Abstract
PA-X is a novel discovered accessory protein encoded by the PA mRNA. Our previous study demonstrated that PA-X decreases the virulence of a highly pathogenic H5N1 strain A/Chicken/Jiangsu/k0402/2010 in mice. However, the underlying mechanism of virulence attenuation associated with PA-X is still unknown. In this study, we compared two PA-X-deficient mutant viruses and the parental virus in terms of induction of pathology and manipulation of host response in the mouse lung, stimulation of cell death and PA nuclear accumulation. We first found that down-regulated PA-X expression markedly aggravated the acute lung injury of the infected mice early on day 1 post-infection (p.i.). We then determined that loss of PA-X expression induced higher levels of cytokines, chemokines and complement-derived peptides (C3a and C5a) in the lung, especially at early time point’s p.i. In addition, in vitro assays showed that the PA-X-deficient viruses enhanced cell death and increased expression of reactive oxygen species (ROS) in mammalian cells. Moreover, we also found that PA nuclear accumulation of the PA-X-null viruses accelerated in MDCK cells. These results demonstrate that PA-X decreases the level of complement components, ROS, cell death and inflammatory response, which may together contribute to the alleviated lung injury and the attenuation of the virulence of H5N1 virus in mice.
Similar content being viewed by others
Introduction
Influenza A virus (IAV) can infect diverse host species [1] and remains a severe threat to public health. Pathogenicity of IAV varies greatly among different virus subtypes due to its genetic variability. Moreover, IAV can translate multiple viral proteins from a single gene segment, which further complicates the pathogenesis of IAV. In 1970s, IAV genome was mapped for the first time and 10 viral proteins were indentified, namely PB2, PB1, PA, HA, NP, NA, M1, M2, NS1 and NS2 [2, 3]. Later, in 2001, the first accessory protein, PB1-F2, which was translated from the alternative open reading frame (ORF), was discovered [4]. Since then, another seven viral proteins, PB1-N40 [5], PA-X [6], M42 [7], NS3 [8], PA-N155 and PA-N182 [9] and PB2-S1 [10] have also been identified.
The PA-X protein is a 29-kD frameshifting product that encoded from the segment 3 of IAV genome [6]. Recently, many functions have been identified associated with this small protein, including modulation of viral replication [11–15] and pathogenicity [6, 12–14], suppression of the host antiviral and immune response [6, 11, 16–20] and endonucleolytic activity [21]. Jagger et al. [6] for the first time identified the PA-X protein and demonstrated that PA-X decreases the pathogenicity of 1918 H1N1 virus in mice and modulates the host response. Gao et al. [13, 14, 22] further confirmed the role of the PA-X protein in modulating virulence of influenza viruses in mice, including 2009 H1H1, avian H5N1 and H9N2 viruses. Besides the role in regulating viral virulence, the major function of PA-X is the host shutoff activity [6, 11, 16–20]. Desmet et al. [16] found that the PA-X protein contributes to the host cell shutoff and inhibits host antiviral response. Khaperskyy et al. [17] have verified that PA-X strongly inhibits the formation of the stress granule (SG, a host antiviral mechanism that detects the stress of virus infection) and arrests cap-dependent mRNA translation. Hayashi et al. [11] demonstrated that the PA-X protein has a strong impact on both the host innate and acquired immune response to influenza virus. Oishi et al. [18] further mapped the critical six basic amino acids in the C-terminal of PA-X that contribute to the PA-X shutoff activity.
Our previous study revealed that PA-X attenuates the pathogenicity of highly pathogenic avian influenza virus (HPAIV) of the H5N1 subtype in mice, ducks and chickens [15]. In this study, we aim to further investigate the mechanism of virulence attenuation caused by the PA-X protein in a mouse model. By systematically comparing the pathological changes in mouse lung, we found that reduced PA-X expression significantly aggravated the acute lung injury early on day 1 post-infection. The PA-X-deficient viruses induced higher production of cytokines, chemokines and complement components in the lung. We then showed that loss of PA-X expression enhanced cell death and increased the expression of reactive oxygen species (ROS) in cells. Moreover, accelerated PA nuclear accumulation in MDCK cells was also observed for PA-X-null mutants.
Materials and methods
Ethics statement
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the Ministry of Science and Technology of the People’s Republic of China. The protocols for animal experiments were approved by Jiangsu Administrative Committee for Laboratory Animals (approval number: SYXK-SU-2007-0005) and complied with the guidelines of Jiangsu laboratory animal welfare and ethics of Jiangsu Administrative Committee of Laboratory Animals. All experiments involving live viruses and animals were performed in negative-pressure isolators with HEPA filters in a biosafety level 3 (BSL3) animal facilities in accordance with the institutional biosafety manual.
Viruses and cells
A/Chicken/Jiangsu/k0402/2010 (CK10) was isolated from a dead chicken and it is highly pathogenic in mice, guinea pigs, chickens and mallard ducks [23]. To reduce the PA-X expression of the CK10 virus, recombinant viruses harboring mutations at the ribosomal frameshifting sites of the CK10 PA gene were generated (CK-PAX5 and CK-PAX3) [15]. The CK-PAX5 virus carries five nucleotide mutations (T568A, C569G, T573C, C574A and T576A, numbering from the A of the AUG codon) in the PA gene, and the CK-PAX3 virus harbors three mutations (T573C, C574A and T576A, numbering from the A of the AUG codon). The PA-X expression for the CK-PAX3 virus was 47 % relative to that of r-CK10 virus, while the corresponding value for the CK-PAX5 virus was 85 % [15]. Virulence test results indicate that the 50 % lethal dose of the mouse (MLD50) of CK-PAX3 (MLD50 = 100.5 EID50) and CK-PAX5 (MLD50 = 100.83 EID50) were 7.5- and 3.5-fold lower than that of r-CK10 (MLD50 = 101.375 EID50), respectively [15].
MDCK and A549 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) (Life technologies, Carlsbad, CA) supplemented with 10 % fetal calf serum (FCS) (Life technologies) and antibiotics and were incubated at 37 °C in 6 % CO2.
Animal experiments
Groups of 23 mice were infected intranasally (i.n.) with 105.0 50 % embryo infectious dose (EID50) of r-CK10, CK-PAX5, CK-PAX3 or with PBS as mock control. On days 1, 2 and 3 post-infections (p.i.), six mice per group were euthanized and the lungs were collected for histological observation, measurement of the levels of cytokines, chemokines and complements. The remaining mice were observed for clinical signs in a period of 2 weeks. Mice were humanely killed when they lost >25 % of their initial body weight.
Histopathological examination
Histopathological examination was performed as previously described [24]. Briefly, the collected lung tissues were fixed in 10 % phosphate-buffered formalin and processed for paraffin embedding. Then the tissue sections were stained with hematoxylin and eosin (H&E) and observed for histopathological changes. Images were captured with a Zeiss Axioplan 2IE epifluorescence microscope. Lesions were scored according to the following standards: 0, no visible changes; 1, mild interstitial pneumonia, edema and hemorrhage; 2, moderate interstitial pneumonia, mild bronchopneumonia and hemorrhage; 3, severe interstitial pneumonia and bronchopneumonia, severe hemorrhage, diffuse necrosis of the alveolar epithelial cell.
Quantitation of cytokines, chemokines and complement-derived peptides C3a and C5a
Levels of the selected cytokines and chemokines (IFN-α, IFN-β, IFN-γ, MX-1, IL-6, TNF-α, CXCL9, CXCL10, CXCL11, CCL3, CCL4 and CCL5) in the infected mouse lung were determined using ELISA kits (Bio-Swamp, Wuhan, CN) according to the manufacturer’s instructions. The production of the complement-derived peptides (C3a and C5a) was also measured using the ELISA method (Changjin Biological Technology Co., Ltd, Shanghai, CN). The absorbance of samples was measured at 450 nm using a 96-well microplate reader (Bio-Tek, Vermont, USA). Levels of cytokines, chemokines and complement-derived components were calculated based on the corresponding standard curves. All samples were measured in triplicate, and the experiments were repeated three times independently.
Cell death analysis
To explore the role of PA-X in virus-induced cell apoptosis, MDCK and A549 cells were inoculated with r-CK10, CK-PAX3 or CK-PAX5 at a MOI of 1. At 12 and 24 h p.i., both inoculated and non-inoculated cells were trypsinized and washed three times with PBS. Cells were then stained with FITC-conjugated annexin V and propidium iodide (PI) (Roche, Mannheim, Germany) for 15 min at room temperature in the dark. The stained cells were then analyzed using a flow cytometer (BD, San Jose, USA). Apoptotic cells are those with high annexin V but low PI staining, whereas necrotic cells are highly stained with both two dyes.
Measurement of the reactive oxygen species (ROS)
ROS levels were determined using the fluorescent marker 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA) (Beyotime, China) according to the manufacturer’s instructions. Briefly, MDCK cells were seeded in 24-well plates and infected with the indicated viruses at a MOI of 1. At 12 and 24 h p.i., cells were collected and washed twice with cold PBS. The cells were then centrifuged and re-suspended in 100 μl of DCFH-DA (10 μM/L) for incubation at 37 °C for 20 min. The cells were then washed twice with PBS. Precipitated cells were then suspended with 100 μl of PBS for flow cytometry analysis. Fluorescence intensity was analyzed with FACSAria flow cytometer using the FACSDiva software (Becton Dickinson Immunocytometry System) [25].
Immunofluorescence
MDCK cells were grown in 12-well plates and infected with the indicated viruses at a MOI of 2. At the indicated time points, cells were fixed with PBS containing 4 % paraformaldehyde for 20 min, saturated with PBS containing 0.5 % Triton X-100 for 10 min and then blocked with 10 % bovine serum albumin in PBS for 30 min. Cells were incubated with rabbit antiserum against PA (Genetex, TX, USA) at 37 °C for 1 h. Cells were washed three times with PBS and incubated at 37 °C for 1 h with fluorescein isothiocyanate (FITC)-coupled donkey anti-rabbit IgG secondary antibody (Santa Cruz Biotechnology, TX, USA). After incubation with secondary antibody, cells were washed three times with PBS and incubated with DAPI for 10 min. Cells were observed using a Leica fluorescence microscope. The accumulation of PA in the nuclei of infected cells was determined as the ratio of cells showing red fluorescence in the nucleus to the total number of cells counted (n = 100).
Statistical analysis
Statistical analysis was performed using the SPSS Statistics software. The independent samples T test was used for data analysis. p < 0.05 was considered as significant.
Results
PA-X-deficient mutant viruses aggravate the acute lung injury
We previously reported that loss of PA-X expression increases the virulence of the PA-X-deficient viruses (CK-PAX5 and CK-PAX3) in a mouse model [15]. The 50 % lethal dose in mice (MLD50) values of CK-PAX3 and CK-PAX5 were 7.5- and 3.5-fold lower than that of r-CK10, respectively. Moreover, mean death time (MDT) of the mice infected with CK-PAX3 was lower than that of r-CK10 virus. In this study, to investigate the potential role of PA-X in the pathogenesis, groups of mice were infected i.n. with 105.0 EID50 of the r-CK10, CK-PAX5 or CK-PAX3 virus, and mice were monitored for morbidity and mortality in a period of 2 weeks. As shown in Fig. 1, the PA-X-deficient viruses induced more severe weight loss compared with the parental virus r-CK10, especially the CK-PAX3 virus. In addition, both the PA-X mutant viruses-infected mice began to die as early as 4 days post-infection (p.i.), whereas the corresponding number for the r-CK10 virus was 6 days p.i. We then systematically compared the viral replication and histopathological changes in the mouse lung caused by these recombinants. As shown in Fig. 2f, on day 1 p.i., both the PA-X-deficient viruses replicated significantly higher viral titers than the r-CK10 virus. However, on day 2 and 3 p.i., the replication efficiency for all the viruses-infected groups was similar. Interestingly, on day 1 p.i., we also found that the pathological changes of the PA-X-deficient viruses-infected mice were more severe than that of r-CK10 virus. Notably, the CK-PAX3-infected mice exhibited severe interstitial pneumonia and bronchopneumonia that were characterized by serious thickened alveolar wall, disappeared alveolar structure, severe infiltration of necrotic alveolar epithelial cells and inflammatory cells in the alveolar interstitial and severe necrosis and desquamation of the bronchial epithelial cells (Fig. 2a). However, r-CK10 induced mild bronchopneumonia with the lesion of bronchial epithelial cells and few inflammatory cells infiltration (Fig. 2c). By statistical analysis, we found that the lung lesion score that induced by the CK-PAX3 virus was significantly higher than that of the parental virus (Fig. 2e). On day 2 and 3 p.i., there was no apparent difference in the histopathological changes between the PA-X-deficient viruses and the parental virus (Fig. 2e). Therefore, loss of PA-X expression alleviated the early acute lung injury (ALI) caused by highly pathogenic H5N1 virus.
Pathogenicity of recombinant viruses in 6-week-old BABL/c mice. a Mean weight loss of mice infected with 105.0 EID50 of r-CK10, CK-PAX5 and CK-PAX3 (n = 5). Mice were humanly killed when they lost ≥25 % of their initial body weight. Error bar represents stand deviation (SD). b Survival rate of mice infected with the indicated viruses or mock PBS (n = 5)
Representative histopathological changes in H&E (hematoxylin and eosin)-stained lung tissues on day 1 p.i. and viral replication in the mouse lung a CK-PAX3-infected mouse lung. Severe interstitial pneumonia and bronchopneumonia characterized by serious thickened alveolar wall, disappeared alveolar structure, severe infiltration of necrotic alveolar epithelial cells and inflammatory cells in the alveolar interstitial (shown as asterisk) and severe necrosis and desquamation of the bronchial epithelial cells (shown as). b CK-PAX5-infected mouse lung. Moderate interstitial pneumonia and bronchopneumonia with mild thickened alveolar wall (shown as triangle), necrosis and desquamation of the bronchial epithelial cells and accompany by infiltration of inflammatory cells (shown as black arrow) and inflammatory cells infiltration around the bronchus (shown as white arrow). c r-CK10-infected mouse lung. Mild bronchopneumonia characterized by necrosis and desquamation of the bronchial epithelial cells and a small quantity of inflammatory cells infiltration (shown as black arrow). d Mock control. No obvious histopathology was observed in the PBS-infected mouse lung. Bar = 100 μm. e Histological changes (expressed as scores). Value shown are the mean ± SD of the results from six individuals (*p < 0.05). f Viral replication in the mouse lung. Value shown are the mean ± SD of the results from six individuals (*p < 0.05). Asterisk indicates significant difference between the PA-X-deficient virus (CK-PAX3 or CK-PAX5) and the parental virus
PA-X-deficient viruses augment cytokine and chemokine response in the pneumonic lung
To further investigate the potential mechanism of different lung pathological manifestations caused by the recombinants and the parental virus, we then evaluated the role of PA-X in the inflammatory response by testing the production of some representative cytokines and chemokines in mouse lung (Fig. 3). In general, the mutants, especially the CK-PAX3 virus, induced a stronger cytokine and chemokine response than r-CK10. At day 1 p.i., compared to the parental virus, CK-PAX3 significantly elevated levels of CCL-3, CCL-4, CCL-5, CXCL-9, CXCL-10, IFN-α, IFN-β, IL-6 and TNF-α and CK-PAX5 significantly up-regulated the production of CCL-4, CCL-5, CXCL-9, IFN-β and IL-6. However, at day 2 and 3 p.i., the levels of the most tested cytokines and chemokines were comparable among all viruses. Nevertheless, significant differences in CXCL-10, IFN-β and TNF-α level between CK-PAX3 and r-CK10 were still observed at day 2 p.i. In addition, at day 3 p.i., the two mutant viruses still induced higher levels of IFN-γ, IL-6 and MX1 compared to r-CK10. Therefore, the PA-X-deficient viruses induced a stronger inflammatory response in the lung than the parental virus.
Cytokines and chemokines expression in the mouse lung. Groups of 18 6-week-old female BABL/c mice were intranasally infected with 105.0 EID50 of the indicated virus strain or received PBS intranasally (Control). On day 1, 2 and 3 p.i., six mice of each group were euthanized; the lungs were collected for homogenate preparation. Lung concentrations of cytokines were measured by ELISA. Cytokine or chemokine expression was expressed as the mean concentration ± SEM. *p < 0.05 and **p < 0.01, asterisk or double asterisk indicates significant difference between the PA-X-deficient virus (CK-PAX3 or CK-PAX5) and the parental virus
Reduced PA-X expression results in the excessive C3a and C5a activation in the lung
Substantial studies have shown that ALI occurring after HPAIV H5N1 virus infection is closely related to an abnormal host innate immune response [26–29]. Since the complement system plays a key role in the innate immunity and aberrant complement activation contributes to ALI caused by influenza virus [30–32], we then determined whether change in PA-X expression could affect the expression of complement-derived peptides C3a and C5a that are the central effectors of the complement system [30–37] (Fig. 4). The results showed that all the tested viruses induced higher level of C5a when compared to that of C3a in the mouse lung. In addition, on day 1, 2 and 3 p.i., the PA-X-deficient viruses activated C5a to significantly higher levels compared to r-CK10. The CK-PAX3 virus appeared as a hyperinducer of C3a compared to r-CK10 at all three time points, whereas CK-PAX5 only enhanced C3a expression at day 2 p.i. Therefore, these data demonstrated that the reduction of PA-X expression significantly activated the C3a and C5a expression.
C3a and C5a expression in the mouse lung. Groups of 18 6-week-old female BABL/c mice were intranasally infected with 105.0 EID50 of the indicated virus strain or received PBS intranasally (Mock). On day 1, 2 and 3 p.i., six mice of each group were euthanized; the lungs were collected for homogenate preparation. ELISA Kit was used to analyze the expression of the C3a and C5a complements. Data are presented as mean ± SEM. *p < 0.05 and **p < 0.01, when compared to the indicated group
PA-X-deficient viruses enhance cell death in MDCK and A549 cells
Since in vivo study showed great differences in lung pathological changes caused by these viruses, we then analyzed whether they differ in inducing cell damage because the degree of cell death is associated with the severity of tissue damage. At 12 and 24 h p.i., significantly more apoptotic cells were detected in cells inoculated with all tested viruses when compared to the mock control in MDCK and A549 cells (Fig. 5a, b, d, e). Moreover, in these two cell lines, the proportion of apoptotic cells was significantly higher in cells infected with CK-PAX3 than that in r-CK10-infected cells and CK-PAX5-infected cells at 12 h p.i. (Fig. 5a, b, d, e). At 24 h p.i., infection with both CK-PAX3 and CK-PAX5 produced significantly more apoptotic cells when compared to r-CK10 in MDCK and A549 cells (Fig. 5b, e).
Apoptosis and necrosis detected by annexin V and PI double staining in MDCK and A549 cells. At 12 and 24 h p.i., both infected and non-infected MDCK cells (a) and A549 cells (d) were harvested and stained with FITC annexin V and PI and then subjected to flow cytometry. Apoptotic cells are those with high annexin V and low PI staining, whereas necrotic cells are highly stained with both annexin V and PI. Values shown are the proportion of dead cells among the total number of cells analyzed. b–e Quantitative analysis of apoptosis and necrosis induced by the recombinants by flow cytometry in MDCK cells (b and c) and A549 cells (e and f). Values shown are the average proportion of apoptotic or necrotic cells of the total number of cells ± SD of the results from three independent experiments. *p < 0.05 and **p < 0.01, when compared to the indicated group
Regarding necrosis, in MDCK cells, only CK-PAX3 induced significantly more necrotic cells than the mock control at 24 h p.i. (Fig. 5c). In A549 cells, except for r-CK10 virus, all the viruses induced significantly more necrotic cells than the mock control both at 12 and 24 h p.i. (Fig. 5f). Moreover, a higher percentage of necrotic cells were observed for CK-PAX3 compared to r-CK10 at 12 h p.i. Taken together, these results showed that loss of PA-X expression significantly enhanced cell death both in MDCK and A549 cells.
PA-deficient viruses increase ROS expression in MDCK cells
Accumulated studies have shown that ROS contributes to the AIL and plays a role in influenza virus pathogenesis [38–47]. To evaluate the role of PA-X in ROS production, we compared the ROS expression level in MDCK cells inoculated with the parental virus and PA-X-deficient viruses. As shown in Fig. 6, at 12 and 24 h p.i., significantly higher expression levels of ROS were observed for all the tested viruses when compared to the mock control group. Moreover, at 24 h p.i., the two PA-X-deficient viruses induced significantly higher expression level of ROS than the parental virus. Therefore, loss of PA-X expression increased the ROS expression level in the process of influenza virus infection.
ROS expression in MDCK cells. ROS levels were determined using the fluorescent marker 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA) according to the manufacturer’s instructions. Fluorescence intensity was analyzed with a FACSAria flow cytometer with the FACSDiva software. Values shown are the average expression levels ± SD of the results from three independent experiments. *p < 0.05 and **p < 0.01, when compared to the indicated group
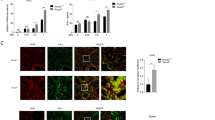
PA-X-deficient viruses enhance PA nuclear accumulation
To assess why reduced PA-X expression can increase viral replication [15], we next investigated the effect of mutations at the frameshifting sites on the PA nuclear accumulation. As shown in Fig. 7, the PA-X-deficient viruses and r-CK10 virus differed greatly in terms of PA nuclear accumulation. An earlier transportation of the PA protein from the cytoplasm to the nucleus was observed during PA-X-deficient virus infection. At 1 h p.i., the PA protein was detected in the nucleus of 6 % of the CK-PAX3-infected cells, whereas only 3 % of the r-CK10-infected cells displayed PA nuclear localization. At 6 h p.i., 5 % of the r-CK10-infected cells showed PA nuclear accumulation, whereas higher proportion of cells inoculated with CK-PAX5 (12 %) and CK-PAX3 (14 %) underwent nuclear translocation of PA. At 12 h p.i, the CK-PAX3-infected cells still showed higher levels of PA nuclear accumulation when compared to r-CK10. However, no significant differences were observed among the three viruses at 24 h p.i. These results indicated that loss of PA-X expression enhances PA nuclear accumulation that may contribute to the increased viral replication of the PA-X-deficient viruses [15].
PA nuclear accumulation of the recombinants in MDCK cells. MDCK cells were infected with r-CK10 (a), CK-PAX5 (b) or CK-PAX3 (c) at a MOI of 2; cell cultures were fixed and processed for immunofluorescence observation at the indicated times. Cell nuclei were stained with DAPI. d The PA nuclear accumulation in the infected cells was determined as the ratio of cells showing red fluorescence in the nucleus to the total number of cells counted. The values shown are mean ± SD of results for three independent experiments (*p < 0.05). Asterisk indicates significant difference between the PA-X-deficient virus CK-PAX3 or CK-PAX5 and the parental virus
Discussion
Our previous study has demonstrated that PA-X decreases the virulence of HPAIV of the H5N1 subtype in a mouse model [15]. In the present study, we further explored the potential role of the PA-X protein in the pathogenesis of H5N1 virus in this animal model. We first found that loss of PA-X expression aggravated the early ALI caused by the H5N1 virus infection (Fig. 2). We then found that the reduced PA-X expression resulted in increased levels of cytokines, chemokines and complements in the mouse lung (Figs. 3, 4). Moreover, the PA-X-deficient viruses induced a stronger cell death (Fig. 5) and higher ROS production (Fig. 6) in mammalian cells. We also found that loss of PA-X expression increased viral replication and enhanced PA nuclear accumulation (Figs. 1, 7). Taken together, our findings suggest that the PA-X protein may alleviate the acute lung injury caused by the lethal H5N1 AIVs through blunting the viral replication and virus-induced host innate immune response, especially the cytokine, complements, ROS and cell death response (Fig. 8).
Function and effects of the influenza A virus PA-X protein. This picture illustrates that the PA-X protein inhibits the influenza viral replication, virus-induced inflammation (including the cytokine and complement response, and inflammatory cells infiltration), cell death and ROS expression. The pleiotropic effects of PA-X protein contribute to the alleviation of the acute lung injury induced by influenza virus and the subsequently reduced mortality of the infected mouse
ALI that is manifested clinically as the acute respiratory distress syndrome (ARDS) is a common cause of mortality from severe influenza. HPAIV of H5N1 subtype often causes severe pneumonia that further results in ALI [48, 49] and subsequent death [26, 48]. The typical pathology of ALI in H5N1 influenza virus infection is characterized by rapid, massive inflammatory cell infiltration, edema formation and a marked increase in cytokines. In this study, we found that PA-X alleviated ALI caused by H5N1 HPAIV in mice (Fig. 2). However, the potential pathogenic mechanism for PA-X-associated attenuation of acute lung injury is currently unknown. Previous histological and pathological indicators strongly suggest a key role of an excessive host response in mediating ALI, and various effector arms of the host response can act deleteriously to initiate or exacerbate pathological damage in this viral pneumonia, including cytokines and chemokines [50–53], cell death [54–60], oxidative stress (ROS production) [38, 42–45], complement response [30–37] and other factors.
Using microarray analysis, Jagger et al. [6] reported that loss of PA-X expression markedly increases the expression level of the genes associated with the inflammatory and apoptotic pathways. In this study, by testing the expression levels of some representative cytokines and chemokines in the mouse lung, we demonstrated that cytokine/chemokine levels were greatly elevated in the PA-X-deficient virus-infected mouse lung compared with that of the parental virus (Fig. 3). To be noted of, early on day 1 p.i., the CK-PAX3 virus induced significantly higher expression level of CCL3, CCL4, CCL5, CXCL9, CXCL10, IFN-α, IFN-β, IL-6 and TNF-α when compared to the parental virus (Fig. 3). Although the innate immune response is critical for influenza virus clearance, an aberrant cytokine response often results in immunopathology and subsequent organ dysfunction and host death [50–53]. We also found that reduced PA-X expression significantly increased virus-induced apoptosis and necrosis in MDCK and A549 cells (Fig. 5). Previous studies have demonstrated that influenza virus-induced severe host cell apoptosis [54–60] contributes to the viral pathogenicity. Thus, we supposed that the accelerated cell apoptosis and massively increased cytokine response induced by the mutants aggravates the cellular damage and organ dysfunction, which may contribute to the severe ALI that caused by the PA-X-deficient viruses in mice.
Cellular metabolisms produce different varieties of reactive oxygen species (ROS) as by-products. Usually, these ROS play an important role in cell signaling, cytokines, growth factors, ion transport, transcription, neuromodulation and immunomodulation [61, 62]. However, an excessive production of ROS can lead to oxidative stress [63, 64] that is associated with several aspects of viral disease pathogenesis including apoptosis [65–67], viral replication [68–70], inflammatory response and dysfunction of immune system [71, 72]. During influenza virus infection, a rapid influx of inflammatory cells often results in an increased ROS production [73]. A number of studies have demonstrated that ROS plays a role in the occurrence of the acute lung injury induced by influenza virus [39, 40, 42, 44, 47] and it is an attractive and promising target for therapeutic intervention [38, 43, 45, 46, 74]. In this study, significantly higher expression level of ROS was detected in the PA-X-deficient virus-infected cells compared to the parental virus at 24 h p.i (Fig. 6). In addition, loss of PA-X expression induced significantly stronger cytokine response and cell death compared to the wild-type virus (Figs. 3 and 5). Therefore, we surmised that the increased level of ROS expression related to the reduced PA-X expression may further aggravate the inflammatory and cell death response, which contribute to the serious lung damage induced by the PA-X-deficient viruses.
Generally, the complement system (CS) plays an important role in the innate and adaptive immune response to pathogens [75–78]. However, aberrant complement activation contributes to the pathogenesis of many inflammatory and immunological diseases [79]. More importantly, accumulating studies have shown that the complement system has been implicated in the development of the acute lung injury induced by respiratory viruses, including influenza virus [30, 31, 37], severe acute respiratory syndrome coronavirus (SARS-Cov) [80] and the Middle East respiratory syndrome coronavirus (MERS-Cov) [81]. In the complement system, the complement-derived peptides C3a and C5a, products of the CS cascade, are the central effector molecules which contribute to the excessive inflammatory response induced by pathogens [30–37]. In this study, we showed that loss of PA-X expression significantly increased the expression levels of C3a and C5a at 24 h p.i. in the mouse lung, especially for the C5a level (Fig. 4). Substantial studies have also demonstrated that C5a mediates ALI through multiple biological activities, e.g., activation of inflammatory cells migration into lung tissue, generation of ROS, up-regulation of adhesion molecules, stimulation of ‘cytokine storm’ (reviewed in [32]). To be note of, we showed that the PA-X-deficient viruses triggered a stronger ROS and inflammatory response than the parental virus (Figs. 3, 6). Therefore, it is possible that the PA-X protein alleviates the lung injury partially by down-regulating the C3a and C5a expression and subsequently reduced ROS and cytokine production.
The polymerase of influenza virus has to enter into the nucleus of the infected cells to accomplish the transcription and replication of the viral genome. Previous studies have shown that the level of the polymerase nuclear accumulation is associated with the virulence of IAV. Huarte et al. reported that PA T157A mutation not only delays the PA nuclear accumulation but also decreases viral replication and virulence in mice [82]. Gabriel et al. [83] showed that the enhanced binding of importin α1 with the PB2 and NP proteins accelerates the nuclear transport of these proteins in mammalian cells and results in higher efficiency of viral replication. Hu et al. [84] also revealed that PA K237E mutation increases PA nuclear accumulation in DEF cells, which is associated with the enhanced viral replication and virulence in mallard ducks. Recently, Gao et al. [22] also found that PA-X reduces PA nuclear accumulation and inhibits viral replication in A549 cells. In the present study, we determined that the reduction of PA-X expression accelerates the transportation of the PA protein from the cytoplasm to the nucleus of the infected cell (Fig. 7). The faster nuclear accumulation of the PA protein may account for the increased virus replication and virulence of the PA-X-deficient virus [15]. Interestingly, Jagger et al. [6] found that PA-X have no effect on viral replication of the 1918 H1N1 virus in MDCK cells, embryonated eggs and mouse lung. However, we found that loss of PA-X expression not only increased viral replication of the highly pathogenic H5N1 virus in mammalian cells (including A549 cells, Vero and MDCK cells) but also enhanced viral replication in mouse organs [15]. Gao et al. [22] also showed that reduced PA-X expression strengthened viral replication of the pandemic 2009 H1N1 and highly pathogenic H5N1 virus in A549 cells and mouse lung. Currently, the underlying mechanism for the different observation in viral replication of the PA-X mutants is unclear. However, virus subtypes might account partially for this phenotype difference. Moreover, the potential mechanism of the PA-X protein in delaying transportation of the PA protein into the nucleus is currently unknown. One possible explanation is that the PA-X protein may affect the expression level of the critical cellular factors that are prerequisite for nuclear transportation system by host shutoff mechanism [6, 16, 17].
A large amount of studies have also demonstrated that another accessory protein, PB1-F2, acts as a virulence regulatory factor in the pathogenesis of influenza virus. Multifunctional roles have been attributed to this protein, including inducing cell death, increasing pathogenesis and cytokine dysfunction, enhancing secondary bacterial pneumonia and so on [Reviewed in 85–89]. However, apparently the PA-X protein behaves in an opposite way by decreasing cell death, diminishing cytokine and inflammatory response and decreasing the pathogenicity of H5N1 and H1N1 influenza viruses [6, 11, 12, 15] (see also Fig. 8). These two accessory proteins may act in concert with maintaining the balance of the pathogenicity of influenza virus. In the future, it is very interesting to investigate the effect of simultaneous deletion of the PA-X and PB1-F2 proteins on the pathogenesis and transmission of the highly pathogenic influenza virus.
In summary, our study clearly demonstrates that PA-X alleviates ALI caused by HPAIV of the H5N1 subtype in mice. Loss of PA-X expression accelerated the nuclear accumulation of the PA protein, aggravated the host cell death and augmented the ROS production, complements and cytokines response. Therefore, this study provides further information for understanding the role of PA-X, a new accessory protein, in the complicated pathogenesis of the influenza virus. Future studies are still needed to investigate the underlying mechanism of PA-X in regulating virulence, particularly in the viral–host interaction interface.
References
Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y (1992) Evolution and ecology of influenza A viruses. Microbiol Rev 56(1):152–179
Palese P (1977) The genes of influenza virus. Cell 10(1):1–10. doi:10.1016/0092-8674(77)90133-7
Lamb RA, Lai CJ (1980) Sequence of interrupted and uninterrupted mRNAs and cloned DNA coding for the two overlapping nonstructural proteins of influenza virus. Cell 21(2):475–485. doi:10.1016/0092-8674(80)90484-5
Chen W, Calvo PA, Malide D, Gibbs J, Schubert U, Bacik I, Basta S, O’Neill R, Schickli J, Palese P, Henklein P, Bennink JR, Yewdell JW (2001) A novel influenza A virus mitochondrial protein that induces cell death. Nat Med 7(12):1306–1312. doi:10.1038/nm1201-1306
Wise HM, Foeglein A, Sun J, Dalton RM, Patel S, Howard W, Anderson EC, Barclay WS, Digard P (2009) A complicated message: Identification of a novel PB1-related protein translated from influenza A virus segment 2 mRNA. J Virol 83(16):8021–8031. doi:10.1128/JVI.00826-09
Jagger BW, Wise HM, Kash JC, Walters KA, Wills NM, Xiao YL, Dunfee RL, Schwartzman LM, Ozinsky A, Bell GL, Dalton RM, Lo A, Efstathiou S, Atkins JF, Firth AE, Taubenberger JK, Digard P (2012) An overlapping protein-coding region in influenza A virus segment 3 modulates the host response. Science 337(6091):199–204. doi:10.1126/science.1222213
Wise HM, Hutchinson EC, Jagger BW, Stuart AD, Kang ZH, Robb N, Schwartzman LM, Kash JC, Fodor E, Firth AE, Gog JR, Taubenberger JK, Digard P (2012) Identification of a novel splice variant form of the influenza A virus M2 ion channel with an antigenically distinct ectodomain. PLoS Pathog 8(11):e1002998. doi:10.1371/journal.ppat.1002998
Selman M, Dankar SK, Forbes NE, Jia J-J, Brown EG (2012) Adaptive mutation in influenza A virus non-structural gene is linked to host switching and induces a novel protein by alternative splicing. Emerg Microb Infect 1(11):e42. doi:10.1038/emi.2012.38
Muramoto Y, Noda T, Kawakami E, Akkina R, Kawaoka Y (2013) Identification of novel influenza A virus proteins translated from PA mRNA. J Virol 87(5):2455–2462. doi:10.1128/JVI.02656-12
Yamayoshi S, Watanabe M, Goto H, Kawaoka Y (2015) Identification of A novel viral protein expressed from the PB2 segment of influenza A virus. J Virol. doi:10.1128/JVI.02175-15
Hayashi T, MacDonald LA, Takimoto T (2015) Influenza A virus protein PA-X contributes to viral growth and suppression of the host antiviral and immune responses. J Virol 89(12):6442–6452. doi:10.1128/JVI.00319-15
Alvarado-Facundo E, Gao Y, Ribas-Aparicio RM, Jimenez-Alberto A, Weiss CD, Wang W (2015) Influenza virus M2 protein ion channel activity helps to maintain pandemic 2009 H1N1 virus hemagglutinin fusion competence during transport to the cell surface. J Virol 89(4):1975–1985. doi:10.1128/JVI.03253-14
Gao HJ, Xu GL, Sun YP, Qi L, Wang JL, Kong WL, Sun HL, Pu J, Chang KC, Liu JH (2015) PA-X is a virulence factor in avian H9N2 influenza virus. J Gen Virol 96:2587–2594. doi:10.1099/jgv.0.000232
Gao H, Sun H, Hu J, Qi L, Wang J, Xiong X, Wang Y, He Q, Lin Y, Kong W, Seng LG, Pu J, Chang KC, Liu X, Liu J, Sun Y (2015) Twenty amino acids at the C-terminus of PA-X are associated with increased influenza A virus replication and pathogenicity. J Gen Virol 96(8):2036–2049. doi:10.1099/vir.0.000143
Hu J, Mo Y, Wang X, Gu M, Hu Z, Zhong L, Wu Q, Hao X, Hu S, Liu W, Liu H, Liu X (2015) PA-X decreases the pathogenicity of highly pathogenic H5N1 influenza A virus in avian species by inhibiting virus replication and host response. J Virol 89(8):4126–4142. doi:10.1128/JVI.02132-14
Desmet EA, Bussey KA, Stone R, Takimoto T (2013) Identification of the N-terminal domain of the influenza virus PA responsible for the suppression of host protein synthesis. J Virol 87(6):3108–3118. doi:10.1128/JVI.02826-12
Khaperskyy DA, Emara MM, Johnston BP, Anderson P, Hatchette TF, McCormick C (2014) Influenza a virus host shutoff disables antiviral stress-induced translation arrest. PLoS Pathog 10(7):e1004217. doi:10.1371/journal.ppat.1004217
Oishi K, Yamayoshi S, Kawaoka Y (2015) Mapping of a region of the PA-X protein of influenza A virus that is important for its shutoff activity. J Virol 89(16):8661–8665. doi:10.1128/Jvi.01132-15
Khaperskyy DA, McCormick C (2015) Timing is everything: coordinated control of host shutoff by influenza A virus NS1 and PA-X proteins. J Virol 89(13):6528–6531. doi:10.1128/Jvi.00386-15
Hayashi T, Chaimayo C, Takimoto T (2015) Impact of influenza PA-X on host response. Oncotarget 6(23):19364–19365. doi:10.18632/oncotarget.5127
Bavagnoli L, Cucuzza S, Campanini G, Rovida F, Paolucci S, Baldanti F, Maga G (2015) The novel influenza A virus protein PA-X and its naturally deleted variant show different enzymatic properties in comparison to the viral endonuclease PA. Nucleic Acids Res 43(19):9405–9417. doi:10.1093/nar/gkv926
Gao H, Sun Y, Hu J, Qi L, Wang J, Xiong X, Wang Y, He Q, Lin Y, Kong W, Seng LG, Sun H, Pu J, Chang KC, Liu X, Liu J (2015) The contribution of PA-X to the virulence of pandemic 2009 H1N1 and highly pathogenic H5N1 avian influenza viruses. Sci Rep 5:8262. doi:10.1038/srep08262
Huang CH, Chen CJ, Yen CT, Yu CP, Huang PN, Kuo RL, Lin SJ, Chang CK, Shih SR (2013) Caspase-1 deficient mice are more susceptible to influenza A virus infection with PA variation. J Infect Dis 208(11):1898–1905. doi:10.1093/infdis/jit381
Sun Y, Qin K, Wang J, Pu J, Tang Q, Hu Y, Bi Y, Zhao X, Yang H, Shu Y, Liu J (2011) High genetic compatibility and increased pathogenicity of reassortants derived from avian H9N2 and pandemic H1N1/2009 influenza viruses. Proc Natl Acad Sci USA 108(10):4164–4169. doi:10.1073/pnas.1019109108
Itoh S, Taketomi A, Harimoto N, Tsujita E, Rikimaru T, Shirabe K, Shimada M, Maehara Y (2010) Antineoplastic effects of gamma linolenic Acid on hepatocellular carcinoma cell lines. J Clin Biochem Nutr 47(1):81–90. doi:10.3164/jcbn.10-24
Szretter KJ, Gangappa S, Lu X, Smith C, Shieh WJ, Zaki SR, Sambhara S, Tumpey TM, Katz JM (2007) Role of host cytokine responses in the pathogenesis of avian H5N1 influenza viruses in mice. J Virol 81(6):2736–2744. doi:10.1128/JVI.02336-06
Perrone LA, Plowden JK, Garcia-Sastre A, Katz JM, Tumpey TM (2008) H5N1 and 1918 pandemic influenza virus infection results in early and excessive infiltration of macrophages and neutrophils in the lungs of mice. PLoS Pathog 4(8):e1000115. doi:10.1371/journal.ppat.1000115
Cameron CM, Cameron MJ, Bermejo-Martin JF, Ran L, Xu L, Turner PV, Ran R, Danesh A, Fang Y, Chan PK, Mytle N, Sullivan TJ, Collins TL, Johnson MG, Medina JC, Rowe T, Kelvin DJ (2008) Gene expression analysis of host innate immune responses during Lethal H5N1 infection in ferrets. J Virol 82(22):11308–11317. doi:10.1128/JVI.00691-08
Baskin CR, Bielefeldt-Ohmann H, Tumpey TM, Sabourin PJ, Long JP, Garcia-Sastre A, Tolnay AE, Albrecht R, Pyles JA, Olson PH, Aicher LD, Rosenzweig ER, Murali-Krishna K, Clark EA, Kotur MS, Fornek JL, Proll S, Palermo RE, Sabourin CL, Katze MG (2009) Early and sustained innate immune response defines pathology and death in nonhuman primates infected by highly pathogenic influenza virus. Proc Natl Acad Sci USA 106(9):3455–3460. doi:10.1073/pnas.0813234106
Garcia CC, Weston-Davies W, Russo RC, Tavares LP, Rachid MA, Alves-Filho JC, Machado AV, Ryffel B, Nunn MA, Teixeira MM (2013) Complement C5 activation during influenza A infection in mice contributes to neutrophil recruitment and lung injury. PLoS One 8(5):e64443. doi:10.1371/journal.pone.0064443
Sun S, Zhao G, Liu C, Wu X, Guo Y, Yu H, Song H, Du L, Jiang S, Guo R, Tomlinson S, Zhou Y (2013) Inhibition of complement activation alleviates acute lung injury induced by highly pathogenic avian influenza H5N1 virus infection. Am J Respir Cell Mol Biol 49(2):221–230. doi:10.1165/rcmb.2012-0428OC
Wang R, Xiao H, Guo R, Li Y, Shen B (2015) The role of C5a in acute lung injury induced by highly pathogenic viral infections. Emerg Microb Infect 4(5):e28. doi:10.1038/emi.2015.28
Nascimento EJM, Silva AM, Cordeiro MT, Brito CA, Gil LHVG, Braga-Neto U, Marques ETA (2009) Alternative complement pathway deregulation is correlated with dengue severity. PLoS One. doi:10.1371/journal.pone.0006782
Kanmura S, Uto H, Sato Y, Kumagai K, Sasaki F, Moriuchi A, Oketani M, Ido A, Nagata K, Hayashi K, Stuver SO, Tsubouchi H (2010) The complement component C3a fragment is a potential biomarker for hepatitis C virus-related hepatocellular carcinoma. J Gastroenterol 45(4):459–467. doi:10.1007/s00535-009-0160-5
Huber-Lang M, Sarma VJ, Lu KT, McGuire SR, Padgaonkar VA, Guo RF, Younkin EM, Kunkel RG, Ding J, Erickson R, Curnutte JT, Ward PA (2001) Role of C5a in multiorgan failure during sepsis. J Immunol 166(2):1193–1199
Hawlisch H, Belkaid Y, Baelder R, Hildeman D, Gerard C, Kohl J (2005) C5a negatively regulates toll-like receptor 4-induced immune responses. Immunity 22(4):415–426. doi:10.1016/j.immuni.2005.02.006
Sun S, Zhao G, Liu C, Fan W, Zhou X, Zeng L, Guo Y, Kou Z, Yu H, Li J, Wang R, Li Y, Schneider C, Habel M, Riedemann NC, Du L, Jiang S, Guo R, Zhou Y (2015) Treatment with anti-C5a antibody improves the outcome of H7N9 virus infection in African green monkeys. Clin Infect Dis 60(4):586–595. doi:10.1093/cid/ciu887
Kash JC, Xiao Y, Davis AS, Walters KA, Chertow DS, Easterbrook JD, Dunfee RL, Sandouk A, Jagger BW, Schwartzman LM, Kuestner RE, Wehr NB, Huffman K, Rosenthal RA, Ozinsky A, Levine RL, Doctrow SR, Taubenberger JK (2014) Treatment with the reactive oxygen species scavenger EUK-207 reduces lung damage and increases survival during 1918 influenza virus infection in mice. Free Radic Biol Med 67:235–247. doi:10.1016/j.freeradbiomed.2013.10.014
Akaike T, Noguchi Y, Ijiri S, Setoguchi K, Suga M, Zheng YM, Dietzschold B, Maeda H (1996) Pathogenesis of influenza virus-induced pneumonia: involvement of both nitric oxide and oxygen radicals. Proc Natl Acad Sci USA 93(6):2448–2453
Akaike T, Ando M, Oda T, Diii T, Ijiri S, Araki S, Maeda H (1990) Dependence on O2- generation by xanthine oxidase of pathogenesis of influenza virus infection in mice. J Clin Invest 85(3):739–745. doi:10.1172/JCI114499
Maeda H, Akaike T (1991) Oxygen free radicals as pathogenic molecules in viral diseases. Proc Soc Exp Biol Med 198(2):721–727
Imai Y, Kuba K, Neely GG, Yaghubian-Malhami R, Perkmann T, van Loo G, Ermolaeva M, Veldhuizen R, Leung YHC, Wang HL, Liu HL, Sun Y, Pasparakis M, Kopf M, Mech C, Bavari S, Peiris JSM, Slutsky AS, Akira S, Hultqvist M, Holmdahl R, Nicholls J, Jiang CY, Binder CJ, Penninger JM (2008) Identification of oxidative stress and toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell 133(2):235–249. doi:10.1016/j.cell.2008.02.043
Vlahos R, Stambas J, Selemidis S (2012) Suppressing production of reactive oxygen species (ROS) for influenza A virus therapy. Trends Pharmacol Sci 33(1):3–8. doi:10.1016/j.tips.2011.09.001
He G, Dong C, Luan Z, McAllan BM, Xu T, Zhao L, Qiao J (2013) Oxygen free radical involvement in acute lung injury induced by H5N1 virus in mice. Influenza Other Respir Viruses 7(6):945–953. doi:10.1111/irv.12067
Ye S, Lowther S, Stambas J (2015) Inhibition of reactive oxygen species production ameliorates inflammation induced by influenza A viruses via upregulation of SOCS1 and SOCS3. J Virol 89(5):2672–2683. doi:10.1128/JVI.03529-14
Vlahos R, Selemidis S (2014) NADPH oxidases as novel pharmacologic targets against influenza A virus infection. Mol Pharmacol 86(6):747–759. doi:10.1124/mol.114.095216
Domej W, Oettl K, Renner W (2014) Oxidative stress and free radicals in COPD implications and relevance for treatment. Int J Chronic Obstr 9:1207–1224. doi:10.2147/Copd.S51226
Tran TH, Nguyen TL, Nguyen TD, Luong TS, Pham PM, Nguyen VV, Pham TS, Vo CD, Le TQ, Ngo TT, Dao BK, Le PP, Nguyen TT, Hoang TL, Cao VT, Le TG, Nguyen DT, Le HN, Nguyen KT, Le HS, Le VT, Christiane D, Tran TT, de Menno J, Schultsz C, Cheng P, Lim W, Horby P, Farrar J (2004) Avian influenza A (H5N1) in 10 patients in Vietnam. N Engl J Med 350(12):1179–1188. doi:10.1056/NEJMoa040419
Chotpitayasunondh T, Ungchusak K, Hanshaoworakul W, Chunsuthiwat S, Sawanpanyalert P, Kijphati R, Lochindarat S, Srisan P, Suwan P, Osotthanakorn Y, Anantasetagoon T, Kanjanawasri S, Tanupattarachai S, Weerakul J, Chaiwirattana R, Maneerattanaporn M, Poolsavathitikool R, Chokephaibulkit K, Apisarnthanarak A (2004) Dowell SF (2005) Human disease from influenza A (H5N1), Thailand. Emerg Infect Dis 11(2):201–209. doi:10.3201/eid1102.041061
Kobasa D, Jones SM, Shinya K, Kash JC, Copps J, Ebihara H, Hatta Y, Kim JH, Halfmann P, Hatta M, Feldmann F, Alimonti JB, Fernando L, Li Y, Katze MG, Feldmann H, Kawaoka Y (2007) Aberrant innate immune response in lethal infection of macaques with the 1918 influenza virus. Nature 445(7125):319–323. doi:10.1038/nature05495
Suzuki K, Okada H, Itoh T, Tada T, Mase M, Nakamura K, Kubo M, Tsukamoto K (2009) Association of increased pathogenicity of Asian H5N1 highly pathogenic avian influenza viruses in chickens with highly efficient viral replication accompanied by early destruction of innate immune responses. J Virol 83(15):7475–7486. doi:10.1128/JVI.01434-08
Oldstone MB (2013) Lessons learned and concepts formed from study of the pathogenesis of the two negative-strand viruses lymphocytic choriomeningitis and influenza. Proc Natl Acad Sci USA 110(11):4180–4183. doi:10.1073/pnas.1222025110
Vogel AJ, Harris S, Marsteller N, Condon SA, Brown DM (2014) Early cytokine dysregulation and viral replication are associated with mortality during lethal influenza infection. Viral Immunol 27(5):214–224. doi:10.1089/vim.2013.0095
Hinshaw VS, Olsen CW, Dybdahl-Sissoko N, Evans D (1994) Apoptosis: a mechanism of cell killing by influenza A and B viruses. J Virol 68(6):3667–3673
Nichols JE, Niles JA, Roberts NJ Jr (2001) Human lymphocyte apoptosis after exposure to influenza A virus. J Virol 75(13):5921–5929. doi:10.1128/JVI.73.13.5921-5929.2001
Lam WY, Tang JW, Yeung AC, Chiu LC, Sung JJ, Chan PK (2008) Avian influenza virus A/HK/483/97(H5N1) NS1 protein induces apoptosis in human airway epithelial cells. J Virol 82(6):2741–2751. doi:10.1128/JVI.01712-07
Price GE, Smith H, Sweet C (1997) Differential induction of cytotoxicity and apoptosis by influenza virus strains of differing virulence. J Gen Virol 78(Pt 11):2821–2829
Schultz-Cherry S, Dybdahl-Sissoko N, Neumann G, Kawaoka Y, Hinshaw VS (2001) Influenza virus ns1 protein induces apoptosis in cultured cells. J Virol 75(17):7875–7881
Brydon EW, Morris SJ, Sweet C (2005) Role of apoptosis and cytokines in influenza virus morbidity. FEMS Microbiol Rev 29(4):837–850. doi:10.1016/j.femsre.2004.12.003
Tumpey TM, Lu X, Morken T, Zaki SR, Katz JM (2000) Depletion of lymphocytes and diminished cytokine production in mice infected with a highly virulent influenza A (H5N1) virus isolated from humans. J Virol 74(13):6105–6116
Gloire G, Legrand-Poels S, Piette J (2006) NF-kappa B activation by reactive oxygen species: fifteen years later. Biochem Pharmacol 72(11):1493–1505. doi:10.1016/j.bcp.2006.04.011
Lander HM (1997) An essential role for free radicals and derived species in signal transduction. FASEB J 11(2):118–124
Israel N, Gougerot-Pocidalo MA (1997) Oxidative stress in human immunodeficiency virus infection. Cell Mol Life Sci 53(11–12):864–870
Dalle-Donne I, Rossi R, Colombo R, Giustarini D, Milzani A (2006) Biomarkers of oxidative damage in human disease. Clin Chem 52(4):601–623. doi:10.1373/clinchem.2005.061408
Gougeon ML, Montagnier L (1993) Apoptosis in AIDS. Science 260(5112):1269–1270
Pace GW, Leaf CD (1995) The role of oxidative stress in HIV disease. Free Radic Biol Med 19(4):523–528. doi:10.1016/0891-5849(95)00047-2
Paracha UZ, Fatima K, Alqahtani M, Chaudhary A, Abuzenadah A, Damanhouri G, Qadri I (2013) Oxidative stress and hepatitis C virus. Virol J 10:251. doi:10.1186/1743-422X-10-251
Baeuerle PA, Baltimore D (1988) Activation of DNA-binding activity in an apparently cytoplasmic precursor of the NF-kappa B transcription factor. Cell 53(2):211–217. doi:10.1016/0092-8674(88)90382-0
Staal FJ, Roederer M, Herzenberg LA (1990) Intracellular thiols regulate activation of nuclear factor kappa B and transcription of human immunodeficiency virus. Proc Natl Acad Sci USA 87(24):9943–9947
Simon HU, Yousefi S, Schranz C, Schapowal A, Bachert C, Blaser K (1997) Direct demonstration of delayed eosinophil apoptosis as a mechanism causing tissue eosinophilia. J Immunol 158(8):3902–3908
Dibbert B, Weber M, Nikolaizik WH, Vogt P, Schoni MH, Blaser K, Simon HU (1999) Cytokine-mediated Bax deficiency and consequent delayed neutrophil apoptosis: a general mechanism to accumulate effector cells in inflammation. P Natl Acad Sci USA 96(23):13330–13335. doi:10.1073/pnas.96.23.13330
Smith JA (1994) Neutrophils, host defense, and inflammation: a double-edged sword. J Leukoc Biol 56(6):672–686
Vlahos R, Stambas J, Bozinovski S, Broughton BR, Drummond GR, Selemidis S (2011) Inhibition of Nox2 oxidase activity ameliorates influenza A virus-induced lung inflammation. PLoS Pathog 7(2):e1001271. doi:10.1371/journal.ppat.1001271
Selemidis S, Seow HJ, Broughton BR, Vinh A, Bozinovski S, Sobey CG, Drummond GR, Vlahos R (2013) Nox1 oxidase suppresses influenza a virus-induced lung inflammation and oxidative stress. PLoS One 8(4):e60792. doi:10.1371/journal.pone.0060792
Kopf M, Abel B, Gallimore A, Carroll M, Bachmann MF (2002) Complement component C3 promotes T-cell priming and lung migration to control acute influenza virus infection. Nat Med 8(4):373–378. doi:10.1038/nm0402-373
Carroll MC (2004) The complement system in regulation of adaptive immunity. Nat Immunol 5(10):981–986. doi:10.1038/ni1113ni1113
Kim AHJ, Dimitriou ID, Holland MCH, Mastellos D, Mueller YM, Altman JD, Lambris JD, Katsikis PD (2004) Complement C5a receptor is essential for the optimal generation of antiviral CD8(+) T cell responses. J Immunol 173(4):2524–2529
Stoermer KA, Morrison TE (2011) Complement and viral pathogenesis. Virology 411(2):362–373. doi:10.1016/j.virol.2010.12.045
Kohl J (2001) Anaphylatoxins and infectious and non-infectious inflammatory diseases. Mol Immunol 38(2–3):175–187. doi:10.1016/S0161-5890(01)00041-4
Huang KJ, Su IJ, Theron M, Wu YC, Lai SK, Liu CC, Lei HY (2005) An interferon-gamma-related cytokine storm in SARS patients. J Med Virol 75(2):185–194. doi:10.1002/jmv.20255
Zhou J, Chu H, Li C, Wong BH, Cheng ZS, Poon VK, Sun T, Lau CC, Wong KK, Chan JY, Chan JF, To KK, Chan KH, Zheng BJ, Yuen KY (2014) Active replication of Middle East respiratory syndrome coronavirus and aberrant induction of inflammatory cytokines and chemokines in human macrophages: implications for pathogenesis. J Infect Dis 209(9):1331–1342. doi:10.1093/infdis/jit504
Huarte M, Falcon A, Nakaya Y, Ortin J, Garcia-Sastre A, Nieto A (2003) Threonine 157 of influenza virus PA polymerase subunit modulates RNA replication in infectious viruses. J Virol 77(10):6007–6013
Gabriel G, Herwig A, Klenk HD (2008) Interaction of polymerase subunit PB2 and NP with importin alpha1 is a determinant of host range of influenza A virus. PLoS Pathog 4(2):e11. doi:10.1371/journal.ppat.0040011
Hu J, Hu Z, Mo Y, Wu Q, Cui Z, Duan Z, Huang J, Chen H, Chen Y, Gu M, Wang X, Hu S, Liu H, Liu W, Liu X (2013) The PA and HA gene-mediated high viral load and intense innate immune response in the brain contribute to the high pathogenicity of H5N1 avian influenza virus in mallard ducks. J Virol 87(20):11063–11075. doi:10.1128/JVI.00760-13
Conenello GM, Palese P (2007) Influenza A virus PB1-F2: a small protein with a big punch. Cell Host Microbe 2(4):207–209. doi:10.1016/j.chom.2007.09.010
Krumbholz A, Philipps A, Oehring H, Schwarzer K, Eitner A, Wutzler P, Zell R (2011) Current knowledge on PB1-F2 of influenza A viruses. Med Microbiol Immunol 200(2):69–75. doi:10.1007/s00430-010-0176-8
Varga ZT, Palese P (2011) The influenza A virus protein PB1-F2: killing two birds with one stone? Virulence 2(6):542–546. doi:10.4161/viru.2.6.17812
Chakrabarti AK, Pasricha G (2013) An insight into the PB1F2 protein and its multifunctional role in enhancing the pathogenicity of the influenza A viruses. Virology 440(2):97–104. doi:10.1016/j.virol.2013.02.025
Kosik I, Krejnusova I, Praznovska M, Russ G (2013) The multifaceted effect of PB1-F2 specific antibodies on influenza A virus infection. Virology 447(1–2):1–8. doi:10.1016/j.virol.2013.08.022
Acknowledgments
The precious contribution of the following colleagues is warmly acknowledged—Maozhi Hu, Qiuxiang Yan and Yipeng Sun. Maozhi Hu and Qiuxiang Yan are appreciated for their technical support of flow cytometry. Yipeng Sun is greatly acknowledged for her kindly help in histopathology observation. This work was supported by the National Natural Science Foundation of China (31502076), by the Jiangsu Provincial Natural Science Foundation of China (BK20150444), by the Postdoctoral Science Foundation of Jiangsu Province (1501015B), by the Natural Science Foundation of the Higher Education Institutions of Jiangsu Province, China (15KJB230006), by the National Key Technologies R&D Program of China (2015BAD12B01), by the earmarked fund for Modern Agro-industry Technology Research System (nycytx-41-G07) and by A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Jiao Hu, Yiqun Mo and Zhao Gao authors have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Hu, J., Mo, Y., Gao, Z. et al. PA-X-associated early alleviation of the acute lung injury contributes to the attenuation of a highly pathogenic H5N1 avian influenza virus in mice. Med Microbiol Immunol 205, 381–395 (2016). https://doi.org/10.1007/s00430-016-0461-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-016-0461-2