characterizing the profile of obese patients who are metabolically healthy. Int J Obes 35:971–981
Prince RL, Kuk JL, Ambler KA, Dhaliwal J, Ball GD (2014) Predictors of metabolically healthy obesity in children. Diabetes Care 37:1462–1468
Reinehr T, Wolters B, Knop C, Lass N, Holl RW (2015) Strong effect of pubertal status on metabolic health in obese children: a longitudinal study. J Clin Endocrinol Metab 100:301–308
Senechal M, Wicklow B, Wittmeier K, Hay J, MacIntosh AC, Eskicioglu P, Venugopal N, McGavock JM (2013) Cardiorespiratory fitness and adiposity in metabolically healthy overweight and obese youth. Pediatrics 132:e85–92
The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents (2004). Pediatrics 114:555–576
Tsiros MD, Sinn N, Coates AM, Howe PR, Buckley JD (2008) Treatment of adolescent overweight and obesity. Eur J Pediatr 167:9–16. doi:10.1007/s00431-007-0575-z
Vukovic R, Mitrovic K, Milenkovic T, Todorovic S, Soldatovic I, Sipetic-Grujicic S, Zdravkovic D (2013) Insulin-sensitive obese children display a favorable metabolic profile. Eur J Pediatr 172:201–206
Weghuber D, Zelzer S, Stelzer I, Paulmichl K, Kammerhofer D, Schnedl W, Molnar D, Mangge H (2013) High risk vs. “metabolically healthy” phenotype in juvenile obesity—neck subcutaneous adipose tissue and serum uric acid are clinically relevant. Exp Clin Endocrinol Diabetes 121:384–390
WHO (2009) WHO AnthroPlus for personal computers: software for assessing growth of the world’s children and adolescents World Health Organization. http://www.who.int/growthref. Accessed January, 12 2012
Zachurzok-Buczynska A, Klimek K, Firek-Pedras M, Malecka-Tendera E (2011) Are metabolic syndrome and its components in obese children influenced by the overweight status or the insulin resistance? Endokrynol Pol 62:102–108
Zimmet P, Alberti KG, Kaufman F, Tajima N, Silink M, Arslanian S, Wong G, Bennett P, Shaw J, Caprio S (2007) The metabolic syndrome in children and adolescents—an IDF consensus report. Pediatr Diabetes 8:299–306
Zwiauer KF (2000) Prevention and treatment of overweight and obesity in children and adolescents. Eur J Pediatr 159(Suppl 1):S56–68
Acknowledgments
We gratefully acknowledge the kind assistance of the doctors and nurses of the Mother and Child Health Care Inistitute of Serbia “Dr Vukan Cupic.” This study was supported by the Ministry of Education, Science, and Technological Development of the Republic of Serbia through project no. 172026, 2011–2014.
Compliance with ethical standards
The authors declare that they have no conflict of interest. Informed consents were obtained for all participants and the study was approved by the Hospital Ethics Committee and performed in accordance with the 1964 Declaration of Helsinki and its later amendments.
Conflict of interest
The authors declare that they have no competing interests.
Authors’ contributions
Rade Vukovic designed the research study, conducted statistical analyses, and wrote the first draft of the manuscript. Tatjana Milenkovic, Katarina Mitrovic, Sladjana Todorovic, Ljiljana Plavsic, Ana Vukovic, and Dragan Zdravkovic contributed to study design and revised the manuscript. All authors reviewed and approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Beat Steinmann
Rights and permissions
About this article
Cite this article
Vukovic, R., Milenkovic, T., Mitrovic, K. et al. Preserved insulin sensitivity predicts metabolically healthy obese phenotype in children and adolescents. Eur J Pediatr 174, 1649–1655 (2015). https://doi.org/10.1007/s00431-015-2587-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-015-2587-4