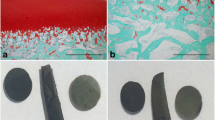
Abstract
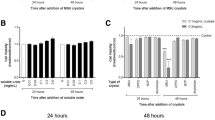
Multinucleated cells termed chondroclasts have been observed on the deep surface of resorbed hyaline cartilage but the relationship of these cells to macrophages and osteoclasts and their role in rheumatoid arthritis (RA) and other arthritic conditions is uncertain. Multinucleated cells in RA and other arthritic conditions showing evidence of cartilage resorption were characterised immunohistochemically for expression of macrophage/osteoclast markers. Mature human osteoclasts formed from circulating monocytes and tissue macrophages were cultured for up to 4 days on slices of human cartilage and glycosaminoglycan (GAG) release was measured. Multinucleated cells resorbing unmineralised cartilage were seen in osteoarthritis, RA, septic arthritis, avascular necrosis and in four cases of giant cell tumour of bone that had extended through the subchondral bone plate. Chondroclasts expressed an osteoclast-like phenotype (TRAP+, cathepsin K+, MMP9+, CD14−, HLA-DR−, CD45+, CD51+ and CD68+). Both macrophages and osteoclasts cultured on cartilage released GAG. These findings indicate that chondroclasts have an osteoclast-like phenotype and that mature human osteoclasts are capable of cartilage matrix resorption. Resorption of unmineralised subchondral cartilage by chondroclasts and macrophages can be a feature of joint destruction in inflammatory and non-inflammatory arthropathies as well as inflammatory and neoplastic subchondral bone lesions.



Similar content being viewed by others
Abbreviations
- RANKL:
-
Receptor activator of nuclear factor kappa B ligand
- M-CSF:
-
Macrophage-colony stimulating factor
- MMP:
-
Matrix metalloproteinase
- TRAP:
-
Tartrate-resistant acid phosphatase
- OA:
-
Osteoarthritis
- GAG:
-
Glycosaminoglycan
References
Bromley M, Woolley DE (1984) Histopathology of the rheumatoid lesion. Identification of cell types at sites of cartilage erosion. Arthritis Rheum 27:857–863
Bromley M, Woolley DE (1984) Chondroclasts and osteoclasts at subchondral sites of erosion in the rheumatoid joint. Arthritis Rheum 27:968–975
Bromley M, Bertfield H, Evanson JM, Woolley DE (1985) Bidirectional erosion of cartilage in the rheumatoid knee joint. Ann Rheum Dis 44:676–681
Novack DV, Teitelbaum SL (2008) The osteoclast: friend or foe? Annu Rev Pathol 3:457–484
Fujikawa Y, Quinn JM, Sabokbar A, McGee JO, Athanasou NA (1996) The human osteoclast precursor circulates in the monocyte fraction. Endocrinology 137:4058–4060
Quinn JM, Elliott J, Gillespie MT, Martin TJ (1998) A combination of osteoclast differentiation factor and macrophage colony stimulating factor is sufficient for both human and mouse osteoclast formation in vitro. Endocrinology 139:4424–4427
Chambers TJ, Thomson BM, Fuller K (1984) Effect of substrate composition on bone resorption by rabbit osteoclasts. J Cell Sci 70:61–71
Fuller K, Chambers TJ (1995) Localisation of mRNA for collagenase in osteocytic, bone surface and chondrocytic cells but not osteoclasts. J Cell Sci 108:2221–2230
Andersen TL, del Carmen OM, Kirkegaard T, Lenhard T, Foged NT, Delaissé JM (2004) A scrutiny of matrix metalloproteinases in osteoclasts: evidence for heterogeneity and for the presence of MMPs synthesized by other cells. Bone 35:1107–1119
Murphy G, Lee MH (2005) What are the roles of metalloproteinases in cartilage and bone damage? Ann Rheum Dis 64:44–47
Cawston TE, Young DA (2010) Proteinases involved in matrix turnover during cartilage and bone breakdown. Cell Tissue Res 339:221–235
Knowles H, Athanasou NA (2008) Acute hypoxia and osteoclast activity: a balance between enhanced resorption and increased apoptosis. J Pathol 218:256–264
Chambers TJ, Fuller K, McSheehy PM, Pringle JA (1985) The effects of calcium regulating hormones on bone resorption by isolated human osteoclastoma cells. J Pathol 145:297–305
Lau YS, Sabokbar A, Gibbons CLMH, Athanasou NA (2005) Phenotypic and molecular studies on giant cell tumours of bone and soft tissues. Hum Pathol 36:945–954
Taylor R, Kashima TG, Knowles H, Gibbons CL, Whitwell D, Athanasou NA (2011) Osteoclast formation and function in pigmented villonodular synovitis. J Pathol 225:151–156
Riley GP, Harrall RL, Constant CR, Chard MD, Cawston TE, Hazleman BL (1994) Glycosaminoglycans of human rotator cuff tendons: changes with age and in chronic rotator cuff tendinitis. Ann Rheum Dis 53:367–376
Maggiani F, Forsyth R, Hogendoorn PCW, Krenacs T, Athanasou NA (2011) Immunophenotype of osteoclasts and macrophage polykaryons. J Clin Pathol 64:701–705
Lewinson D, Sibermann (1992) Chondroclasts and endothelial cells collaborate in the process of cartilage resorption. Anat Rec 233:504–514
Shibata S, Suzuki S, Yamashita Y (1997) An ultrastructural study of cartilage resorption at the site of initial endochondral bone formation in the fetal mouse mandibular condyle. J Anat 191:65–76
Szuwart T, Kierdorf H, Kierdorf U, Clemen G (2002) Histochemical and ultrastructural studies of cartilage resorption and acid phosphatise activity during antler growth in fallow deer (Dama dama). Anat Rec 268:66–72
Nordahl J, Andersson G, Reinholt FP (1998) Chondroclasts and osteoclasts in bones of young rats: comparison of ultrastructural and functional features. Calcif Tissue Int 63:401–408
Arana-Chavez VE, Bradaschia-Correa V (2009) Clastic cells: mineralized tissue resorption in health and disease. Int J Biochem Cell Biol 41:446–450
Suzumoto R, Takami M, Sasaki T (2005) Differentiation and function of osteoclasts cultured on bone and cartilage. J Electron Microsc (Tokyo) 54:529–540
Yovich S, Seydel U, Papadimitriou JM, Nicholson GC, Wood DJ, Zheng MH (1998) Evidence that failure of osteoid bone matrix resorption is caused by perturbation of osteoclast polarization. Histochem J 30:267–273
Shimizu H, Sakamoto S, Sakamoto M (1989) Matrix collagen of devitalized bone is resistant to osteoclastic bone resorption. Connect Tissue Res 20:169–175
Chambers TJ, Fuller K (1985) Bone cells predispose bone surfaces to resorption by exposure of mineral to osteoclastic contact. J Cell Sci 76:155–165
Fuller K, Gallagher AC, Chambers TJ (1991) Osteoclast resorption-stimulating activity is associated with the osteoblast cell surface and/or the extracellular matrix. Biochem Biophys Res Commun 27:67–73
Holiday LS, Welgus HG, Flixzar CJ, Veith GM, Jeffrey JJ, Gluck SL (1997) Initiation of osteoclast bone resorption by interstitial collagenase. J Biol Chem 29:22053–22058
Blair HC, Kahn AJ, Crouch EC, Jeffrey JJ, Teitelbaum SL (1986) Isolated osteoclasts resorb the organic and inorganic components of bone. J Cell Biol 102:1164–1172
Dejica VM, Mort JS, Laverty S, Percival MD, Antoniou J, Zukor DJ, Poole AR (2008) Cleavage of type II collagen by cathepsin K in human osteoarthritic cartilage. Am J Pathol 173:161–169
Cowan RW, Mak I, Colterjohn N, Singh G, Chent M (2009) Collagenase expression and activity in the stromal cell from giant cell tumour of bone. Bone 44:865–871
Quinn JMW, McGee JO'D, Athanasou NA (1998) Human tumour-associated macrophages differentiate into osteoclastic bone-resorbing cells. J Pathol 184:31–36
Fujikawa Y, Sabokbar A, Neale S, Athanasou NA (1996) Human osteoclast formation and bone resorption by monocytes and synovial macrophages in rheumatoid arthritis. Ann Rheum Dis 55:1–7
Komuro H, Olee T, Kuhn K, Quach J, Brinson DC, Shikhman A, Valbracht J, Creighton-Achermann L, Lotz M (2001) The osteoprotegerin/receptor activator of nuclear factor kappaB/receptor activator of nuclear factor kappaB ligand system in cartilage. Arthritis Rheum 44:2768–2776
Ota N, Takaishi H, Kosaki N, Takito J, Yoda M, Tohmonda T, Kimura T, Okada Y, Yasuda H, Kawaguchi H, Matsumoto M, Chiba K, Ikegami H, Toyama Y (2009) Accelerated cartilage resorption by chondroclasts during bone fracture healing in osteoprotegerin-deficient mice. Endocrinology 150:4823–4834
Strassle BW, Mark L, Leventhal L, Piesla MJ, Jian Li X, Kennedy JD, Glasson SS, Whiteside GT (2010) Inhibition of osteoclasts prevents cartilage loss and pain in a rat model of degenerative joint disease. Osteoarthr Cartil 18:1319–1328
Svelander L, Erlandsson-Harris H, Astner L, Grabowska U, Klareskog L, Lindstrom E, Hewitt E (2009) Inhibition of cathepsin K reduces bone erosion, cartilage degradation and inflammation evoked by collagen-induced arthritis in mice. Eur J Pharmacol 613:155–162
Acknowledgments
We wish to thank Mrs C Lowe for typing the manuscript and R. Taylor for providing the PVNS cells. This study was supported by the Rosetrees Charitable Trust and EuroBoNet a Network of Excellence, funded by the European Union.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Knowles, H.J., Moskovsky, L., Thompson, M.S. et al. Chondroclasts are mature osteoclasts which are capable of cartilage matrix resorption. Virchows Arch 461, 205–210 (2012). https://doi.org/10.1007/s00428-012-1274-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-012-1274-3