Abstract
An ordered relation of structure and function has been a cornerstone in thinking about brain organization. Like the brain itself, however, this is not straightforward and is confounded both by functional intricacy and structural plasticity (many routes to a given outcome). As a striking case of putative structure–function correlation, this mini-review focuses on the relatively well-characterized pattern of cytochrome oxidase (CO) blobs (aka “patches” or “puffs”) in the supragranular layers of macaque monkey visual cortex. The pattern is without doubt visually compelling, and the semi-dichotomous array of CO+ blobs and CO− interblobs is consistent with multiple studies reporting compartment-specific preferential connectivity and distinctive physiological response properties. Nevertheless, as briefly reviewed here, the finer anatomical organization of this system is surprisingly under-investigated, and the relation to functional aspects, therefore, unclear. Microcircuitry, cell type, and three-dimensional spatiotemporal level investigations of the CO+ CO− pattern are needed and may open new views to structure–function organization of visual cortex, and to phylogenetic and ontogenetic comparisons across nonhuman primates (NHP), and between NHP and humans.
Graphic abstract







Similar content being viewed by others
Availability of data and materials
This will be freely available upon request.
Code availability
NA
References
Adams DL, Economides JR, Horton JC (2015) Co-localization of glutamic acid decarboxylase and vesicular GABA transporter in cytochrome oxidase patches of macaque striate cortex. Vis Neurosci 32:EO26. https://doi.org/10.1017/S0952523815000218
Angelucci A, Bijanzadeh M, Nurminen L, Federer F, Merlin S, Bressloff PC (2017) Circuits and mechanisms for surround modulation in visual cortex. Annu Rev Neurosci 40:425–451. https://doi.org/10.1146/annurev-neuro-072116-031418
Beaulieu C, Kisvarday Z, Somogyi P, Cynader M, Cowey A (1992) Quantitative distribution of GABA-immunopositive and –immunonegative neurons and synapses in the monkey striate cortex (area 17). Cereb Cortex 2:295–309. https://doi.org/10.1093/cercor/2.4.295
Blasdel GG, Lund JS (1983) Termination of afferent axons in macaque striate cortex. J Neurosci 3:1389–1413. https://doi.org/10.1523/JNEUROSCI.03-07-01389.1983
Born RT, Tootell RBH (1991) Spatial frequency tuning of single units in macaque supragranular striate cortex. Proc Natl Acad Sci 88:7066–7070. https://doi.org/10.1073/pnas.88.16.7066
Borra E, Rockland KS (2011) Projections to early visual areas V1 and V2 in the calcarine fissure from parietal association areas in the macaque. Fron Neuroanat 5:35. https://doi.org/10.3389/fnana.2011.00035
Bryant KL, Suwyn C, Reding KM, Smiley JF, Hackett TA, Preuss TM (2012) Evidence for ape and human specializations in geniculostriate projections from VGLUT2 immunohistochemistry. Brain Behav Evol 80:210–221. https://doi.org/10.1159/000341135
Callaway EM (1998) Local circuits in primary visual cortex of the macaque monkey. Annu Rev Neurosci 21:47–74. https://doi.org/10.1023/a:1024165824469
Casagrande VA, Yazar F, Jones KD, Ding Y (2007) The Morphology of the koniocellular axon pathway in the macaque monkey. Cereb Cortex 17:2334–2345. https://doi.org/10.1093/cercor/bhl142
Celio MR, Scharer L, Morrison JH, Norman AW, Bloom FE (1986) Calbindin immunoreactivity alternates with cytochrome c-oxidase-rich zones in some layers of the primate visual cortex. Nature 323:715–717. https://doi.org/10.1038/323715a0
Cembrowski M, Spruston N (2019) Heterogeneity within classical cell types is the rule: lessons from hippocampal pyramidal neurons. Nat Neuro Rev 20:193–204. https://doi.org/10.1038/s41583-019-0125-5
Chatterjee S, Ohki K, Reid RC (2021) Chromatic micromaps in primary visual cortex. Nat Commun 12:2315. https://doi.org/10.1038/s41467-021-22488-3
Coppola JJ, Ward NJ, Jadi MP, Disney AA (2016) Modulatory compartments in cortex and local regulation of cholinergic tone. Physiol Paris 110:3–9. https://doi.org/10.1016/j.jphysparis.2016.08.001
Crowley JC, Katz LC (2002) Ocular dominance development revisited. Curr Opin Neurobiol 12:104–109. https://doi.org/10.1016/s0959-4388(02)00297-0
D’Souza RD, Bista P, Meier AM, Ji W, Burkhalter A (2019) Spatial clustering of inhibition in mouse primary visual cortex. Neuron 104:1–13. https://doi.org/10.1016/j.neuron.2019.09.020
DeFelipe J (2011) The evolution of the brain, the human nature of cortical circuits, and intellectual creativity. Fron Neuroanat 5:29. https://doi.org/10.3389/fnana.2011.00029
Disney AA, Aoki C (2008) Muscarinic acetycholine receptors in macaque V1 are most frequently expressed by parvalbumin-immunoreactive neurons. J Comp Neurol 507:1748–1762. https://doi.org/10.1002/cne.21616
Disney AA, Aoki C, Hawken MJ (2012) Cholinergic suppression of visual responses in primate V1 is mediated by GABAergic inhibition. J Neurophysiol 108:1907–1923. https://doi.org/10.1152/jn.00188.2012:10.1152/jn.00188.201
Dyck RH, Cynader MS (1993) An interdigitated columnar mosaic of cytochrome oxidase, zinc, and neurotransmitter-related molecules in cat and monkey visual cortex. Proc Natl Acad Sci USA 90:9066–9069. https://doi.org/10.1073/pnas.90.19.9066
Dyck RH, Chaudhuri A, Cynader MS (2003) Experience-dependent regulation of the zincergic innervation of visual cortex in adult monkeys. Cereb Cortex 13:1094–1109. https://doi.org/10.1093/cercor/13.10.1094
Economides JR, Sincich LC, Adams DL, Horton JC (2011) Orientation tuning of cytochrome oxidase patches in macaque primary visual cortex. Nat Neurosci 14:1574–1580. https://doi.org/10.1038/nn.2958
Farias MF, Gattass R, Pinon MC, Ungerlieder LG (1997) Tangential distribution of cytochrome oxidase-rich blobs in the primary visual cortex of macaque monkeys. J Comp Neurol 386:217–228. https://doi.org/10.1002/(sici)1096-9861(19970922)386:2%3c217::aid-cne4%3e3.0.co;2-4
Fitzpatrick D, Itoh K, Diamond IT (1983) The laminar organization of the lateral geniculate body and the striate cortex in the squirrel monkey (Saimiri sciureus). J Neurosci 3:673–702. https://doi.org/10.1523/JNEUROSCI.03-04-00673.1983
Fitzpatrick D, Lund JS, Schmechel DE, Towles AC (1987) Distribution of GABAergic neurons and axon terminals in the macaque striate cortex. J Comp Neurol 264:73–91. https://doi.org/10.1002/cne.902640107
Freese JL, Amaral DG (2005) The Organisation of projections from the amygdala to visual cortical areas TE and V1 in the macaque monkey. J Comp Neurol 486:295–317. https://doi.org/10.1002/cne.20520
Gabbott PLA (2003) Radial organisation of neurons and dendrites in human cortical areas 25, 32, and 32’. Brain Res 992:298–304. https://doi.org/10.1016/j.brainres.2003.08.054
Garcia-Marin V, Ahmed TH, Afzal YC, Hawken MJ (2013) Distribution of vesicular glutamate transporter 2 (VGluT2) in the primary visual cortex of the macaque and human. J Comp Neurol 521:130–151. https://doi.org/10.1002/cne.23165
Garcia-Marin V, Sundiang M, Hawken MJ (2015) Reduced density of geniculocortical terminals in foveal layer 4A in the macaque primary visual cortex: relationship to S-cone density. Brain Struct Funct 220:2783–2796. https://doi.org/10.1007/s00429-014-0826-5
Garg AK, Peichao L, Rashid MS, Callaway EM (2019) Color and orientation are jointly coded and spatially organized in primate primary visual cortex. Science 364:1275–1279. https://doi.org/10.1126/science.aaw5868
Hawken MJ, Shapley RM, Disney AA, Garcia-Marin V, Henrie A, Henry CA, Johnson EN et al (2020) Functional clusters of neurons in layer 6 of macaque V1. J Neurosci 40:2445–2457. https://doi.org/10.1523/JNEUROSCI.1394-19.2020
Hendrickson AE, Hunt SP, Wu J-Y (1981) Immunocytochemical localization of glutamic acid decarboxylase in monkey striate cortex. Nature 292:605–607. https://doi.org/10.1038/292605a0
Hendry SHC, Reid RC (2000) The Koniocellular pathway in primate vision. Annu Rev Neurosci 23:127–153. https://doi.org/10.1146/annurev.neuro.23.1.127
Hendry SH, Yoshioka T (1994) A neurochemically distinct third channel in the macaque dorsal lateral geniculate nucleus. Science 264:575–577. https://doi.org/10.1126/science.8160015
Hendry SHC, Jones EG, Burstein N (1988) Activity-Dependent regulation of tachykinin-like immunoreactivity in neurons of monkey visual cortex. J Neurosci 8:1225–1238. https://doi.org/10.1523/JNEUROSCI.08-04-01225.1988
Herrero JL, Roberts MJ, Delicato LS, Gieselmann MA, Dayan P, Thiele A (2008) Acetylcholine contributes through muscarinic receptors to attentional modulation in V1. Nature 454:1110–1112. https://doi.org/10.1038/nature07141
Hevner RF, Wong-Riley MTT (1992) Entorhinal cortex of the human, monkey, and rat: metabolic map as revealed by cytochrome oxidase. J Comp Neurol 326:451–469. https://doi.org/10.1002/cne.903260310
Horton JC (1984) Cytochrome oxidase patches: a new cytoarchitectonic feature of monkey visual cortex. Philos Trans R Soc Lond B Biol Sci 304:199–253. https://doi.org/10.1098/rstb.1984.0021
Horton JC, Adams DL (2005) The cortical column: a structure without a function. Philos Trans R Soc Lond B Biol Sci 360:837–862. https://doi.org/10.1098/rstb.2005.1623
Hubener M, Bolz J (1992) Relationships between dendritic morphology and cytochrome oxidase compartments in monkey striate cortex. J Comp Neurol 324:67–80. https://doi.org/10.1002/cne.903240106
Ichinohe N, Rockland KS (2002) Parvalbumin positive dendrites co-localize with apical dendritic bundles in rat retrosplenial cortex. NeuroReport 13:757–761. https://doi.org/10.1097/00001756-200205070-00005
Ichinohe N, Fujiyama F, Kaneko T, Rockland KS (2003) Honeycomb-like mosaic at the border of layers 1 and 2 in the cerebral cortex. J Neurosci 23:1372–1382. https://doi.org/10.1523/JNEUROSCI.23-04-01372.2003
Ichinohe N, Matsushita A, Ohta K, Rockland KS (2010) Pathway specific utilization of synaptic zinc in the macaque ventral visual cortical areas. Cereb Cortex 20:2818–2831. https://doi.org/10.1093/cercor/bhq028
Ji W, Gamanut R, Bista P, D’Souza RD, Wang Q, Burkhalter A (2015) Modularity in the organization of mouse primary visual cortex. Neuron 87:632–643. https://doi.org/10.1016/j.neuron.2015.07.004
Johnson JK, Casgrande VA (1995) Distribution of calcium-binding proteins within the parallel visual pathways of a primate (Galago crassicaudatus). J Comp Neurol 356:238–260. https://doi.org/10.1002/cne.90356020
Ju N, Li Y, Liu F, Jiang H, Macknik SL, Martinez-Conde S, Tang S (2020) Spatiotemporal functional organization of excitatory synaptic inputs onto macaque V1 neurons. Nat Commun 11:698. https://doi.org/10.1038/s41467-020-14501-y
Karimi A, Odenthal J, Drawitsch F, Boergens KM, Helmstaedter M (2020) Cell-type specific innervation of cortical pyramidal cells at their apical dendrite. Elife 9:e46876. https://doi.org/10.7554/eLife.46876
Kelly SM, Raudales R, He M, Lee JH, Kim Y, Gibb LG, Wu P, Matho K, Osten P, Graybiel AM, Huang ZJ (2018) Radial lineage progression and differential intermediate progenitor amplification underlie striatal compartments and circuit organization. Neuron 99:345–361. https://doi.org/10.1016/j.neuron.2018.06.021
Kim EJ, Zhang Z, Huang L, Ito-Cole T, Jacobs MW, Juavinett AL et al (2020) Extraction of distinct neuronal cell types from within a genetically continuous population. Neuron 107:274–282. https://doi.org/10.1016/j.neuron.2020.04.018
Koestinger G, Martin KAC, Roth S, Rusch ES (2017) Synaptic connections formed by patchy projections of pyramidal cells in the superficial layers of cat visual cortex. Brain Struct Funct 222:3025–3042. https://doi.org/10.1007/s00429-017-1384-4
Kondo H, Tanaka K, Hashikawa T, Jones EG (1999) Neurochemical gradients along monkey sensory cortical pathways: calbindin-immunoreactive pyramidal neurons in layers II and III. Eur J Neuro 11:4197–4203. https://doi.org/10.1046/j.1460-9568.1999.00844.x
Krueger J, Disney AA (2019) Structure and function of dual-source cholinergic modulation in early vision. J Comp Neurol 527:738–750. https://doi.org/10.1002/cne.24590
Kuljis RO, Rakic P (1989) Neuropeptide Y-containing neurons are situated predominantly outside cytochrome oxidase puffs in macaque visual cortex. Vis Neurosci 2:57–62. https://doi.org/10.1017/s0952523800004326
Levanthal AG, Thompson KG, Liu D, Zhou Y, Ault SJ (1995) Concomitant sensitivity to orientation, direction, and color of cells in layers 2, 3, and 4 of monkey striate cortex. J Neurosci 15:1808–1818. https://doi.org/10.1523/JNEUROSCI.15-03-01808.1995
Li M, Song XM, Xu T, Hu D, Roe AW, Li C-Y (2019) Subdomains within orientation columns of primary visual cortex. Sci Adv 5:eaaw0807. https://doi.org/10.1126/sciadv.aaw0807
Lin CS, Nicolelis MA, Schneider JS, Chapin JK (1990) A major direct GABAergic pathway from zona incerta to neocortex. Science 248:1553–1556. https://doi.org/10.1126/science.2360049
Livingstone MS, Hubel DH (1982) Thalamic inputs to cytochrome oxidase-rich regions in monkey visual cortex. Proc Natl Acad Sci USA 79:6098–6101. https://doi.org/10.1073/pnas.79.19.6098
Livingstone MS, Hubel DH (1984a) Anatomy and physiology of a color system in the primate visual cortex. J Neurosci 4:309–356. https://doi.org/10.1523/JNEUROSCI.04-01-00309.1984
Livingstone MS, Hubel DH (1984b) Specificity of intrinsic connections in primate primary visual cortex. J Neurosci 4:2830–2835. https://doi.org/10.1523/JNEUROSCI.04-11-02830.1984
Lund JS (1988) Anatomical organization of macaque monkey striate visual cortex. Annu Rev Neurosci 11:253–258. https://doi.org/10.1146/annurev.ne.11.030188.001345
Lund JS, Boothe RG (1975) Interlaminar connections and pyramidal neuron organization in the visual cortex, area 17, of the macaque monkey. J Comp Neurol 159:305–334. https://doi.org/10.1002/cne.901640303
Malach R (1994) Cortical columns as devices for maximizing neuronal diversity. Trends Neurosci 17:101–104. https://doi.org/10.1016/0166-2236(94)90113-9
Marcondes M, Rosa MGP, Fiorani M, Lima B, Gattass R (2017) Distribution of cytochrome oxidase-rich patches in human primary visual cortex. J Comp Neurol 527:614–624. https://doi.org/10.1002/cne.24435
McGuire BA, Gilbert CD, Rivlin PK, Wiesel TN (1991) Targets of horizontal connections in macaque primary visual cortex. J Comp Neurol 305:370–392. https://doi.org/10.1002/cne.903050303
Merigan WH, Maunsell JHR (1993) How parallel are the primate visual pathways? Annu Rev Neurosci 16:369–402. https://doi.org/10.1146/annurev.ne.16.030193.002101
Mesulam M, Hersh LB, Mash C, Geula C (1992) Differential cholinergic innervation within functional subdivisions of the human cerebral cortex: a choline acetyltransferase study. J Comp Neurol 318:316–328. https://doi.org/10.1002/cne.903180308
Moore B, Li K, Kaas JH, Liao C-C, Boal AM, Mavity-Hudson J, Casagrande V (2019) Cortical projections to the two retinotopic maps of primate pulvinar are distinct. J Comp Neurol 527:577–588. https://doi.org/10.1002/cne.24515
Mrzljak L, Levey AI, Rakic P (1996) Selective expression of m2 muscarinic receptor in the parvocellular channel of the primate visual cortex. Proc Natl Acad Sci USA 93:7337–73340. https://doi.org/10.1073/pnas.93.14.7337
Nakashima AS, Dyck RH (2009) Zinc and cortical plasticity. Brain Res Rev 59:347–373. https://doi.org/10.1016/j.brainresrev.2008.10.003
Nassi JJ, Callaway EM (2009) Parallel processing strategies of the primate visual system. Nat Rev Neurosci 10:360–372. https://doi.org/10.1038/nrn2619
Nie F, Wong-Riley MT (1996) Differential glutamatergic innervation in cytochrome oxidase-rich and –poor regions of the macaque striate cortex: quantitative EM analysis of neurons and neuropil. J Comp Neurol 369:571–590. https://doi.org/10.1002/(SICI)1096-9861(19960610)369:4%3c571::AID-CNE7%3e3.0.CO;2-1
Payne BR, Peters A (1989) Cytochrome oxidase patches and Meynert cells in monkey visual cortex. Neurosci 28:353–363. https://doi.org/10.1016/0306-4522(89)90182-6
Peters A (1994) The organisation of the primary visual cortex in the macaque. In: Peters A, Rockland K (eds) Cerebral cortex, vol 10, primary visual cortex in primates. Plenum Press, New York, pp 1–35
Peters A, Sethares C (1991a) Layer IVA of rhesus monkey primary visual cortex. Cereb Cortex 1:445–462. https://doi.org/10.1093/cercor/1.6.445
Peters A, Sethares C (1991b) Organization of pyramidal neurons in area 17 of monkey visual cortex. J Comp Neurol 306:1–23. https://doi.org/10.1002/cne.903060102
Preuss TM, Coleman G (2002) Human-specific organization of primary visual cortex: alternating compartments of dense Cat-301 and calbindin immunoreactivity in layer 4A. Cereb Cortex 12:671–691. https://doi.org/10.1093/cercor/12.7.671
Purves D, LaMantia AS (1990) Numbers of “blobs” in the primary visual cortex of neonatal and adult monkeys. Proc Natl Acad Sci USA 87:5764–5767. https://doi.org/10.1073/pnas.87.15.5764
Rockland KS (1992) Laminar distribution of neurons projecting from area V1 to V2 in macaque and squirrel monkeys. Cereb Cortex 2:38–47. https://doi.org/10.1093/cercor/2.1.38
Rockland KS, Ojima H (2003) Multisensory convergence in calcarine visual areas in macaque monkey. Int J Psychophysiol 50:19–26. https://doi.org/10.1016/s0167-8760(03)00121-1
Rockland KS, Virga A (1990) Organization of individual cortical axons projecting from area V1 (area 17) to V2 (area 18) in the Macaque monkey. Vis Neurosci 4:11–28. https://doi.org/10.1017/s095252380000273x
Rockland KS, Andresen J, Cowie RJ, Robinson DL (1999) Single axon analysis of pulvinocortical connections to several visual areas in the macaque. J Comp Neurol 406:221–250. https://doi.org/10.1002/(sici)1096-9861(19990405)406:2%3c221::aid-cne7%3e3.0.co;2-k
Rockoff EC, Balaram P, Kaas JH (2014) Patchy distributions of myelin and vesicular glutamate transporter 2 align with cytochrome oxidase blobs and interblobs in the superficial layers of primary visual cortex. Eye Brain 6:19–27. https://doi.org/10.2147/EB.S59797
Sawatari A, Callaway EM (2000) Diversity and cell type specificity of local excitatory connections to neurons in layer 3B of monkey primary visual cortex. Neuron 25:459–471. https://doi.org/10.1016/s0896-6273(00)80908-3
Scholl B, Fitzpatrick D (2020) Cortical synaptic architecture supports flexible sensory computations. Curr Opin Neurobiol 64:41–45. https://doi.org/10.1016/j.conb.2020.01.013
Scholl B, Wilson DE, Fitzpatrick D (2017) Local order within global disorder: synaptic architecture of visual space. Neuron 96:1127–1138. https://doi.org/10.1016/j.neuron.2017.10.017
Sensi SL, Paoletti P, Koh J-Y, Aizenman E, Bush AI, Hershfinkel M (2011) The neurophysiology and pathology of zinc. J Neurosci 31:16076–16085. https://doi.org/10.1523/JNEUROSCI.3454-11.2011
Shipp S, Zeki S (1989) The organization of connections between areas V5 and V1 in macaque monkey visual cortex. Eur J Neurosci 1:309–332. https://doi.org/10.1111/j.1460-9568.1989.tb00798.x
Sincich LC, Horton JC (2005) The circuitry of V1 and V2: Integration of color, form, and motion. Annu Rev Neurosci 28:303–326. https://doi.org/10.1146/annurev.neuro.28.061604.135731
Sincich LC, Park KF, Wohlgemuth MJ, Horton JC (2004) Bypassing V1: a direct geniculate input to area MT. Nat Neurosci 7:1123–1128. https://doi.org/10.1038/nn1318
Stettler DD, Das A, Bennett J, Gilbert CD (2002) Lateral connectivity and contextual interactions in macaque primary visual cortex. Neuron 36:739–750. https://doi.org/10.1016/s0896-6273(02)01029-2
Takahata T (2016) What does cytochrome oxidase histochemistry represent in the visual cortex? Fron Neuroanat 10:79. https://doi.org/10.3389/fnana.2016.00079
Takahata T, Komatsu Y, Watakabe A, Hashikawa T, Tochitani S, Yamamori T (2009) Differential expression of occ’-related genes in adult monkey visual cortex. Cereb Cortex 19:1937–1951. https://doi.org/10.1093/cercor/bhn220
Tigges M, Tigges J, Rees H, Rye D, Levey AI (1997) Distribution of muscarinic cholinergic receptor proteins m1 to m4 in area 17 of normal and monocularly deprived rhesus monkeys. J Comp Neurol 388:130–145 (PMID: 9364243)
Tootell RB, Silverman MS, Hamilton SL, De Valois RL, Switkes E (1988a) Functional anatomy of macaque striate cortex. III. Color. J Neurosci 8:1569–1593. https://doi.org/10.1523/JNEUROSCI.08-05-01569.1988
Tootell RB, Silverman MS, Hamilton SL, Switkes E, De Valois RL (1988b) Functional anatomy of macaque striate cortex. V. Spatial frequency. J Neurosci 8:1610–1624. https://doi.org/10.1523/JNEUROSCI.08-05-01610.1988
Van Brederode JF, Mulligan KA, Hendrickson AE (1990) Calcium-binding proteins as markers for subpopulations of GABAergic neurons in monkey striate cortex. J Comp Neurol 298:1–22. https://doi.org/10.1002/cne.902980102
Vanni S, Hokkanen H, Werner F, Angelucci A (2020) Anatomy and physiology of macaque visual cortical areas V1, V2, and V5/MT: Bases for biologically realistic models. Cereb Cortex 30:3483–3517. https://doi.org/10.1093/cercor/bhz322
Watakabe A, Komatsu Y, Sadakane O, Shimegi S, Takahata T, Higo N, Tochitani S, Hashikawa T, Naito T, Osaki H, Sakamoto H, Okamoto M, Ishikawa A, Hara S, Akasaki T, Sato H, Yamamori T (2009) Enriched expression of serotonin 1B and 2A receptor genes in macaque visual cortex and their bidirectional modulatory effects on neuronal responses. Cereb Cortex 19:1915–1928. https://doi.org/10.1093/cercor/bhn219
Wong-Riley MTT (1994) Primate visual cortex: dynamic metabolic organization and plasticity revealed by cytochrome oxidase. In: Peters A, Rockland K (eds) Cerebral cortex, vol 10, primary visual cortex in primates. Plenum Press, New York, pp 141–200
Wong-Riley M (2010) Energy metabolism of the visual system. Eye Brain 2:99–116. https://doi.org/10.2147/EB.S9078
Wyss JM, van Groen T, Sripanidkulchai K (1990) Dendritic bundling in layer I of granular retrosplenial cortex: intracellular labeling and selectivity of innervation. J Comp Neurol 295:33–42. https://doi.org/10.1002/cne.902950104
Yabuta NH, Callaway EM (1998) Cytochrome-oxidase blobs and intrinsic horizontal connections of layer 2/3 pyramidal neurons in primate V1. Visual Neurosci 15:1007–1027. https://doi.org/10.1017/s0952523898156018
Yao S, Zhou Q, Li S, Takahata T (2021) Immunoreactivity of vesicular glutamate transporter 2 corresponds to Cytochrome Oxidase-Rich subcompartments in the visual cortex of squirrel monkeys. Fron Neuroanat 15:629473. https://doi.org/10.3389/fnana.2021.629473
Yoshioka T, Blasdel GG, Levitt JB, Lund JS (1996) Relation between patterns of intrinsic lateral connectivity, ocular dominance, and cytochrome oxidase-reactive regions in macaque monkey striate cortex. Cereb Cortex 6:297–310. https://doi.org/10.1093/cercor/6.2.297
Zilles K, Palomero-Gallagher N (2017) Multiple transmitter receptors in regions and layers of the human cerebral cortex. Fron Neuroant 11:78. https://doi.org/10.3389/fnana.2017.00078
Acknowledgements
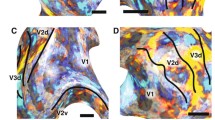
I thank MacBrain Resource for contributing calbindin stained material (in Fig. 1D) for this project (MH113257 to Dr. Alvaro Duque).
Funding
No funding has been received for the present work by the author (ksr). As this is a review paper, issues of informed consent and of treatment of human or animal subjects do not apply. Animal work from previous papers of the Author had been approved at the several relevant institutions (Boston University, Univ. of Iowa, or RIKEN Brain Science Institute) as stated in the original research papers. Credit has been explicitly given to any original research mentioned in this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no direct or indirect conflicts of interest for this work.
Consent to participate
NA (i.e., I am sole Author).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rockland, K.S. Cytochrome oxidase “blobs”: a call for more anatomy. Brain Struct Funct 226, 2793–2806 (2021). https://doi.org/10.1007/s00429-021-02360-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02360-2