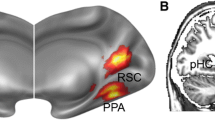
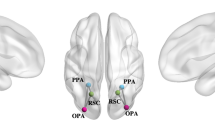
Abstract
Successful navigation is largely dependent on the ability to make correct decisions at navigational decision points. However, the interaction between the brain regions associated with the navigational decision point in a schematic map is unclear. In this study, we adopted a 2D subway paradigm to study the neural basis underlying decision points. Twenty-eight subjects performed a spatial navigation task using a subway map during fMRI scanning. We adopted a voxel-wise general linear model (GLM) approach and found four brain regions, the left hippocampus (HIP), left parahippocampal gyrus (PHG), left ventromedial prefrontal cortex (vmPFC), and right retrosplenial cortex (RSC), activated at a navigational decision point in a schematic map. Using a psychophysiological interactions (PPI) method, we found that (1) both the left vmPFC and right HIP interacted cooperatively with the right RSC, and (2) the left HIP and the left vmPFC interacted cooperatively at the decision point. These findings may be helpful for revealing the neural mechanisms underlying decision points in a schematic map during spatial navigation.



Similar content being viewed by others
Data and code availability statement
The datasets generated and analyzed in the current study are available from the corresponding author upon reasonable request.
Abbreviations
- BA:
-
Brodmann’s area
- HIP:
-
Hippocampus
- mPFC:
-
Medial prefrontal cortex
- OPA:
-
Occipital place area
- PCUN:
-
Precuneus
- PFC:
-
Prefrontal cortex
- PHG:
-
Parahippocampal gyrus
- PPA:
-
Parahippocampal place area
- RSC:
-
Retrosplenial cortex
- vmPFC:
-
Ventromedial prefrontal cortex
- DP:
-
Decision points
- nonDP:
-
Non-decision points
- fMRI:
-
Functional magnetic resonance imaging
- GLM:
-
General linear model
- GRF:
-
Gaussian random field
- HRF:
-
Hemodynamic response function
- PPI:
-
Psychophysiological interactions
- ROI:
-
Region of interest
- TFCE:
-
Threshold-free cluster enhancement
References
Balaguer J, Spiers H, Hassabis D, Summerfield C (2016) Neural mechanisms of hierarchical planning in a virtual subway network. Neuron 90(4):893–903. https://doi.org/10.1016/j.neuron.2016.03.037
Barron HC, Dolan RJ, Behrens TE (2013) Online evaluation of novel choices by simultaneous representation of multiple memories. Nat Neurosci 16(10):1492–1498. https://doi.org/10.1038/nn.3515
Baumann O, Mattingley JB (2021) Extrahippocampal contributions to spatial navigation in humans: a review of the neuroimaging evidence. Hippocampus 31(7):640–657. https://doi.org/10.1002/hipo.23313
Behrens TEJ, Muller TH, Whittington JCR, Mark S, Baram AB, Stachenfeld KL, Kurth-Nelson Z (2018) What is a cognitive map? Organizing knowledge for flexible behavior. Neuron 100(2):490–509. https://doi.org/10.1016/j.neuron.2018.10.002
Belchior H, Lopes-Dos-Santos V, Tort AB, Ribeiro S (2014) Increase in hippocampal theta oscillations during spatial decision making. Hippocampus 24(6):693–702. https://doi.org/10.1002/hipo.22260
Bellmund JLS, Gardenfors P, Moser EI, Doeller CF (2018) Navigating cognition: spatial codes for human thinking. Science. https://doi.org/10.1126/science.aat6766
Bonner MF, Epstein RA (2017) Coding of navigational affordances in the human visual system. Proc Natl Acad Sci USA 114(18):4793–4798. https://doi.org/10.1073/pnas.1618228114
Bottini R, Doeller CF (2020) Knowledge Across Reference Frames: Cognitive Maps and Image Spaces. Trends Cogn Sci. https://doi.org/10.1016/j.tics.2020.05.008
Brown TI, Ross RS, Tobyne SM, Stern CE (2012) Cooperative interactions between hippocampal and striatal systems support flexible navigation. Neuroimage 60(2):1316–1330. https://doi.org/10.1016/j.neuroimage.2012.01.046
Brown TI, Carr VA, LaRocque KF, Favila SE, Gordon AM, Bowles B, Wagner ADJS (2016) Prospective representation of navigational goals in the human hippocampus. Science 352(6291):1323–1326
Byrne P, Becker S, Burgess N (2007) Remembering the past and imagining the future: a neural model of spatial memory and imagery. Psychol Rev 114(2):340–375. https://doi.org/10.1037/0033-295X.114.2.340
Cavada C, Compañy T, Tejedor J, Cruz-Rizzolo RJ, Reinoso-Suárez FJCC (2000) The anatomical connections of the macaque monkey orbitofrontal cortex. A review. Cereb Cortex 10(3):220–242. https://doi.org/10.1093/cercor/10.3.220
Chadwick MJ, Jolly AE, Amos DP, Hassabis D, Spiers HJ (2015) A goal direction signal in the human entorhinal/subicular region. Curr Biol 25(1):87–92. https://doi.org/10.1016/j.cub.2014.11.001
Chersi F, Pezzulo G (2012) Using hippocampal-striatal loops for spatial navigation and goal-directed decision-making. Cogn Process 13(Suppl 1):S125-129. https://doi.org/10.1007/s10339-012-0475-7
Ciaramelli E (2008) The role of ventromedial prefrontal cortex in navigation: a case of impaired wayfinding and rehabilitation. Neuropsychologia 46(7):2099–2105. https://doi.org/10.1016/j.neuropsychologia.2007.11.029
Colombo D, Serino S, Tuena C, Pedroli E, Dakanalis A, Cipresso P, Riva G (2017) Egocentric and allocentric spatial reference frames in aging: a systematic review. Neurosci Biobehav Rev 80:605–621. https://doi.org/10.1016/j.neubiorev.2017.07.012
Constantinescu AO, O’Reilly JX, Behrens TEJS (2016) Organizing conceptual knowledge in humans with a gridlike code. Science 352(6292):1464–1468
Cornwell BR, Johnson LL, Holroyd T, Carver FW, Grillon C (2008) Human hippocampal and parahippocampal theta during goal-directed spatial navigation predicts performance on a virtual Morris water maze. J Neurosci 28(23):5983–5990. https://doi.org/10.1523/JNEUROSCI.5001-07.2008
Dahmani L, Bohbot VD (2015) Dissociable contributions of the prefrontal cortex to hippocampus- and caudate nucleus-dependent virtual navigation strategies. Neurobiol Learn Mem 117:42–50. https://doi.org/10.1016/j.nlm.2014.07.002
De Simoni S, Grover PJ, Jenkins PO, Honeyfield L, Quest RA, Ross E, Sharp DJ (2016) Disconnection between the default mode network and medial temporal lobes in post-traumatic amnesia. Brain 139(Pt 12):3137–3150. https://doi.org/10.1093/brain/aww241
Doeller CF, Barry C, Burgess N (2010) Evidence for grid cells in a human memory network. Nature 463(7281):657–661. https://doi.org/10.1038/nature08704
Ekstrom AD, Spiers HJ, Bohbot VD, Rosenbaum RS (2018) Human spatial navigation. Princeton University Press, Princeton
Epstein RA, Patai EZ, Julian JB, Spiers HJ (2017) The cognitive map in humans: spatial navigation and beyond. Nat Neurosci 20(11):1504–1513. https://doi.org/10.1038/nn.4656
Greicius MD, Supekar K, Menon V, Dougherty RFJCC (2009) Resting-state functional connectivity reflects structural connectivity in the default mode network. Cereb Cortex 19(1):72–78. https://doi.org/10.1093/cercor/bhn059
Grimsen C, Hildebrandt H, Fahle M (2008) Dissociation of egocentric and allocentric coding of space in visual search after right middle cerebral artery stroke. Neuropsychologia 46(3):902–914. https://doi.org/10.1016/j.neuropsychologia.2007.11.028
Hare TA, Camerer CF, Rangel AJS (2009) Self-control in decision-making involves modulation of the vmPFC valuation system. Science 324(5927):646–648. https://doi.org/10.1126/science.1168450
Howard LR, Javadi AH, Yu Y, Mill RD, Morrison LC, Knight R, Spiers HJ (2014) The hippocampus and entorhinal cortex encode the path and Euclidean distances to goals during navigation. Curr Biol 24(12):1331–1340. https://doi.org/10.1016/j.cub.2014.05.001
Igloi K, Doeller CF, Berthoz A, Rondi-Reig L, Burgess N (2010) Lateralized human hippocampal activity predicts navigation based on sequence or place memory. Proc Natl Acad Sci 107(32):14466–14471. https://doi.org/10.1073/pnas.1004243107
Janzen G (2006) Memory for object location and route direction in virtual large-scale space. Q J Exp Psychol (hove) 59(3):493–508. https://doi.org/10.1080/02724980443000746
Janzen G, Jansen C (2010) A neural wayfinding mechanism adjusts for ambiguous landmark information. Neuroimage 52(1):364–370. https://doi.org/10.1016/j.neuroimage.2010.03.083
Janzen G, van Turennout M (2004) Selective neural representation of objects relevant for navigation. Nat Neurosci 7(6):673–677. https://doi.org/10.1038/nn1257
Janzen G, Weststeijn CG (2007) Neural representation of object location and route direction: an event-related fMRI study. Brain Res 1165:116–125. https://doi.org/10.1016/j.brainres.2007.05.074
Janzen G, Jansen C, Turennout MV (2008) Memory consolidation of landmarks in good navigators. Hippocampus 18(1):40–47. https://doi.org/10.1002/hipo.20364
Javadi AH, Emo B, Howard LR, Zisch FE, Yu Y, Knight R, Spiers HJ (2017) Hippocampal and prefrontal processing of network topology to simulate the future. Nat Commun 8:14652. https://doi.org/10.1038/ncomms14652
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17(2):825–841. https://doi.org/10.1006/nimg.2002.1132
Johnson A, Redish AD (2007) Neural ensembles in CA3 transiently encode paths forward of the animal at a decision point. J Neurosci 27(45):12176–12189. https://doi.org/10.1523/jneurosci.3761-07.2007
Kaplan R, Schuck NW, Doeller CF (2017) The role of mental maps in decision-making. Trends Neurosci 40(5):256–259. https://doi.org/10.1016/j.tins.2017.03.002
Kessels RP, van Doormaal A, Janzen G (2011) Landmark recognition in Alzheimer’s dementia: spared implicit memory for objects relevant for navigation. PLoS ONE 6(4):e18611. https://doi.org/10.1371/journal.pone.0018611
Kobayashi Y, Amaral DG (2003) Macaque monkey retrosplenial cortex: II. Cortical afferents. J Comp Neurol 466(1):48–79. https://doi.org/10.1002/cne.10883
Lindquist M (2020) Neuroimaging results altered by varying analysis pipelines. Nature 582(7810):36–37. https://doi.org/10.1038/d41586-020-01282-z
Maguire EA, Burgess N, Donnett JG, Frackowiak RS, Frith CD, O’Keefe JJS (1998) Knowing where and getting there: a human navigation network. Science 280(5365):921–924. https://doi.org/10.1126/science.280.5365.921
Marchette SA, Vass LK, Ryan J, Epstein RA (2015) Outside looking in landmark generalization in the human navigational system. J Neurosci 35(44):14896–14908. https://doi.org/10.1523/JNEUROSCI.2270-15.2015
Miller J, Watrous AJ, Tsitsiklis M, Lee SA, Sheth SA, Schevon CA, Jacobs J (2018) Lateralized hippocampal oscillations underlie distinct aspects of human spatial memory and navigation. Nat Commun 9(1):2423. https://doi.org/10.1038/s41467-018-04847-9
Mullally SL, Vargha-Khadem F, Maguire EA (2014) Scene construction in developmental amnesia: an fMRI study. Neuropsychologia 52:1–10. https://doi.org/10.1016/j.neuropsychologia.2013.11.001
Nau M, Navarro Schröder T, Frey M, Doeller CF (2020) Behavior-dependent directional tuning in the human visual-navigation network. Nat Commun. https://doi.org/10.1038/s41467-020-17000-2
Nieh EH, Schottdorf M, Freeman NW, Low RJ, Lewallen S, Koay SA, Tank DW (2021) Geometry of abstract learned knowledge in the hippocampus. Nature 595(7865):80–84. https://doi.org/10.1038/s41586-021-03652-7
O’Keefe J, Nadel L (1978) The hippocampus as a cognitive map. Oxford University Press, Oxford
O’Reilly JX, Woolrich MW, Behrens TE, Smith SM, Johansen-Berg H (2012) Tools of the trade: psychophysiological interactions and functional connectivity. Soc Cogn Affect Neurosci 7(5):604–609. https://doi.org/10.1093/scan/nss055
Pu Y, Cornwell BR, Cheyne D, Johnson BW (2017) The functional role of human right hippocampal/parahippocampal theta rhythm in environmental encoding during virtual spatial navigation. Hum Brain Mapp 38(3):1347–1361. https://doi.org/10.1002/hbm.23458
Rempel-Clower NL, Barbas HJCC (2000) The laminar pattern of connections between prefrontal and anterior temporal cortices in the rhesus monkey is related to cortical structure and function. Cereb Cortex 10(9):851–865. https://doi.org/10.1093/cercor/10.9.851
Roberts AC, Tomic DL, Parkinson CH, Roeling TA, Cutter DJ, Robbins TW, Everitt BJ (2007) Forebrain connectivity of the prefrontal cortex in the marmoset monkey (Callithrix jacchus): an anterograde and retrograde tract-tracing study. J Comp Neurol 502(1):86–112. https://doi.org/10.1002/cne.21300
Schinazi VR, Epstein RA (2010) Neural correlates of real-world route learning. Neuroimage 53(2):725–735. https://doi.org/10.1016/j.neuroimage.2010.06.065
Schneider B, Koenigs M (2017) Human lesion studies of ventromedial prefrontal cortex. Neuropsychologia 107:84–93. https://doi.org/10.1016/j.neuropsychologia.2017.09.035
Sherrill KR, Erdem UM, Ross RS, Brown TI, Hasselmo ME, Stern CE (2013) Hippocampus and retrosplenial cortex combine path integration signals for successful navigation. J Neurosci 33(49):19304–19313. https://doi.org/10.1523/JNEUROSCI.1825-13.2013
Shine JP, Valdes-Herrera JP, Tempelmann C, Wolbers T (2019) Evidence for allocentric boundary and goal direction information in the human entorhinal cortex and subiculum. Nat Commun 10(1):4004. https://doi.org/10.1038/s41467-019-11802-9
Spiers HJ (2020) The hippocampal cognitive map: one space or many? Trends Cogn Sci 24(3):168–170. https://doi.org/10.1016/j.tics.2019.12.013
Spiers HJ, Barry C (2015) Neural systems supporting navigation. Curr Opin Behav Sci 1:47–55. https://doi.org/10.1016/j.cobeha.2014.08.005
Sun L, Frank SM, Epstein RA, Tse PU (2021) The parahippocampal place area and hippocampus encode the spatial significance of landmark objects. Neuroimage 236:118081. https://doi.org/10.1016/j.neuroimage.2021.118081
Tavares RM, Mendelsohn A, Grossman Y, Williams CH, Shapiro M, Trope Y, Schiller D (2015) A map for social navigation in the human brain. Neuron 87(1):231–243. https://doi.org/10.1016/j.neuron.2015.06.011
Theves S, Fernandez G, Doeller CF (2019) The hippocampus encodes distances in multidimensional feature space. Curr Biol 29(7):1226-1231.e1223. https://doi.org/10.1016/j.cub.2019.02.035
Tolman EC (1948) Cognitive maps in rats and men. Psychol Rev 55(4):189
Vann SD, Aggleton JP, Maguire EA (2009) What does the retrosplenial cortex do? Nat Rev Neurosci 10(11):792–802. https://doi.org/10.1038/nrn2733
Viard A, Doeller CF, Hartley T, Bird CM, Burgess N (2011) Anterior hippocampus and goal-directed spatial decision making. J Neurosci 31(12):4613–4621. https://doi.org/10.1523/JNEUROSCI.4640-10.2011
Xu J, Evensmoen HR, Lehn H, Pintzka CW, Håberg AKJN (2010) Persistent posterior and transient anterior medial temporal lobe activity during navigation. Neuroimage 52(4):1654–1666. https://doi.org/10.1016/j.neuroimage.2010.05.074
Acknowledgements
The authors thank Dr. Jan Balaguer and Prof. Christopher Summerfield for providing the code of the subway paradigm. The authors appreciate Drs. Rhoda E. and Edmund F. Perozzi for editing the manuscript. The multiband accelerated EPI pulse sequence was obtained from the University of Minnesota Center for Magnetic Resonance Research (CMRR) through a C2P agreement. We also appreciate the University of Minnesota CMRR for the development of the sequence.
Funding
This work was supported by the National Natural Science Foundation of China (Grant numbers: 82171914 and 81871338) and the National Key R&D Program of China (Grant numbers: 2018YFC1705006).
Author information
Authors and Affiliations
Contributions
Conceptualization: QQ, YW and RH; methodology: QQ, YW, SZ and SW; software: SW and SZ; investigation: YW, QQ, SW and SZ; formal analysis: QQ and YW; writing—original draft preparation: QQ and YW; writing—review and editing: QQ, YW, SZ, SL, QH and RH; visualization: QQ; funding acquisition: RH; resources: RH; supervision: RH.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing financial interests.
Ethical approval
The study protocol was approved by the Research Review Board of South China Normal University. Written informed consent was obtained from each subject before the study.
Informed consent
Informed consent was obtained from all individual subjects included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qi, Q., Weng, Y., Zheng, S. et al. Task-related connectivity of decision points during spatial navigation in a schematic map. Brain Struct Funct 227, 1697–1710 (2022). https://doi.org/10.1007/s00429-022-02466-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-022-02466-1