Abstract
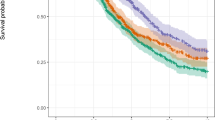
Minichromosome maintenance complex component 6 (MCM6) is involved in initiating DNA replication and is upregulated during licensed G0 phase of the cell cycle. This early expression permits its labeling of more proliferating cells than those by Ki-67. Here using a cohort of 89 endometrioid adenocarcinoma, we report findings made on the prognostic value of MCM6 based on immunohistochemical labeling index (LI) of the protein in comparison with that of Ki67 as no such information is currently available. Additionally, we examined the prognostic values of these markers based on their mRNA expression using a cohort of uterine corpus endometrial carcinoma (UCEC, n = 307) taken from The Cancer Genome Atlas (TCGA) database. Our evidence indicated the presence of a positive correlation between the LI of MCM6 and the histological grade of endometrioid endometrial adenocarcinoma (grade I, 66.7%; grade II, 75.3%; grade III, 81.4%; p < 0.001) and an inverse correlation between the LI of MCM6 and the overall and progression-free survival (p = 0.02 for both). The LI of Ki-67 correlated with grade (p < 0.001), but not survival. The MCM6 and Ki-67 inter-observer intra-class correlation coefficients were excellent: 0.84 (95% confidence interval, 0.83–0.91) and 0.84 (0.77–0.90), respectively. For in silico analyses of the TCGA cohort, both univariate and multivariate Cox analyses (p = 0.003 and p = 0.03, respectively) revealed high MCM6 mRNA Z-scores associated with reduced overall survival. This association was absent for Ki-67. MCM6 is thus a highly reproducible marker of poor prognosis in endometrial cancer. Evaluation of MCM6 should thus be considered in daily practice for risk stratification.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66(1):7–30. https://doi.org/10.3322/caac.21332
Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JWW, Comber H, Forman D, Bray F (2013) Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer 49(6):1374–1403. https://doi.org/10.1016/j.ejca.2012.12.027
Fleming GF, Brunetto VL, Cella D, Look KY, Reid GC, Munkarah AR, Kline R, Burger RA, Goodman A, Burks RT (2004) Phase III trial of doxorubicin plus cisplatin with or without paclitaxel plus filgrastim in advanced endometrial carcinoma: a gynecologic oncology group study. J Clin Oncol Off J Am Soc Clin Oncol 22(11):2159–2166. https://doi.org/10.1200/JCO.2004.07.184
Thigpen JT, Brady MF, Homesley HD, Malfetano J, DuBeshter B, Burger RA, Liao S (2004) Phase III trial of doxorubicin with or without cisplatin in advanced endometrial carcinoma: a gynecologic oncology group study. J Clin Oncol Off J Am Soc Clin Oncol 22(19):3902–3908. https://doi.org/10.1200/JCO.2004.02.088
Petru E, Lück H-J, Stuart G, Gaffney D, Millan D, Vergote I (2009) Gynecologic cancer intergroup (GCIG) proposals for changes of the current FIGO staging system. Eur J Obstet Gynecol Reprod Biol 143(2):69–74. https://doi.org/10.1016/j.ejogrb.2008.12.015
Salvesen HB, Iversen OE, Akslen LA (1998) Identification of high-risk patients by assessment of nuclear Ki-67 expression in a prospective study of endometrial carcinomas. Clin Cancer Res 4:2779–2785
Gassel AM, Backe J, Krebs S, Schon S, Caffier H, Muller-Hermelink HK (1998) Endometrial carcinoma: immunohistochemically detected proliferation index is a prognosticator of long-term outcome. J Clin Pathol 51(1):25–29. https://doi.org/10.1136/jcp.51.1.25
Oreskovic S, Babic D, Kalafatic D, Barisic D, Beketic-Oreskovic L (2004) A significance of immunohistochemical determination of steroid receptors, cell proliferation factor Ki-67 and protein p53 in endometrial carcinoma. Gynecol Oncol 93(1):34–40. https://doi.org/10.1016/j.ygyno.2003.12.038
Chakravarty D, Gupta N, Goda JS, Srinivasan R, Patel FD, Dhaliwal L (2010) Steroid receptors, HER2/neu and Ki-67, in endometrioid type of endometrial carcinoma: correlation with conventional histomorphological features of prognosis. Acta Histochem 112(4):355–363. https://doi.org/10.1016/j.acthis.2009.03.001
Tye BK (1999) MCM proteins in DNA replication. Annu Rev Biochem 68(1):649–686. https://doi.org/10.1146/annurev.biochem.68.1.649
Ritzi M, Baack M, Musahl C, Romanowski P, Laskey RA, Knippers R (1998) Human minichromosome maintenance proteins and human origin recognition complex 2 protein on chromatin. J Biol Chem 273(38):24543–24549. https://doi.org/10.1074/jbc.273.38.24543
Romanowski P, Madine MA (1997) Mechanisms restricting DNA replication to once per cell cycle: the role of Cdc6p and ORC. Trends Cell Biol 7(1):9–10. https://doi.org/10.1016/S0962-8924(97)30077-4
Vigouroux C, Casse J-M, Battaglia-Hsu S-F, Brochin L, Luc A, Paris C, Lacomme S, Gueant JL, Vignaud JM, Gauchotte G (2015) Methyl(R217)HuR and MCM6 are inversely correlated and are prognostic markers in non small cell lung carcinoma. Lung Cancer 89(2):189–196. https://doi.org/10.1016/j.lungcan.2015.05.008
Gauchotte G, Vigouroux C, Rech F, Battaglia-Hsu SF, Soudant M, Pinelli C, Civit T, Taillandier L, Vignaud JM, Bressenot A (2012) Expression of minichromosome maintenance MCM6 protein in meningiomas is strongly correlated with histologic grade and clinical outcome. Am J Surg Pathol 36(2):283–291. https://doi.org/10.1097/PAS.0b013e318235ee03
Kato K, Toki T, Shimizu M, Shiozawa T, Fujii S, Nikaido T, Konishi I (2003) Expression of replication-licensing factors MCM2 and MCM3 in normal, hyperplastic, and carcinomatous endometrium: correlation with expression of Ki-67 and estrogen and progesterone receptors. Int J Gynecol Pathol Off J Int Soc Gynecol Pathol 22(4):334–340. https://doi.org/10.1097/01.pgp.0000092129.10100.5e
Li SS, Xue WC, Khoo US, Ngan HYS, Chan KYK, Tam IYS, Chiu PM, Ip PPC, Tam KF, Cheung ANY (2005) Replicative MCM7 protein as a proliferation marker in endometrial carcinoma: a tissue microarray and clinicopathological analysis. Histopathology 46(3):307–313. https://doi.org/10.1111/j.1365-2559.2005.02069.x
Sun H, Yan L, Tu R, Zhang Y, Ma L, Tang W, Li L, Chen W, Zhan C, Zang R (2016) Expression profiles of endometrial carcinoma by integrative analysis of TCGA data. Gynecol Obstet Investig 82(1):30–38. https://doi.org/10.1159/000445073
Agopiantz M, Forgez P, Casse J-M, Lacomme S, Charra-Brunaud C, Clerc-Urmès I, Morel O, Bonnet C, Guéant JL, Vignaud JM, Gompel A, Gauchotte G (2017) Expression of neurotensin receptor 1 in endometrial adenocarcinoma is correlated with histological grade and clinical outcome. Virchows Arch 471(4):521–530. https://doi.org/10.1007/s00428-017-2215-y
Colombo N, Creutzberg C, Amant F, Bosse T, González-Martín A, Ledermann J, Marth C, Nout R, Querleu D, Mirza MR, Sessa C, ESMO-ESGO-ESTRO Endometrial Consensus Conference Working Group (2016) ESMO-ESGO-ESTRO consensus conference on endometrial cancer: diagnosis, treatment and follow-up. Int J Gynecol Cancer 26(1):2–30. https://doi.org/10.1097/IGC.0000000000000609
Ouldamer L, Bendifallah S, Body G et al (2017) Call for surgical nodal staging in women with ESMO/ESGO/ESTRO high-intermediate risk endometrial cancer: a multicentre cohort analysis from the FRANCOGYN study group. Ann Surg Oncol 24(6):1660–1666. https://doi.org/10.1245/s10434-016-5731-0
Alkushi A, Lim P, Coldman A, Huntsman D, Miller D, Gilks CB (2004) Interpretation of p53 immunoreactivity in endometrial carcinoma: establishing a clinically relevant cut-off level. Int J Gynecol Pathol 23(2):129–137. https://doi.org/10.1097/00004347-200404000-00007
Alvarez T, Miller E, Duska L, Oliva E (2012) Molecular profile of grade 3 endometrioid endometrial carcinoma: is it a type I or type II endometrial carcinoma? Am J Surg Pathol 36(5):753–761. https://doi.org/10.1097/PAS.0b013e318247b7bb
Orbo A, Nilsen MN, Arnes MS et al (2003) Loss of expression of MLH1, MSH2, MSH6, and PTEN related to endometrial cancer in 68 patients with endometrial hyperplasia. Int J Gynecol Pathol Off J Int Soc Gynecol Pathol 22:141–148
Djordjevic B, Barkoh BA, Luthra R, Broaddus RR (2013) Relationship between PTEN, DNA mismatch repair, and tumor histotype in endometrial carcinoma: retained positive expression of PTEN preferentially identifies sporadic non-endometrioid carcinomas. Mod Pathol Off J U S Can Acad Pathol Inc 26(10):1401–1412. https://doi.org/10.1038/modpathol.2013.67
Network CGAR, Kandoth C, Schultz N et al (2013) Integrated genomic characterization of endometrial carcinoma. Nature 497(7447):67–73. https://doi.org/10.1038/nature12113
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G (2000) Gene ontology: tool for the unification of biology. The gene ontology consortium. Nat Genet 25(1):25–29. https://doi.org/10.1038/75556
Core Team R (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Therneau T (2015) _A package for survival analysis in S_. version 2.38
Pohlert T (2014) The Pariwaise multiple comparison of mean ranks package (PMCMR). R package
Wolak ME, Fairbairn DJ, Paulsen YR (2012) Guidelines for estimating repeatability. Methods Ecol Evol 3(1):129–137. https://doi.org/10.1111/j.2041-210X.2011.00125.x
Sorosky JI (2008) Endometrial cancer. Obstet Gynecol 111(2, Part 1):436–447. https://doi.org/10.1097/AOG.0b013e318162f690
Narducci F, Lambaudie E, Sonoda Y, Papageorgiou T, Taïeb S, Cabaret V, Castelain B, Leblanc E, Querleu D (2003) Endometrial cancer: what’s new? Gynecol Obstet Fertil 31(7-8):581–596. https://doi.org/10.1016/S1297-9589(03)00173-5
Soslow RA, Bissonnette JP, Wilton A, Ferguson SE, Alektiar KM, Duska LR, Oliva E (2007) Clinicopathologic analysis of 187 high-grade endometrial carcinomas of different histologic subtypes: similar outcomes belie distinctive biologic differences. Am J Surg Pathol 31(7):979–987. https://doi.org/10.1097/PAS.0b013e31802ee494
Mengel M, von Wasielewski R, Wiese B, Rüdiger T, Müller-Hermelink HK, Kreipe H (2002) Inter-laboratory and inter-observer reproducibility of immunohistochemical assessment of the Ki-67 labelling index in a large multi-centre trial. J Pathol 198(3):292–299. https://doi.org/10.1002/path.1218
Verheijen R, Kuijpers HJ, van Driel R et al (1989) Ki-67 detects a nuclear matrix-associated proliferation-related antigen. II. Localization in mitotic cells and association with chromosomes. J Cell Sci 92(Pt 4):531–540
Chatrath P, Scott IS, Morris LS, Davies RJ, Bird K, Vowler SL, Coleman N (2006) Immunohistochemical estimation of cell cycle phase in laryngeal neoplasia. Br J Cancer 95(3):314–321. https://doi.org/10.1038/sj.bjc.6603262
Scott IS, Morris LS, Bird K, Davies RJ, Vowler SL, Rushbrook SM, Marshall AE, Laskey RA, Miller R, Arends MJ, Coleman N (2003) A novel immunohistochemical method to estimate cell-cycle phase distribution in archival tissue: implications for the prediction of outcome in colorectal cancer. J Pathol 201(2):187–197. https://doi.org/10.1002/path.1444
Kingsbury SR, Loddo M, Fanshawe T, Obermann EC, Prevost AT, Stoeber K, Williams GH (2005) Repression of DNA replication licensing in quiescence is independent of geminin and may define the cell cycle state of progenitor cells. Exp Cell Res 309(1):56–67. https://doi.org/10.1016/j.yexcr.2005.05.027
Eward KL, Obermann EC, Shreeram S et al (2004) DNA replication licensing in somatic and germ cells. J Cell Sci 117(24):5875–5886. https://doi.org/10.1242/jcs.01503
Levine AJ, Momand J, Finlay CA (1991) The p53 tumour suppressor gene. Nature 351(6326):453–456. https://doi.org/10.1038/351453a0
Yemelyanova A, Vang R, Kshirsagar M, Lu D, Marks MA, Shih IM, Kurman RJ (2011) Immunohistochemical staining patterns of p53 can serve as a surrogate marker for TP53 mutations in ovarian carcinoma: an immunohistochemical and nucleotide sequencing analysis. Mod Pathol 24(9):1248–1253. https://doi.org/10.1038/modpathol.2011.85
Schultheis AM, Martelotto LG, De Filippo MR, Piscuglio S, Ng CKY, Hussein YR, Reis-Filho JS, Soslow RA, Weigelt B (2016) TP53 mutational spectrum in endometrioid and serous endometrial cancers. Int J Gynecol Pathol Off J Int Soc Gynecol Pathol 35(4):289–300. https://doi.org/10.1097/PGP.0000000000000243
Kohler MF, Carney P, Dodge R, Soper JT, Clarke-Pearson DL, Marks JR, Berchuck A (1996) p53 overexpression in advanced-stage endometrial adenocarcinoma. Am J Obstet Gynecol 175(5):1246–1252. https://doi.org/10.1016/S0002-9378(96)70036-4
Kohlberger P, Gitsch G, Loesch A, Tempfer C, Kaider A, Reinthaller A, Kainz C, Breitenecker G (1996) p53 protein overexpression in early stage endometrial cancer. Gynecol Oncol 62(2):213–217. https://doi.org/10.1006/gyno.1996.0218
Ozalp S, Yalcin OT, Tanir HM, Kabukcuoglu S, Erol G (2003) p53 overexpression as a prognostic indicator in endometrial carcinoma. Eur J Gynaecol Oncol 24(3-4):275–278
Kudela M, Pilka R, Lubusky M, Hejtmanek P, Dzubak P, Brychtova S (2012) Prognostic importance of selected molecular immunohistochemical markers and DNA ploidy in endometrial cancer. Eur J Gynaecol Oncol 33(2):159–163
Miyaki M, Konishi M, Tanaka K, Kikuchi-Yanoshita R, Muraoka M, Yasuno M, Igari T, Koike M, Chiba M, Mori T (1997) Germline mutation of MSH6 as the cause of hereditary nonpolyposis colorectal cancer. Nat Genet 17(3):271–272. https://doi.org/10.1038/ng1197-271
Nicolaides NC, Papadopoulos N, Liu B, Weit YF, Carter KC, Ruben SM, Rosen CA, Haseltine WA, Fleischmann RD, Fraser CM, Adams MD, Venter JC, Dunlop MG, Hamilton SR, Petersen GM, de la Chapelle A, Vogelstein B, Kinzler KW (1994) Mutations of two PMS homologues in hereditary nonpolyposis colon cancer. Nature 371(6492):75–80. https://doi.org/10.1038/371075a0
An HJ, Kim KI, Kim JY, Shim JY, Kang H, Kim TH, Kim JK, Jeong JK, Lee SY, Kim SJ (2007) Microsatellite instability in endometrioid type endometrial adenocarcinoma is associated with poor prognostic indicators. Am J Surg Pathol 31(6):846–853. https://doi.org/10.1097/01.pas.0000213423.30880.ac
Nelson GS, Pink A, Lee S, Han G, Morris D, Ogilvie T, Duggan MA, Köbel M (2013) MMR deficiency is common in high-grade endometrioid carcinomas and is associated with an unfavorable outcome. Gynecol Oncol 131(2):309–314. https://doi.org/10.1016/j.ygyno.2013.08.003
Acknowledgements
The authors thank all members of Pathology Departments of CHRU (Nancy) and Institut de Cancérologie de Lorraine (Vandoeuvre-lès-Nancy) for their technical support and Ms. Magali LEFEBVRE and Delphine CLABAUT for their advices.
Results are in part generated by the data extracted from the database of TCGA Research Network: http://cancergenome.nih.gov/.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiments reported here were carried out according to the Declaration of Helsinki principles and in agreement with the French laws on biomedical research (institutional review board n°DC2008-459; CNIL declaration n°1209171).
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Supplementary Figure S1
MCM6 and Ki-67 labeling index and p53 status. (A) Significant correlation between overexpression of MCM6 staining and p53 mutated status (p = 0.003). (B) No association between Ki-67 labeling expression and p53 status (p = 0.23) (boxplots; Wilcoxon tests). (JPEG 42 kb)
Supplementary Figure S2
MCM6 and Ki-67 labeling index and microsatellite instability (MSI). (A) Significant correlation between overexpression of MCM6 staining and microsatellite instability (p = 0.03). (B) No association between Ki-67 labeling expression and tumors showing loss of MSH6 and/or PMS2 (p = 0.41) (boxplots; Wilcoxon tests). (JPEG 42 kb)
Supplementary Figure S3
Correlation curve between marker of proliferation Ki-67 (Ki-67), and minichromosome maintenance complex component 6 (MCM6) expression in endometrioid endometrial adenocarcinoma (rho = 0.55, p < 0.001) (Spearman’s correlation test). (GIF 154 kb)
Supplementary Figure S4
Survival analyses in the uterine corpus endometrioid carcinoma cohort of TCGA. Kaplan-Meier curves with log-rank tests. (A) Correlation between minichromosome maintenance complex component 6 (MCM6) mRNA Z-score and overall survival (p = 0.04) (threshold: 75th percentile [0.45]). (B) Lack of significant correlation between marker of proliferation Ki-67 (Ki-67), mRNA Z-score and overall survival (p = 0.2)(threshold: 75th percentile [0.50]). (JPEG 65 kb)
Supplementary Figure S5
Expression of minichromosome maintenance complex component 6 (MCM6) sorted by molecular subgroups (A) (p < 0.001). Expression of marker of proliferation Ki-67 (Ki-67), sorted by molecular subgroups (B) (p < 0.001) (boxplots; Kruskal-Wallis tests). (JPEG 52 kb)
Rights and permissions
About this article
Cite this article
Hotton, J., Agopiantz, M., Leroux, A. et al. Minichromosome maintenance complex component 6 (MCM6) expression correlates with histological grade and survival in endometrioid endometrial adenocarcinoma. Virchows Arch 472, 623–633 (2018). https://doi.org/10.1007/s00428-017-2278-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-017-2278-9