Abstract
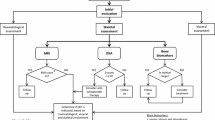
In individuals with non-neuronopathic Gaucher disease, childhood manifestations are usually predictive of a more severe phenotype. Although children with Gaucher disease are at risk of irreversible disease complications, early intervention with an optimal dose of enzyme therapy can prevent the development of complications and ensure adequate, potentially normal, development through childhood and adolescence. Very few, if any, children diagnosed by signs and symptoms should go untreated. Evidence suggests that disease severity, disease progression and treatment response in different organs where glucocerebroside accumulates are often non-uniform in affected individuals. Therefore, serial monitoring of the affected compartments is important. This should include a thorough physical examination at 6- to 12-monthly intervals. Neurological assessment should be performed to rule out neurological involvement and should be undertaken periodically thereafter in children who are considered to have risk factors for developing neuronopathic disease. Haematological and biochemical markers, such as haemoglobin, platelet counts and chitotriosidase levels, should be assessed every 3 months initially, but when clinical goals have been met through treatment with enzyme therapy, the frequency can be reduced to every 12 to 24 months. Careful monitoring of bone disease is vitally important, as the resulting sequelae are associated with the greatest level of morbidity. By combining various imaging modalities, the skeletal complications of non-neuronopathic Gaucher disease can be effectively monitored so that irreversible skeletal pathology is avoided and pain due to bone involvement is diminished or eliminated. Monitoring must include regular psychosocial, functional status and quality-of-life evaluation, as well as consistent assessment of therapeutic goal attainment and necessary dosage adjustments based on the patient’s progress. Conclusion: Through comprehensive and serial monitoring, ultimately, a therapeutic dose of enzyme therapy that achieves sustained benefits can be found for each child with non-neuronpathic Gaucher disease.

Similar content being viewed by others
Abbreviations
- ACE :
-
angiotensin-converting enzyme
- BMD :
-
bone mineral density
- DXA :
-
dual-energy X-ray absorptiometry
- QoL :
-
quality-of-life
- TRAP :
-
tartrate-resistant acid phosphatase
References
Aerts JMFG, Hollak CEM (1997) Plasma and metabolic abnormalities in Gaucher’s disease. Baillière’s Clin Haematol 10: 691–709
Allison JW, James CA, Arnold GL, Stine KC, Becton DL, Bell JM (1998) Reconversion of bone marrow in Gaucher disease treated with enzyme therapy documented by MR. Pediatr Radiol 28: 237–240
Ashkenazi A, Zaizov R, Matoth Y (1986) Effect of splenectomy on destructive bone changes in children with chronic (Type I) Gaucher disease. Eur J Pediatr 145: 138–141
Babyn PS, Ranson M, McCarville ME (1998) Normal bone marrow: signal characteristics and fatty conversion. Magn Reson Imaging Clin N Am 6: 473–495
Bamiou DE, Campbell P, Liasis A, Page J, Sirimanna T, Boyd S, Vellodi A, Harris C (2001) Audiometric abnormalities in children with Gaucher disease type 3. Neuropediatrics 32: 136–141
Barton NW, Brady RO, Dambrosia JM, Di Bisceglie AM, Doppelt SH, Hill SC, Mankin HJ, Murray GJ, Parker RI, Argoff CE (1991) Replacement therapy for inherited enzyme deficiency – macrophage targeted glucocerebrosidase for Gaucher’s disease. N Engl J Med 324: 1464–1470
Bembi B, Ciana G, Mengel E, Terk MR, Martini C, Wenstrup RJ (2002) Bone complications in children with Gaucher disease. Br J Radiol 75[Suppl 1]: A37–A44
Beutler E, Grabowski GA (2001) Gaucher disease. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic and molecular bases of inherited disease, 8th edn, vol III. McGraw-Hill, New York, pp 3635–3668
Boot RG, Renkema GH, Verhoek M, Strijland A, Bliek J, de Meulemeester TM, Mannens MM, Aerts JM (1998) The human chitotriosidase gene. Nature of inherited enzyme deficiency. J Biol Chem 273: 25680–25685
Casal JA, Lacerda L, Perez LF, Pinto RA, Clara Sa Miranda M, Carlos Tutor J (2002) Relationships between serum markers of monocyte/macrophage activation in type 1 Gaucher’s disease. Clin Chem Lab Med 40: 52–55
Cassidy L, Taylor D, Harris C (2000) Abnormal supranuclear eye movements in the child: a practical guide to examination and interpretation. Surv Ophthalmol 44: 479–506
Charrow J, Esplin JA, Gribble TJ, Kaplan P, Kolodny EH, Pastores GM, Scott CR, Wappner RS, Weinreb NJ, Wisch JS (1998) Gaucher disease: recommendations on diagnosis, evaluation, and monitoring. Arch Intern Med 158: 1754–1760
Charrow J, Andersson HC, Kaplan P, Kolodny EH, Mistry P, Pastores G, Rosenbloom BE, Scott CR, Wappner RS, Weinreb NJ, Zimran A (2000) The Gaucher Registry: demographics and disease characteristics of 1698 patients with Gaucher disease. Arch Intern Med 160: 2835–2843
Cohen IJ (2000) Managing Gaucher. Isr Med Assoc J 2: 260
Cohen IJ, Zaizov R (1998) Reply to a commentary by Elstein et al. Blood Cells Mol Dis 24: 306–308
Cohen IJ, Kornreich L, Mekhmandarov S, Katz K, Zaizov R (1996) Effective treatment of painful bone crises in type I Gaucher’s disease with high dose prednisolone. Arch Dis Child 75: 218–222
Cohen IJ, Katz K, Kornreich L, Horev G, Frish A, Zaizov R (1998) Low-dose high-frequency enzyme replacement therapy prevents fractures without complete suppression of painful bone crises in patients with severe juvenile onset type I Gaucher disease. Blood Cells Mol Dis 24: 296–302
Damiano AM, Pastores GM, Ware JE Jr (1998) The health-related quality of life of adults with Gaucher’s disease receiving enzyme replacement therapy: results from a retrospective study. Qual Life Res 7: 373–386
Ek M, Guijt M, Oosterveld P, Hollak CEM (1999) Quality of life in patients with Gaucher disease in the Netherlands (abstract). 3rd Meeting of the European Working Group on Gaucher Disease, Lemnos, Greece
Ellis KJ, Shypailo RJ, Hardin DS, Perez MD, Motil KJ, Wong WW, Abrams SA (2001) Z score prediction model for assessment of bone mineral content in pediatric diseases. J Bone Miner Res 16: 1658–1664
Erikson A (2001) Remaining problems in the management of patients with Gaucher disease. J Inherit Metab Dis 24[Suppl 2]: 122–126
Giraldo P, Pocovi M, Perez-Calvo J, Rubio-Felix D, Giralt M (2000) Report of the Spanish Gaucher’s disease registry: clinical and genetic characteristics. Haematologica 85: 792–799
Grabowski GA, Leslie N, Wenstrup R (1998) Enzyme therapy for Gaucher disease: the first 5 years. Blood Rev 12: 115–133
Grabowski GA, Andria G, Baldellou A, Campbell PE, Cohen IJ, Harris CM, Kaplan P, Mengel E, Pocovi M, Vellodi A (2003) Paediatric non-neuronopathic Gaucher disease: presentation, diagnosis and assessment. Eur J Pediatr (in press)
Harris CM, Taylor DSI, Vellodi A (1999) Ocular motor abnormalities in Gaucher disease. Neuropediatrics 30: 289–293
Hayes RP, Grinzald KA, Duffey EB, Elsas LJ II (1998) The impact of Gaucher disease and its treatment on quality of life. Qual Life Res 7: 521–534
Hermann G, Goldblat J, Levy RN, Goldsmith SJ, Desnick RJ, Grabowski GA (1986) Gaucher’s disease type I: assessment of bone involvement by CT and scintigraphy. AJR Am J Roentol 147: 943–948
Hicks CL, von Baeyer CL, Spafford PA, van Korlaar I, Goodenough B (2001) The Faces Pain Scale-Revised: toward a common metric in pediatric pain measurement. Pain 93: 173–183
Hill SC, Damaska BM, Ling A, Patterson K, Di Bisceglie AM, Brady RO, Barton NW (1992) Gaucher disease: abdominal MR imaging findings in 46 patients. Radiology 184: 561–566
Hollak CE, Maas M, Aerts JM (2001) Clinically relevant therapeutic endpoints in type 1 Gaucher disease. J Inherit Metab Dis 24[Suppl 2]: 97–105
Iuliano-Burns S, Mirwald RL, Bailey DA (2001) Timing and magnitude of peak height velocity and peak tissue velocities for early, average, and late maturing boys and girls. Am J Human Biol 13: 1–8
Kaplan P, Mazur A, Manor O, Charrow J, Esplin J, Gribble TJ, Wappner RS, Wisch JS, Weinreb NJ (1996) Acceleration of retarded growth in children with Gaucher disease after treatment with alglucerase. J Pediatr 129: 149–153
Kaplan P, Andersson HC, Charrow J, Kolodny EH, Mistry P, Pastores GM, Rosenbloom BE, Scott CR, Wappner RS, Weinreb NJ, for the International Collaborative Gaucher Group (2001) Growth improvement in response to enzyme replacement therapy (ERT) among children with Gaucher disease: the Gaucher Registry (abstract). 51st Annual Meeting of the American Society of Human Genetics, San Diego, CA, October 2001
Katz K, Cohen IJ, Ziv N, Grunebaum M, Zaizov R, Yosipovitch Z (1987) Fractures in children who have Gaucher disease. J Bone Joint Surg Am 69: 1361–1370
Katz K, Mechlis-Frish S, Cohen IJ, Horev G, Zaizov R, Lubin E (1991) Bone scans in the diagnosis of bone crisis in patients who have Gaucher disease. J Bone Joint Surg Am 73: 513–517
Katz K, Sabato S, Horev G, Cohen IJ, Yosipovitch Z (1993) Spinal involvement in children and adolescents with Gaucher disease. Spine 18: 332–335
Kauli R, Zaizov R, Lazar L, Pertzelan A, Laron Z, Galatzer A, Phillip M, Yaniv Y, Cohen IJ (2000) Delayed growth and puberty in patients with Gaucher disease type 1: natural history and effect of splenectomy and/or enzyme replacement therapy. Isr Med Assoc J 2: 158–163
Kocher MS, Hall JE (2000) Surgical management of spinal involvement in children and adolescents with Gaucher’s disease. J Pediatr Orthop 20: 383–388
Maas M, Poll LW, Terk MR (2002) Imaging and quantifying skeletal involvement in Gaucher disease. Br J Radiol 75[Suppl 1]: A13–A24
Masek BJ, Sims KB, Bove CM, Korson MS, Short P, Norman DK (1999) Quality of life assessment in adults with type 1 Gaucher disease. Qual Life Res 8: 263–268
Moore SG, Dawson KL (1990) Red and yellow marrow in the femur: age related changes in appearance at MR imaging. Radiology 175: 219–223
Pastores GM, Patel MJ, Firooznia H (2000) Bone and joint complications related to Gaucher disease. Curr Rheumatol Rep 2: 175–180
Richards SM, Olson TA, McPherson JM (1993) Antibody response in patients with Gaucher disease after repeated infusions with macrophage-targeted glucocerebrosidase. Blood 82: 1402–1405
Rite S, Baldellou A, Giraldo P, Labarta JI, Giralt M, Rubio-Felix D, Guallar A, Perez-Calvo JI, Mayayo E, Ferrandez A, Pocovi M (2002) Insulin-like growth factors in childhood-onset Gaucher disease. Pediatr Res 52: 109–112
Rosenthal DI, Doppelt SH, Mankin HJ, Dambrosia JM, Xavier RJ, McKusick KA, Rosen BR, Baker J, Niklason LT, Hill SC (1995) Enzyme replacement therapy for Gaucher disease: skeletal responses to macrophage-targeted glucocerebrosidase. Pediatrics 96: 629–635
Vellodi A, Bembi B, de Villemeur TB, Collin-Histed T, Erikson A, Mengel E, Rolfs A, Tylki-Szymanska A, Neuronopathic Gaucher Disease Task Force of the European Working Group on Gaucher Disease (2001) Management of neuronopathic Gaucher disease: a European consensus. J Inherit Metab Dis 24: 319–327
Waitches G, Zawin JK, Poznanski AK (1994) Sequence and rate of bone marrow conversion in the femora of children as seen on MR imaging: are accepted standards accurate? AJR Am J Roentgenol 162: 1399–1406
Ware JE, Snow KK, Kosinski M, Gandek B (1976) SF-36 Health Survey Manual and Interpretation Guide. New England Medical Center, The Health Institute, Boston, Massachussets, USA
Zhao H, Bailey L, Ponce E, Leslie N, Grabowski, GA (2003) Correlations of clinical, chitotriosidase and neutralizing antibody titres during enzyme therapy in Gaucher disease. BCMD 30: 90–96
Acknowledgements
This publication was supported by an educational grant from Genzyme Corporation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baldellou, A., Andria, G., Campbell, P.E. et al. Paediatric non-neuronopathic Gaucher disease: recommendations for treatment and monitoring. Eur J Pediatr 163, 67–75 (2004). https://doi.org/10.1007/s00431-003-1363-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-003-1363-z