Abstract
Background
Transplacental passage of IgGs is impaired in HIV + pregnant women, possibly determining an inadequate immunological protection in their children. We aimed to determine the impact of maternal immunological IgG profile and immunoactivation status on the efficiency of transplacental passage of IgG subclasses in HIV + mothers.
Methods
16 mother/infants pairs were studied in Malawi. Mothers received antiretroviral therapy (ART) from the third trimester of pregnancy. Determinations of pre-ART levels of maternal sCD14, of IgG subclasses in mothers at delivery and in their 1-month-old infants, were performed using commercial ELISA kits.
Results
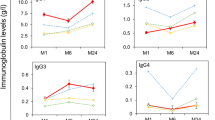
At delivery, after a median of 10 weeks of ART, 12/16 mothers were hypergammaglobulinemic, with IgG levels (20.5 mg/ml, 95% CI:18.8–26.8) directly correlated to the plasmatic levels of sCD14 (r = 0.640, p = 0.014). IgG1 levels (17.9 mg/ml) accounted for 82% of IgG, IgG3 and IgG4 levels were in the normal range. A profound deficit of IgG2 was observed both in mothers (0.60 mg/ml) and in infants (0.14 mg/ml). Placental transfer ratio (range 0.16–0.42) did not show a selective impairment between the different IgG subclasses. The transplacental passage of all IgG subclasses was decreased in the presence of maternal IgG over 16 mg/ml (significantly for IgG1, p = 0.031) and of high levels of sCD14 (p = 0.063).
Conclusions
Transplacental passage was reduced for all IgG subclasses and inversely correlated to high levels of maternal IgGs and to the degree of immunoactivation. The profound depression of IgG2 in mothers suggests that IgG2 neonatal levels mostly reflect the maternal deficit rather than a selective impairment of IgG2 transfer.



Similar content being viewed by others
References
Afran L, Garcia Knight M, Nduati E, Urban BC, Heyderman RS, Rowland-Jones SL (2014) HIV-exposed uninfected children: a growing population with a vulnerable immune system? Clin Exp Immunol 176:11–22. https://doi.org/10.1111/cei.12251
de Moraes-Pinto MI, Verhoeff F, Chimsuku L, Milligan PJ, Wesumperuma L, Broadhead RL, Brabin BJ, Johnson PM, Hart CA (1998) Placental antibody transfer: influence of maternal HIV infection and placental malaria. Arch Dis Child Fetal Neonatal 79:F202
Wilkinson AL, Pedersen SH, Urassa M, Michael D, Andreasen A, Todd J, Kinung’hi SM, Changalucha J, McDermid JM (2017) Maternal systemic or cord blood inflammation is associated with birth anthropometry in a Tanzanian prospective cohort. Trop Med Int Health 22:52–62. https://doi.org/10.1111/tmi.12799
Tanko RF, Soares AP, Müller TL, Garrett NJ, Samsunder N, Abdool Karim Q, Abdool Karim SS, Riou C, Burgers WA (2017) Effect of Antiretroviral therapy on the memory and activation profiles of B cells in HIV-infected African women. J Immunol 198:1220–1228. https://doi.org/10.4049/jimmunol.1601560
Ljunggren K, Broliden PA, Morfeldt-Manson L, Jondal M, Wahren B (1988) IgG subclass response to HIV in relation to antibody-dependent cellular cytotoxicity at different clinical stages. Clin Exp Immunol 73:343–347
Baum LL (2010) Role of humoral immunity in host defense against HIV. Curr HIV/AIDS Rep 7:11–18. https://doi.org/10.1007/s11904-009-0036-6
Palmeira P, Quinello C, Silveira-Lessa AL, Zago CA, Carneiro-Sampaio M (2012) IgG placental transfer in healthy and pathological pregnancies. Clin Dev Immunol 2012: 985646. https://doi.org/10.1155/2012/985646
Ruck C, Reikie BA, Marchant A, Kollmann TR, Kakkar F. Linking susceptibility to infectious diseases to immune system abnormalities among HIV-exposed uninfected infants. Front Immunol 7: 310. https://doi.org/10.3389/fimmu.2016.00310
de Moraes-Pinto MI, Almeida AC, Kenj G, Filgueiras TE, Tobias W, Santos AM, Carneiro-Sampaio MM, Farhat CK, Milligan PJ, Johnson PM, Hart CA (1996) Placental transfer and maternally acquired neonatal IgG immunity in HIV infection. J Infect Dis 173:1077–1084
Jones CE, Naidoo S, De Beer C, Esser M, Kampmann B, Hesseling AC (2011) Maternal HIV infection and antibody responses against vaccine-preventable diseases in uninfected infants. JAMA 305:576–584. https://doi.org/10.1001/jama.2011.100
Babakhanyan A, Ekali GL, Dent A, Kazura J, Nguasong JT, Fodjo BAY, Yuosembom EK, Esemu LF, Taylor DW, Leke RGF (2016) Maternal human immunodeficiency virus-associated hypergammaglobulinemia reduces transplacental transfer of immunoglobulin G to plasmodium falciparum antigens in Cameroonian neonates. Open Forum Infect Dis 3:ofw092. https://doi.org/10.1093/ofid/ofw092
Moro L, Bardají A, Nhampossa T, Mandomando I, Serra-Casas E, Sigaúque B, Cisteró P, Chauhan VS, Chitnis CE, Ordi J, Dobaño C, Alonso PL, Menéndez C, Mayor A (2015) Malaria and HIV infection in Mozambican pregnant women are associated with reduced transfer of antimalarial antibodies to their newborns. J Infect Dis 211:1004–101. https://doi.org/10.1093/infdis/jiu547
Farquhar C, Nduati R, Haigwood N, Sutton W, Mbori-Ngacha D, Richardson B, John-Stewart G (2005) High maternal HIV-1 viral load during pregnancy is associated with reduced placental transfer of measles IgG antibody. J Acquir Immune Defic Syndr 40:494–497
Gaensbauer JT, Rakhola JT, Onyango-Makumbi C, Mubiru M, Westcott JE, Krebs NF, Asturias EJ, Fowler MG, McFarland E, Janoff EN (2014) Impaired Haemophilus influenza type-b transplacental antibody transmission and declining antibody avidity through the first year of life represent potential vulnerabilities for HIV-exposed but uninfected infants. Clin Vaccine Immunol 21:1661–1667. https://doi.org/10.1128/CVI.00356-14
Baroncelli S, Galluzzo CM, Mancinelli S, Andreotti M, Jere H, Amici R, Marazzi MC, Vella S, Palombi L, Giuliano M (2016) Antibodies against pneumococcal capsular polysaccharide in Malawian HIV-positive mothers and their HIV-exposed uninfected children. Infect Dis 48:317–321. https://doi.org/10.3109/23744235.2015.1115895
Abu-Raya B, Smolen KK, Willems F, Kollmann TR, Marchant A (2016) Transfer of maternal antimicrobial immunity to HIV-exposed uninfected newborns. Front Immunol 31:338
Ben-Hur H, Gurevich P, Elhayany A, Avinoach I, Schneider DF, Zusman I (2005) Transport of maternal immunoglobulins through the human placental barrier in normal pregnancy and during inflammation. Int J Mol Med 16:401–407
Cumberland P, Shulman CE, Maple PA, Bulmer JN, Dorman EK, Kawuondo K, Marsh K, Cutts FTM (2007) Maternal HIV infection and placental malaria reduce transplacental antibody transfer and tetanus antibody levels in newborns in Kenya. J Infect Dis 196:550–557. https://doi.org/10.1086/519845
Wilcox CR, Holder B, Jones CE (2017) Factors affecting the FcRn-mediated transplacental transfer of antibodies and implications for vaccination in pregnancy. Front Immunol 2017 8:1294. https://doi.org/10.3389/fimmu.2017.01294
Vidarsson G, Gillian Dekkers G, Rispens T (2014) IgG Subclasses and allotypes: from structure to effector functions. Front Immunol 5:520. https://doi.org/10.3389/fimmu.2016.00338
Giuliano M, Andreotti M, Liotta G, Jere H, Sagno JB, Maulidi M, Mancinelli S, Buonomo E, Scarcella P, Pirillo MF, Amici R, Ceffa S, Vella S, Palombi L, Marazzi MC (2013) Maternal antiretroviral therapy for the prevention of mother-to-child transmission of HIV in Malawi: maternal and infant outcomes two years after delivery. PLoS One 8:e68950. https://doi.org/10.1371/journal.pone.0068950
McLean-Tooke A, OʼSullivan M, Easter T, Loh R (2013) Differences between total IgG and sum of the IgG subclasses in clinical samples. Pathology 45:675–677. https://doi.org/10.1097/PAT.0000000000000003
Bosire R, Farquhar C, Nduati R, Broliden K, Luchters S, Van de Perre P, De Vincenzi I, Merkel M, Wachuka V, Mbori-Ngacha D, John-Stewart G, Lohman-Payne B, Reilly M (2017) Higher transplacental pathogen-specific antibody transfer among pregnant women randomized to triple antiretroviral treatment versus short course zidovudine. Pediatr Infect Dis J. https://doi.org/10.1097/INF.0000000000001749. (Epub ahead of print)
Okoko BJ, Wesumperuma LH, Ota MO, Pinder M, Banya W, Gomez SF, McAdam KP, Hart AC (2001) The influence of placental malaria infection and maternal hypergammaglobulinemia on transplacental transfer of antibodies and IgG subclasses in a rural West African population. J Infect Dis 184:627–632. https://doi.org/10.1086/322808
Pirillo MF, Liotta G, Andreotti M, Jere H, Sagno JB, Scarcella P, Mancinelli S, Buonomo E, Amici R, Marazzi MC, Vella S, Palombi L, Giuliano M (2017) CMV infection in a cohort of HIV-exposed infants born to mothers receiving antiretroviral therapy during pregnancy and breastfeeding. Med Microbiol Immunol 206:23–29. https://doi.org/10.1007/s00430-016-0478-6
Freeman ML, Lederman MM, Sara Gianella S (2016) Partners in crime: the role of CMV in immune dysregulation and clinical outcome during HIV infection. Curr HIV/AIDS Rep 13:10–19. https://doi.org/10.1007/s11904-016-0297-9
Moir S, Fauci AS (2009) B cells in HIV infection and disease. Nat Rev Immunol 9:235–245. https://doi.org/10.1038/nri2524
Ruffin N, Thang PH, Rethi B, Nilsson A, Chiodi F (2011) The impact of inflammation and immune activation on B cell differentiation during HIV-1 infection. Front Immunol 2:90. https://doi.org/10.3389/fimmu.2011.00090
Brenchley JM, Price DA, Schacker TW, Asher TE, Silvestri G, Rao S, Kazzaz Z, Bornstein E, Lambotte O, Altmann D, Blazar BR, Rodriguez B, Teixeira-Johnson L, Landay A, Martin JN, Hecht FM, Picker LJ, Lederman MM, Deeks SG, Douek DC (2006) Microbial translocation is a cause of systemic immune activation in chronic HIV. infection Nat Med 12:1365–1371. https://doi.org/10.1038/nm1511
Hashira S, Okitsu-Negishi S, Yoshino K (2000) Placental transfer of IgG subclasses in a Japanese population. Pediatr Int 42:337–342
Schauer U, Stemberg F, Rieger CH, Borte M, Schubert S, Riedel F, Herz U, Renz H, Wick M, Carr-Smith HD, Bradwell AR, Herzog W (2003) IgG subclass concentrations in certified reference material 470 and reference values for children and adults determined with the binding site reagents. Clin Chem 49:1924–1929
Schur PH, Borel H, Gelfand EW, Alper CA, Rosen FS (1970) Selective gamma-g globulin deficiencies in patients with recurrent pyogenic infections. N Engl J Med 283:631–634. https://doi.org/10.1056/NEJM197009172831205
Zhang H, Li P, Wu D, Xu D, Hou Y, Wang Q, Li M, Li Y, Zeng X, Zhang F, Shi Q (2015) Serum IgG subclasses in autoimmune diseases. Medicine (Baltimore) 94:e387. https://doi.org/10.1097/MD.0000000000000387
Engelhart S, Glynn RJ, Schur PH (2017) Disease associations with isolated elevations of each of the four IgG subclasses. Semin Arthritis Rheum. https://doi.org/10.1016/j.semarthrit.2017.03.0211. (Epub ahead of print)
Weiss PJ, Wallace MR, Oldfield EC 3rd, O’Brien J, Janoff EN (1995) Response of recent human immunodeficiency virus seroconverters to the pneumococcal polysaccharide vaccine and Haemophilus influenzae type b conjugate vaccine. J Infect Dis 171:1217–1222
Reimer CB, Black CM, Holman RC, Wells TW, Ramirez RM, Sa-Ferreira JA, Nicholson JK, McDougal JS (1988) Hypergammaglobulinemia associated with human immunodeficiency virus infection. Monogr Allergy 23:83–96
De Milito A, Nilsson A, Titanji K, Thorstensson R, Reizenstein E, Narita M, Grutzmeier S, Sönnerborg A, Chiodi F (2004) Mechanisms of hypergammaglobulinemia and impaired antigen-specific humoral immunity in HIV-1 infection. Blood 103:2180–2186
Macchia D, Almerigogna F, Parronchi P, Ravina A, Maggi E, Romagnani S (1993) Membrane tumour necrosis factor-alpha is involved in the polyclonal B-cell activation induced by HIV-infected human T cells. Nature 363:464–466
Reuter MA, Pombo C, Betts MR (2012) Cytokine production and dysregulation in HIV pathogenesis: lessons for development of therapeutics and vaccines. Cytokine Growth Factor Rev 23:181–191. https://doi.org/10.1016/j.cytogfr.2012.05.005
Hosmalin A, Lebon P (2006) Type I interferon production in HIV-infected patients. J Leukoc Biol 80:984–993
Ferreira MU, Kimura EAS, Katzin AM, Santos-Neto LL, Ferrari JO, Villalobos JM, de Carvalho ME (1998) The IgG-subclass distribution of naturally acquired antibodies to Plasmodium falciparum, in relation to malaria exposure and severity. Ann Trop Med Parasitol 92:245–256
Tongren JE, Drakeley CJ, McDonald SL, Reyburn HG, Manjurano A, Nkya WM, Lemnge MM, Gowda CD, Todd JE, Corran PH, Riley EM (2006) Target antigen, age, and duration of antigen exposure independently regulate immunoglobulin G subclass switching in malaria. Infect Immun 74:257–264
Rouhani M, Zakeri S, Mehrizi AA, Djadid ND (2015) Comparative analysis of the profiles of IgG subclass-specific responses to Plasmodium falciparum apical membrane antigen-1 and merozoite surface protein-1 in naturally exposed individuals living in malaria hypoendemic settings. Iran Malar J 14:58. https://doi.org/10.1186/s12936-015-0547-0
National Malaria Control Programme (NMCP) [Malawi] and ICF International. 2014. Malawi Malaria Indicator Survey (MIS) 2014. Lilongwe, Malawi, and Rockville, Maryland, USA: NMCP and ICF International. http://dhsprogram.com/pubs/pdf/MIS18/MIS18.pdf. Accessed: Jan 2018
Lepage N, Huang SH, Nieuwenhuys E, Filler G (2010) Pediatric reference intervals for immunoglobulin G and its subclasses with Siemens immunonephelometric assays. Clin Biochem 43:694–696. https://doi.org/10.1016/j.clinbiochem.2010.02.003
Malek A (2003) Ex vivo human placenta models: transport of immunoglobulin G and its subclasses. Vaccine 21:3362–3364. https://doi.org/10.1097/PAT.0000000000000003
Simister NE (2003) Placental transport of immunoglobulin G. Vaccine 21:3365–3369. https://doi.org/10.1016/S0264-410X(03)00334-7
Parker A, Irure Ventura J, Sims D, Echeverría de Carlos A, Gómez de la Torre R, Tricas Aizpún L, Ocejo-Vinyals JG, López-Hoyos M, Wallis G, Harding S (2017) Measurement of the IgG2 response to Pneumococcal capsular polysaccharides may identify an antibody deficiency in individuals referred for immunological investigation. J Immunoassay Immunochem 38:514–522. https://doi.org/10.1080/15321819.2017.1340897
Hib and pneumococcal deaths among children 1–59 months of age, WHO 2008. http://www.who.int/immunization/monitoring_surveillance/burden/estimates/Pneumo_hib/en/ Accessed 12 August 2017
Heyderman RS, Madhi SA, French N, Cutland C, Ngwira B, Kayambo D, Mboizi R, Koen A, Jose L, Olugbosi M, Wittke F, Slobod K, Dull PM. Group B streptococcus vaccination in pregnant women with or without HIV in Africa: a non-randomised phase 2, open-label, multicentre trial. Lancet Infect Dis 2016 May;16(5):546–555. https://doi.org/10.1016/S1473-3099(15)00484-3
Dangor Z, Nunes MC, Kwatra G, Lala SG, Madhi SA (2017) Vaccination of HIV-infected pregnant women: implications for protection of their young infants. Trop Dis Travel Med Vaccines 2017 3:1. https://doi.org/10.1186/s40794-016-0044-7
Wilson C, Ebling R, Henig C, Adler T, Nicolaevski R, Barak M, Cazabon J, Maisin D, Lepoutre T, Gruson D, Hughes RG, Parker AR (2013) Significant, quantifiable differences exist between IgG subclass standards WHO67/97 and ERM-DA470k and can result in different interpretation of results. Clin Biochem 46:1751–1755. https://doi.org/10.1016/j.clinbiochem.2013.07.014
Pensieroso S, Cagigi A, Palma P, Nilsson A, Capponi C, Freda E, Bernardi S, Thorstensson R, Chiodi F, Rossi P (2009) Timing of HAART defines the integrity of memory B cells and the longevity of humoral responses in HIV-1 vertically-infected children. Proc Natl Acad Sci USA 106:7939–7944. https://doi.org/10.1073/pnas.0901702106
Moir S, Malaspina A, Ho J, Wang W, Dipoto AC, O’Shea MA, Roby G, Mican JM, Kottilil S, Chun TW, Proschan MA, Fauci AS (2008) Normalization of B cell counts and subpopulations after antiretroviral therapy in chronic HIV disease. J Infect Dis 197:572–579. https://doi.org/10.1086/526789
World Health Organization. WHO Case Definitions of HIV for Surveillance and Revised Clinical Staging and Immunological Classification of HIV-Related Disease in Adults. and Children; 2007. http://www.who.int/hiv/pub/guidelines/HIVstaging150307.pdf?ua=1. Accessed 5 Jan 2018
Acknowledgements
We thank Alessandra Mattei for providing secretarial help, Marco Mirra, Massimiliano Di Gregorio, Stefano Lucattini and Luca Fucili for IT support and Ferdinando Costa and Patrizia Cocco, for technical support. We are grateful to Giuseppe Lucania for the technical support with BN ProSpec® System Analyzer, and to all the mothers and their neonates who participated in our study.
Funding
This work was supported by a grant from the Ministry of Health, Rome, Italy (grant no. 3C04/1) and by Esther-Italy, Ministry of Health (grant no. 9M34).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study received ethics approval by the National Health Research Committee of Malawi (approval number #486).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Baroncelli, S., Galluzzo, C.M., Liotta, G. et al. Deficit of IgG2 in HIV-positive pregnant women is responsible of inadequate IgG2 levels in their HIV-uninfected children in Malawi. Med Microbiol Immunol 207, 175–182 (2018). https://doi.org/10.1007/s00430-018-0537-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-018-0537-2