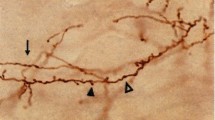
Abstract
Neural connections are implemented by axons of different diameters, whose spectrum increases depending on species and areas. Axon diameter determines conduction velocity and is proportional to the size of the cell body of origin. We describe that in motor, callosal connections of the monkey thick axons distribute larger boutons than thin axons, suggesting that faster axons also release more neurotransmitter at their termination, probably activating more powerfully their targets.









Similar content being viewed by others
References
Anderson JC, Binzegger T, Martin KA, Rockland KS (1998) The connection from cortical area V1 to V5: a light and electron microscopic study. J Neurosci 18:10525–10540
Anderson JC, Binzegger T, Douglas RJMK (2002) Chance or design? Some specific considerations concerning synaptic boutons in cat visual cortex. J Neurocytol 13:211–229
Berbel P, Innocenti GM (1988) The developmental of the corpus callosum in cats: a light- and electron-microscopic study. J Comp Neurol 276:132–156
Caminiti R, Ghaziri H, Galuske R et al (2009) Evolution amplified processing with temporally dispersed slow neuronal connectivity in primates. Proc Natl Acad Sci USA 106:19551–19556. doi:10.1073/pnas.0907655106
Caminiti R, Carducci F, Piervincenzi C et al (2013) Diameter, length, speed, and conduction delay of callosal axons in macaque monkeys and humans: comparing data from histology and magnetic resonance imaging diffusion tractography. J Neurosci 33:14501–14511. doi:10.1523/JNEUROSCI.0761-13.2013
Ding Y, Casagrande VA (1998) Synaptic and neurochemical characterization of parallel pathways to the cytochrome oxidase blobs of promate visual cortex. J Comp Neurol 391:429–433
Gasser HS, Erlanger J (1926) The role played by the sizes of the constituent fibers of a nerve trunk in determining the form of its action potential. Am J Physiol 80:522–547
Gasser HS, Grundfest H (1939) Axon diameters in relation to the spike dimensions and the conduction velocity in mammalian A fibers. Americ J Physiol 127:393–414
Groh A, de Kock CP, Wimmer VC et al (2008) Driver or coincidence detector: modal switch of a corticothalamic giant synapse controlled by spontaneous activity and short-term depression. J Neurosci 28:9652–9663. doi:10.1523/JNEUROSCI.1554-08.2008
Harata NC, Aravanis AM, Tsien RW (2006) Kiss-and-run and full-collapse fusion as modes of exoendocytosis in neurosecretion. J Neurochem 97:1546–1570
Hellwig B, Schiiz A, Aertsen A (1994) Synapses on axon collaterals of pyramidal cells are spaced at random intervals: a Golgi study in the mouse cerebral cortex. Biol Cybemet 12:1–12
Holderith N, Lorincz A, Katona G, Rózsa B, Kulik A, Watanabe M, Nusser Z (2012) Release probability of hippocampal glutamatergic terminals scales with the size of the active zone. Nat Neurosci 15:988–997. doi:10.1038/nn.3137
Houzel JC, Milleret C, Innocenti GM (1994) Morphology of callosal axons interconnecting areas 17 and 18 of the cat. Eur J Neurosci 6:898–917. doi:10.1111/j.1460-9568.1994.tb00585.x
Innocenti GM (1995) Exuberant development of connections, and its possible permissive role in cortical evolution. Trends Neurosci 18:397–402. doi:10.1016/0166-2236(95)93936-R
Innocenti GM (2011) Development and evolution. Two determinants of cortical connectivity. In: Progress in brain research, p 65–75
Innocenti GM, Lehmann P, Houzel JC (1994) Computational structure of visual callosal axons. Eur J Neurosci 6:918–935. doi:10.1111/j.1460-9568.1994.tb00586.x
Innocenti GM, Vercelli A, Caminiti R (2014) The diameter of cortical axons depends both on the area of origin and target. Cereb Cortex 24:2178–2188. doi:10.1093/cercor/bht070
Innocenti GM, Caminiti R, Aboitiz F et al (2015) Comments on the paper by Horowitz et al (2014). Brain Struct Funct 220:1789–1790. doi:10.1007/s00429-014-0974-7
Innocenti GM, Dyrby TB, Winther-Andersen K, Rouiller EM, Caminiti R (2016) The crossed projection to the striatum in two species of monkey and in humans: behavioral and evolutionary significance. Cereb Cortex (in press)
Innocenti GM, Carlén M, Dyrby TB (2016b) The diameter of cortical axons and their relevance to neural computing. Axons and Brain architecture. Elsevier, Amsterdam, pp 317–335
Künzle H (1976) Alternating afferent zones of high and low axon terminal density within the macaque motor cortex. Brain Res 106:365–370. doi:10.1016/0006-8993(76)91031-3
Makarov VA, Schmidt KE (2008) Stimulus-dependent interaction between the visual areas 17 and 18 of the 2 hemispheres of the ferret (Mustela putorius). Cereb Cortex 18:1951–1960. doi:10.1093/cercor/bhm222
Merchán-Pérez A, Rodríguez JR, González S et al (2014) Three-dimensional spatial distribution of synapses in the neocortex: a dual-beam electron microscopy study. Cereb Cortex 24:1579–1588. doi:10.1093/cercor/bht018
Montes J, Peña JM, De Felipe J et al (2015) The influence of synaptic size on AMPA receptor activation: a monte carlo model. PLoS One 10:1–18. doi:10.1371/journal.pone.0130924
Olivares R, Montiel J, Aboitiz F (2001) Species differences and similarities in the fine structure of the mammalian corpus callosum. Brain Behav Evol 57:98–105
Parent M, Parent A (2006) Single-axon tracing study of corticostriatal projections arising from primary motor cortex in primates. J Comp Neurol 496:202–213. doi:10.1002/cne.20925
Perge JA, Niven JE, Mugnaini E et al (2012) Why do axons differ in caliber? J Neurosci 32:626–638. doi:10.1523/JNEUROSCI.4254-11.2012
Petrof I, Sherman SM (2013) Functional significance of synaptic terminal size in glutamatergic sensory pathways in thalamus and cortex. J Physiol 591:3125–3131
Pierce JP, Lewin GR (1994) An ultrastructural size principle. Neuroscience 58:441–446. doi:10.1016/0306-4522(94)90071-X
Rockland KS (1992) Configuration, in serial recronstruction, of individual axons projecting from area V2 to V4 in the macaque monkey. Cereb Cortex 2:353–374
Rollenhagen A, Klook K, Sätzler K, Qi G, Anstötz M, Feldmeyer D, Lübke JHR (2015) Structural determinants underly the high efficacy of synaptic transmission and plasticity at synaptic boutons in layer 4 of the adult rat barrel cortex. Brain Struct Funct 220:3185–3209
Rouiller EM, Welker E (2000) A comparative analysis of the morphology of the corticothalamic projections in mammals. Brain Res Bull 53:727–741
Rushton WAH (1951) A theory of the effects of fibre size in medullated nerve. J Physiol 115:101–122
Schikorski T, Stevens CF (1997) Quantitative ultrastructural analysis of hippocampal excitatory synapses. J Neurosci 17:5858–5867
Segev I, Schneidman E (1999) Axons as computing devices: basic insights gained from models. J Physiol Paris 93:263–270. doi:10.1016/S0928-4257(00)80055-8
Senn W, Wyler K, Clamann HP et al (1997) Size principle and information theory. Biol Cybern 76:11–22. doi:10.1007/s004220050317
Swadlow HA (1985) Physiological properties of individual cerebral axons studied in vivo for as long as one year. J Neurophysiol 54:1346–1362
Tarusawa E, Matsui K, Budisantoso T et al (2009) Input-specific intrasynaptic arrangements of ionotropic glutamate receptors and their impact on postsynaptic responses. J Neurosci 29:12896–12908. doi:10.1523/JNEUROSCI.6160-08.2009
Tettoni L, Gheorghita-Baechler F, Bressoud R et al (1998) Constant and variable aspects of axonal phenotype in cerebral cortex. Cereb Cortex 8:543–552. doi:10.1093/cercor/8.6.543
Tomasi S, Caminiti R, Innocenti GM (2012) Areal differences in diameter and length of corticofugal projections. Cereb Cortex 22:1463–1472. doi:10.1093/cercor/bhs011
Wang SS, Shultz JR, Burish MJ et al (2009) Shaping of white matter composition by biophysical scaling constraints. J Neurosci 28:4047–4056. doi:10.1523/JNEUROSCI.5559-05.2008
Waxman SG, Bennett MVL (1972) Relative conduction velocities of small myelinated and non-myelinated fibers in the central nervous system. Nature 238:217–219
Zhang Q, Li Y, Tsien RW (2009) The dynamic control of kiss-and-run and vesicular reuse probed with single nanoparticles. Science 323:1448–1453
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2016_1266_MOESM1_ESM.tif
Supplementary Fig. 1. Thick axon terminals were sampled at a slightly more superficial location than thin axon terminals and unlike the latter they showed a moderate tendency to larger diameter with depth (TIFF 1926 kb)
429_2016_1266_MOESM2_ESM.tif
Supplementary Fig. 2. Linear relation between axonal size and soma size in callosally projecting, neurons in motor and prefrontal areas of the monkey. The neurons were retrogradely filled with dextrane. Modified from Tomasi et al. (2012) (TIFF 1287 kb)
Rights and permissions
About this article
Cite this article
Innocenti, G.M., Caminiti, R. Axon diameter relates to synaptic bouton size: structural properties define computationally different types of cortical connections in primates. Brain Struct Funct 222, 1169–1177 (2017). https://doi.org/10.1007/s00429-016-1266-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-016-1266-1