Abstract
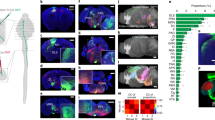
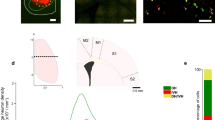
The descending corticospinal (CS) projection has been considered a key element for motor control, which results from direct and indirect modulation of spinal cord pre-motor interneurons in the intermediate gray matter of the spinal cord, which, in turn, influences motoneurons in the ventral horn. The CS tract (CST) is also involved in a selective and complex modulation of sensory information in the dorsal horn. However, little is known about the spinal network engaged by the CST and the organization of CS projections that may encode different cortical outputs to the spinal cord. This study addresses the issue of whether the CS system exerts parallel control on different spinal networks, which together participate in sensorimotor integration. Here, we show that in the adult rat, two different and partially intermingled CS neurons in the sensorimotor cortex activate, with different time latencies, distinct spinal cord neurons located in the dorsal horn and intermediate zone of the same segment. The fact that different populations of CS neurons project in a segregated manner suggests that CST is composed of subsystems controlling different spinal cord circuits that modulate motor outputs and sensory inputs in a coordinated manner.







Similar content being viewed by others
Abbreviations
- CST:
-
Corticospinal tract
- CS:
-
Corticospinal
- PAD:
-
Primary afferent depolarization
- EFPs:
-
Evoked field potentials
- SD:
-
Standard deviation
- M1:
-
Primary motor cortex
- S1:
-
Primary somatosensory cortex
- M2:
-
Secondary motor cortex
- S2:
-
Secondary somatosensory cortex
- HL:
-
Hind limb
- FL:
-
Fore limb
References
Abdelmoumene M, Besson JM, Aleonard P (1970) Cortical areas exerting presynaptic inhibitory action on the spinal cord in cat and monkey. Brain Res 20:327–329. doi:10.1016/0006-8993(70)90301-X
Akintunde A, Buxton DF (1992) Differential sites of origin and collateralization of corticospinal neurons in the rat: a multiple fluorescent retrograde tracer study. Brain Res 575:86–92. doi:10.1016/0006-8993(92)90427-B
Alstermark B, Pettersson LG (2014) Skilled reaching and grasping in the rat: lacking effect of corticospinal lesion. Front Neurol 5:103. doi:10.3389/fneur.2014.00103
Alstermark B, Ogawa J, Isa T (2004) Lack of monosynaptic corticomotoneuronal EPSPs in rats: disynaptic EPSPs mediated via reticulospinal neurons and polysynaptic EPSPs via segmental interneurons. J Neurophysiol 91:1832–1839. doi:10.1152/jn.00820.2003
Andersen P, Eccles JC, Sears TA (1964) Cortically evoked depolarization of primary afferent fibers in the spinal cord. J Neurophysiol 27:63–77
Armand J (1982) The origin, course and terminations of corticospinal fibers in various mammals. Prog Brain Res 57:329–360. doi:10.1016/S0079-6123(08)64136-9
Babalian A, Liang F, Rouiller EM (1993) Cortical influences on cervical motoneurons in the rat: recordings of synaptic responses from motoneurons and compound action potential from corticospinal axons. Neurosci Res 16:301–310. doi:10.1016/0168-0102(93)90041-N
Baker MR, Javid M, Edgley SA (2001) Activation of cerebellar climbing fibres to rat cerebellar posterior lobe from motor cortical output pathways. J Physiol 536:825–839. doi:10.1111/j.1469-7793.2001.00825.x
Benaglia T, Chauveau D, Hunter DR, Young DS (2009) Mixtools: an R package for analyzing finite mixture models. J Stat Softw 32:1–29
Bourane S, Grossmann KS, Britz O, Dalet A, Del Barrio MG, Stam FJ, Garcia-Campmany L, Koch S, Goulding M (2015) Identification of a spinal circuit for light touch and fine motor control. Cell 160:503–515. doi:10.1016/j.cell.2015.01.011
Canedo A (1997) Primary motor cortex influences on the descending and ascending systems. Prog Neurobiol 51:287–335. doi:10.1016/S0301-0082(96)00058-5
Carpenter D, Lundberg A, Norrsell U (1963) Primary afferent depolarization evoked from the sensorimotor cortex. Acta Physiol Scand 59:126–142. doi:10.1111/j.1748-1716.1963.tb02729.x
Casale EJ, Light AR, Rustioni A (1988) Direct projection of the corticospinal tract to the superficial laminae of the spinal cord in the rat. J Comp Neurol 278:275–286. doi:10.1002/cne.902780210
Condés-Lara M, Martínez-Lorenzana G, Rojas-Piloni G, Rodríguez-Jiménez J (2007) Branched oxytocinergic innervations from the paraventricular hypothalamic nuclei to superficial layers in the spinal cord. Brain Res 1160:20–29. doi:10.1016/j.brainres.2007.05.031
Conte WL, Kamishina H, Reep RL (2009) The efficacy of the fluorescent conjugates of cholera toxin subunit B for multiple retrograde tract tracing in the central nervous system. Brain Struct Funct 213:367–373. doi:10.1007/s00429-009-0212-x
Coulter JD, Jones EG (1977) Differential distribution of corticospinal projections from individual cytoarchitectonic fields in the monkey. Brain Res 129:335–340. doi:10.1016/0006-8993(77)90012-9
Dunkerley GB, Duncan D (1969) A light and electron microscopic study of the normal and the degenerating corticospinal tract in the rat. J Comp Neurol 137:155–183. doi:10.1002/cne.901370204
Edgley SA, Jankowska E, Hammar I (2004) Ipsilateral actions of feline corticospinal tract neurons on limb motoneurons. J Neurosci 24:7804–7813. doi:10.1523/JNEUROSCI.1941-04.2004
Eguibar JR, Quevedo J, Jimenez I, Rudomin P (1994) Selective cortical control of information flow through different intraspinal collaterals of the same muscle afferent fiber. Brain Res 643:328–333. doi:10.1016/0006-8993(94)90042-6
Fink AJ, Croce KR, Huang ZJ, Abbott LF, Jessell TM, Azim E (2014) Presynaptic inhibition of spinal sensory feedback ensures smooth movement. Nature 509:43–48. doi:10.1038/nature13276
Galea MP, Darian-Smith I (1994) Multiple corticospinal neuron populations in the macaque monkey are specified by their unique cortical origins, spinal terminations, and connections. Cereb Cortex 4:166–194. doi:10.1093/cercor/4.2.166
Griffin DM, Hoffman DS, Strick PL (2015) Corticomotoneuronal cells are “functionally tuned”. Science 350:667–670. doi:10.1126/science.aaa8035
Groh A, Meyer HS, Schmidt EF, Heintz N, Sakmann B, Krieger P (2010) Cell-type specific properties of pyramidal neurons in neocortex underlying a layout that is modifiable depending on the cortical area. Cereb Cortex 20:826–836. doi:10.1093/cercor/bhp152
Hantman AW, Jessell TM (2010) Clarke’s column neurons as the focus of a corticospinal corollary circuit. Nat Neurosci 13:1233–1239. doi:10.1038/nn.2637
Hattox AM, Nelson SB (2007) Layer V neurons in mouse cortex projecting to different targets have distinct physiological properties. J Neurophysiol 98:3330–3340. doi:10.1152/jn.00397.2007
Hultborn H, Meunier S, Morin C, Pierrot-Deseilligny E (1987a) Assessing changes in presynaptic inhibition of I a fibres: a study in man and the cat. J Physiol 389:729–756. doi:10.1113/jphysiol.1987.sp016680
Hultborn H, Meunier S, Pierrot-Deseilligny E, Shindo M (1987b) Changes in presynaptic inhibition of Ia fibres at the onset of voluntary contraction in man. J Physiol 389:757–772. doi:10.1113/jphysiol.1987.sp016681
Jankowska E, Padel Y, Tanaka R (1976) Disynaptic inhibition of spinal motoneurones from the motor cortex in the monkey. J Physiol 258:467–487. doi:10.1113/jphysiol.1976.sp011431
Jones EG, Wise SP (1977) Size, laminar and columnar distribution of efferent cells in the sensory-motor cortex of monkeys. J Comp Neurol 175:391–438. doi:10.1002/cne.901750403
Joosten EA, Gribnau AA (1988) Unmyelinated corticospinal axons in adult rat pyramidal tract. An electron microscopic tracer study. Brain Res 459:173–177. doi:10.1016/0006-8993(88)90300-9
Kamiyama T, Kameda H, Murabe N, Fukuda S, Yoshioka N, Mizukami H, Ozawa K, Sakurai M (2015) Corticospinal tract development and spinal cord innervation differ between cervical and lumbar targets. J Neurosci 35:1181–1191. doi:10.1523/JNEUROSCI.2842-13.2015
Killackey HP, Koralek KA, Chiaia NL, Rhodes RW (1989) Laminar and areal differences in the origin of the subcortical projection neurons of the rat somatosensory cortex. J Comp Neurol 282:428–445. doi:10.1002/cne.902820309
Lawrence DG, Kuypers HG (1968) The functional organization of the motor system in the monkey. I. The effects of bilateral pyramidal lesions. Brain 91:1–14. doi:10.1093/brain/91.1.1
Leenen LP, Meek J, Posthuma PR, Nieuwenhuys R (1985) A detailed morphometrical analysis of the pyramidal tract of the rat. Brain Res 359:65–80. doi:10.1016/0006-8993(85)91413-1
Lemon RN (2008) Descending pathways in motor control. Ann Rev Neurosci 31:195–218. doi:10.1146/annurev.neuro.31.060407.125547
Lemon RN, Griffiths J (2005) Comparing the function of the corticospinal system in different species: organizational differences for motor specialization? Muscle Nerve 32:261–279. doi:10.1002/mus.20333
Lomeli J, Quevedo J, Linares P, Rudomin P (1998) Local control of information flow in segmental and ascending collaterals of single afferents. Nature 395:600–604. doi:10.1038/26975
Maeda H, Fukuda S, Kameda H, Murabe N, Isoo N, Mizukami H, Ozawa K, Sakurai M (2016) Corticospinal axons make direct synaptic connections with spinal motoneurons innervating forearm muscles early during postnatal development in the rat. J Physiol 594:189–205. doi:10.1113/JP270885
Maier MA, Perlmutter SI, Fetz EE (1998) Response patterns and force relations of monkey spinal interneurons during active wrist movement. J Neurophysiol 80:2495–2513
Martinez-Lorenzana G, Machín R, Avendaño C (2001) Definite segregation of cortical neurons projecting to the dorsal column nuclei in the rat. Neuroreport 12:413–416
Mediratta NK, Nicoll JA (1983) Conduction velocities of corticospinal axons in the rat studied by recording cortical antidromic responses. J Physiol 336:545–561. doi:10.1113/jphysiol.1983.sp014597
Miller MW (1987) The origin of corticospinal projection neurons in rat. Exp Brain Res 67:339–351. doi:10.1007/BF00248554
Moreno-Lopez Y, Perez-Sanchez J, Martinez-Lorenzana G, Condes-Lara M, Rojas-Piloni G (2013) Cortical presynaptic control of dorsal horn C-afferents in the rat. PLoS One 8:e69063. doi:10.1371/journal.pone.0069063
Nakajima K, Maier MA, Kirkwood PA, Lemon RN (2000) Striking differences in transmission of corticospinal excitation to upper limb motoneurons in two primate species. J Neurophysiol 84:698–709
Nelson RJ (1996) Interactions between motor commands and somatic perception in sensorimotor cortex. Curr Opin Neurobiol 6:801–810. doi:10.1016/S0959-4388(96)80031-6
Papp EA, Leergaard TB, Calabrese E, Johnson GA, Bjaalie JG (2014) Waxholm space atlas of the Sprague Dawley rat brain. Neuroimage 97:374–386. doi:10.1016/j.neuroimage.2014.04.001
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates. Academic Press, London
Porter R, Lemon R (1993) Corticospinal function and voluntary movement. Oxford University Press, Oxford
Rathelot JA, Strick PL (2009) Subdivisions of primary motor cortex based on cortico-motoneuronal cells. PNAS 106:918–923. doi:10.1073/pnas.0808362106
Rojas-Piloni G, Martinez-Lorenzana G, Condes-Lara M, Rodriguez-Jimenez J (2010) Direct sensorimotor corticospinal modulation of dorsal horn neuronal C-fiber responses in the rat. Brain Res 1351:104–114. doi:10.1016/j.brainres.2010.06.010
Rudomin P, Schmidt RF (1999) Presynaptic inhibition in the vertebrate spinal cord revisited. Exp Brain Res 129:1–37. doi:10.1007/s002210050933
Rudomin P, Solodkin M, Jimenez I (1986) PAD and PAH response patterns of group Ia- and Ib-fibers to cutaneous and descending inputs in the cat spinal cord. J Neurophysiol 56:987–1006
Schieber MH (2007) Chapter 2 Comparative anatomy and physiology of the corticospinal system. Hand Clin Neurol 82:15–37
Seki K, Perlmutter SI, Fetz EE (2003) Sensory input to primate spinal cord is presynaptically inhibited during voluntary movement. Nat Neurosci 6:1309–1316. doi:10.1038/nn1154
Sengul G (2013) Atlas of the spinal cord of the rat, mouse, marmoset, rhesus, and human, 1st edn. Elsevier Academic Press, Boston
Shepherd GM (2013) Corticostriatal connectivity and its role in disease. Nat Rev Neurosci 14:278–291. doi:10.1038/nrn3469
Shinoda Y, Yamaguchi T, Futami T (1986) Multiple axon collaterals of single corticospinal axons in the cat spinal cord. J Neurophysiol 55:425–448
Stewart M, Quirk GJ, Amassian VE (1990) Corticospinal responses to electrical stimulation of motor cortex in the rat. Brain Res 508:341–344. doi:10.1016/0006-8993(90)90421-7
Sunshine MD, Cho FS, Lockwood DR, Fechko AS, Kasten MR, Moritz CT (2013) Cervical intraspinal microstimulation evokes robust forelimb movements before and after injury. J Neural Eng 10(3):036001. doi:10.1088/1741-2560/10/3/036001
Suter BA, Shepherd GM (2015) Reciprocal interareal connections to corticospinal neurons in mouse M1 and S2. J Neurosci 35:2959–2974. doi:10.1523/JNEUROSCI.4287-14.2015
Tosolini and Morris (2012) Spatial characterization of the motor neuron columns supplying the rat forelimb. Neuroscience 200:19–30. doi:10.1016/j.neuroscience.2011.10.054
Towe AL, Harding GW (1970) Extracellular microelectrode sampling bias. Exp Neurol 29:366–381. doi:10.1016/0014-4886(70)90065-8
Ullan J, Artieda J (1981) Somatotopy of the corticospinal neurons in the rat. Neurosci Lett 21:13–18. doi:10.1016/0304-3940(81)90049-5
Welniarz Q, Dusart I, Roze E (2016) The corticospinal tract: evolution, development, and human disorders. Dev Neurobiol. doi:10.1002/dneu.22455
Acknowledgements
We thank Jessica Gonzalez Norris for revising the grammatical aspects of the manuscript; Cutberto Dorado, Nydia Hernández, Alejandra Castilla, Martín García Servin, Adriana Gonzalez Gallardo, and Anaid Antaramian for technical assistance as well as Edna Hurtado who performed preliminary experiments. Supported by grants from CONACYT Fronteras de la Ciencia 846 (GRP) and 181508 (LC) and PAPIIT (IN200615). Rafael Olivares Moreno is a doctoral student from Programa de Doctorado en Ciencias Biomédicas, Universidad Nacional Autónoma de México (UNAM) and received fellowship 317553 from CONACYT.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2017_1447_MOESM1_ESM.tif
Supplementary material 1 Supplementary Fig. 1 Relationship between the number of pulses of the cortical stimulation train and amplitude of the intraspinal LFP. Relationship between the number of pulses of the cortical stimulation train and magnitude of the short-latency (green triangles) and long-latency (red circles) components of the intraspinal EFP. The continuous lines are the respective best sigmoidal fittings. Inset shows representative superimposed averaged (n = 16) intraspinal EFPs produced with 1, 3, and 5 pulses in the same experiment. Green and red arrows indicate short- and long-latency components of the EFP, respectively (TIFF 5214 kb)
429_2017_1447_MOESM2_ESM.tif
Supplementary material 2 Supplementary Fig. 2 Spinal cord injection sites of all the experiments analyzed. a Cervical spinal cord sections showing the core of the neuronal tracer’s injection sites (dashed lines) in three different experiments (cholera toxin subunit B conjugated with Alexa 488 in green and Alexa 594 in red). b Average of the injection site areas corresponding to DH and IZ. c–d The same but for the injections in lumbar segments. The measured areas of the injection sites in cervical and lumbar segments show a similar size (p > 0.5, Mann–Whitney U). Scale bars 500 µm (TIFF 30694 kb)
429_2017_1447_MOESM3_ESM.wmv
Supplementary material 3 Supplementary video 1 Identification of retrogradely labeled CS cells in large-scale mosaic images (WMV 5799 kb)
429_2017_1447_MOESM4_ESM.wmv
Supplementary material 4 Supplementary video 2 3D model of the rat brain showing the landmarks and density maps of the CS cells projecting to the dorsal horn (red) and intermediate zone (green) (WMV 7651 kb)
Rights and permissions
About this article
Cite this article
Olivares-Moreno, R., Moreno-Lopez, Y., Concha, L. et al. The rat corticospinal system is functionally and anatomically segregated. Brain Struct Funct 222, 3945–3958 (2017). https://doi.org/10.1007/s00429-017-1447-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-017-1447-6