Abstract
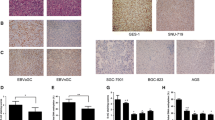
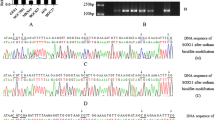
SOX9 is a member of the SOX [Sry-related high-mobility group (HMG) box] family and is required for the development and differentiation of multiple cell lineages. To clarify the significance of SOX9 in gastric carcinoma (GC), immunohistochemical expression of SOX9 and the CpG island methylation status of SOX9 were evaluated and compared with clinicopathological factors including overall survival. SOX9 expression was immunohistochemically evaluated in 382 GC tumors and the methylation status was examined in 121 GC tumors. SOX9 expression and its methylation status in six GC cell lines, their Epstein–Barr virus (EBV)-infected cell lines, and two EBV-associated GC cell lines was also examined. The SOX9 expression increased from non-neoplastic mucosa to early cancer. High expression of SOX9 was seen in 212 cases (56%). SOX9 expression was inversely related to advanced tumor stage, vessel infiltration, nodal metastasis, and EBV infection. Fifty-eight (48%) of 121 GC tumors had a methylated promoter in GC and the methylated status was related to low expression. The expression and methylation status were not related to prognosis. Three of six cell lines had increased methylation through EBV infection and decreased SOX9 expression. Upregulation of SOX9 is related to GC development. Downregulation by promoter methylation is related to GC progression and EBV infection. SOX9 is closely related to GC carcinogenesis and EBV-associated GC carcinogenesis.




Similar content being viewed by others
References
Uozaki H, Fukayama M (2008) Epstein–Barr virus and gastric carcinoma—viral carcinogenesis through epigenetic mechanisms. Int J Clin Exp Pathol 1:198–216
Uozaki H, Barua RR, Sun M et al (2011) Transcriptional factor typing with SOX2, HNF4aP1, and CDX2 closely relates to tumor invasion and Epstein–Barr virus status in gastric cancer. Int J Clin Exp Pathol 4:230–240
Park DY, Srivastava A, Kim GH et al (2010) CDX2 expression in the intestinal-type gastric epithelial neoplasia: frequency and significance. Mod Pathol 23:54–61
Haveri H, Westerholm-Ormio M, Lindfors K et al (2008) Transcription factors GATA-4 and GATA-6 in normal and neoplastic human gastrointestinal mucosa. BMC Gastroenterol 8:9
Lefebvre V, Dumitriu B, Penzo-Mendez A et al (2007) Control of cell fate and differentiation by Sry-related high-mobility-group box (Sox) transcription factors. Int J Biochem Cell Biol 39:2195–2214
Kent J, Wheatley SC, Andrews JE et al (1996) A male-specific role for SOX9 in vertebrate sex determination. Development 122:2813–2822
Lefebvre V, Huang W, Harley VR et al (1997) SOX9 is a potent activator of the chondrocyte-specific enhancer of the pro alpha1(II) collagen gene. Mol Cell Biol 17:2336–2346
Stolt CC, Lommes P, Sock E et al (2003) The Sox9 transcription factor determines glial fate choice in the developing spinal cord. Genes Dev 17:1677–1689
Perez-Alcala S, Nieto MA, Barbas JA (2004) LSox5 regulates RhoB expression in the neural tube and promotes generation of the neural crest. Development 131:4455–4465
Foster JW, Dominguez-Steglich MA, Guioli S et al (1994) Campomelic dysplasia and autosomal sex reversal caused by mutations in an SRY-related gene. Nature 372:525–530
De Santa BP, Bonneaud N, Boizet B et al (1998) Direct interaction of SRY-related protein SOX9 and steroidogenic factor 1 regulates transcription of the human anti-Mullerian hormone gene. Mol Cell Biol 18:6653–6665
Blache P, van de Wetering M, Duluc I et al (2004) SOX9 is an intestine crypt transcription factor, is regulated by the Wnt pathway, and represses the CDX2 and MUC2 genes. J Cell Biol 166:37–47
Fukayama M, Hino R, Uozaki H (2008) Epstein–Barr virus and gastric carcinoma: virus–host interactions leading to carcinoma. Cancer Sci 99:1726–1733
Lu B, Fang Y, Xu J et al (2008) Analysis of SOX9 expression in colorectal cancer. Am J Clin Pathol 130:897–904
Thomsen MK, Ambroisine L, Wynn S et al (2010) SOX9 elevation in the prostate promotes proliferation and cooperates with PTEN loss to drive tumor formation. Cancer Res 70:979–987
Japanese Gastric Cancer Association (1998) Japanese classification of gastric carcinoma—2nd English edition. Gastric Cancer 1:10–24
Lauren P (1965) The two histological main types of gastric carcinoma. Diffuse and so-called intestinal type carcinoma. An attempt at histoclinical classification. Acta Pathol Microbiol Scand 64:31–49
Sinicrope FA, Ruan SB, Cleary KR et al (1995) bcl-2 and p53 oncoprotein expression during colorectal tumorigenesis. Cancer Res 55:237–241
Iwasaki Y, Chong JM, Hayashi Y et al (1998) Establishment and characterization of a human Epstein–Barr virus-associated gastric carcinoma in SCID mice. J Virol 72:8321–8326
Imai S, Nishikawa J, Takada K (1998) Cell-to-cell contact as an efficient mode of Epstein–Barr virus infection of diverse human epithelial cells. J Virol 72:4371–4378
Shimizu N, Yoshiyama H, Takada K (1996) Clonal propagation of Epstein–Barr virus (EBV) recombinants in EBV-negative Akata cells. J Virol 70:7260–7263
Aleman A, Adrien L, Lopez-Serra L et al (2008) Identification of DNA hypermethylation of SOX9 in association with bladder cancer progression using CpG microarrays. Br J Cancer 98:466–473
Chang MS, Uozaki H, Chong JM et al (2006) CpG island methylation status in gastric carcinoma with and without infection of Epstein–Barr virus. Clin Cancer Res 12:2995–3002
Stolt CC, Wegner M (2010) SoxE function in vertebrate nervous system development. Int J Biochem Cell Biol 42:437–440
Akiyama H (2008) Control of chondrogenesis by the transcription factor Sox9. Mod Rheumatol 18:213–219
Thomsen MK, Francis JC, Swain A (2008) The role of Sox9 in prostate development. Differentiation 76:728–735
Barrionuevo F, Scherer G (2010) SOX E genes: SOX9 and SOX8 in mammalian testis development. Int J Biochem Cell Biol 42:433–436
Moniot B, Biau S, Faure S et al (2004) SOX9 specifies the pyloric sphincter epithelium through mesenchymal-epithelial signals. Development 131:3795–3804
Mori-Akiyama Y, van den Born M, van Es JH et al (2007) SOX9 is required for the differentiation of paneth cells in the intestinal epithelium. Gastroenterology 133:539–546
Bastide P, Darido C, Pannequin J et al (2007) Sox9 regulates cell proliferation and is required for Paneth cell differentiation in the intestinal epithelium. J Cell Biol 178:635–648
Yasui W, Oue N, Sentani K et al (2009) Transcriptome dissection of gastric cancer: identification of novel diagnostic and therapeutic targets from pathology specimens. Pathol Int 59:121–136
Chen Y, Zhang Y-Z, Zhou Z-G et al (2006) Identification of differently expressed genes in human colorectal adenocarcinoma. World J Gastroenterol 12:1025–1032
Jiang SS, Fang W-T, Hou Y-H et al (2010) Upregulation of SOX9 in lung adenocarcinoma and its involvement in the regulation of cell growth and tumorigenicity. Clin Cancer Res 16:4363–4373
Wang H, Leav I, Ibaragi S et al (2008) SOX9 is expressed in human fetal prostate epithelium and enhances prostate cancer invasion. Cancer Res 68:1625–1630
Passeron T, Valencia JC, Namiki T et al (2009) Upregulation of SOX9 inhibits the growth of human and mouse melanomas and restores their sensitivity to retinoic acid. J Clin Invest 119:954–963
Chakravarty G, Moroz K, Makridakis NM et al (2011) Prognostic significance of cytoplasmic SOX9 in invasive ductal carcinoma and metastatic breast cancer. Exp Biol Med (Maywood) 236:145–155
Clevers H (2006) Wnt/beta-catenin signaling in development and disease. Cell 127:469–480
Vidal VP, Ortonne N, Schedl A (2008) SOX9 expression is a general marker of basal cell carcinoma and adnexal-related neoplasms. J Cutan Pathol 35:373–379
Ramocki NM, Wilkins HR, Magness ST et al (2008) Insulin receptor substrate-1 deficiency promotes apoptosis in the putative intestinal crypt stem cell region, limits Apcmin/+ tumors, and regulates Sox9. Endocrinology 149:261–267
Choi IS, Wu TT (2005) Epigenetic alterations in gastric carcinogenesis. Cell Res 15:247–254
Acknowledgment
This study was supported by grants-in-aid for scientific research on priority areas (20249022, 20590337) and the Global COE Research Program “Comprehensive center of education and research for chemical biology of the diseases” from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Conflicts of interest
We declare that we have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, M., Uozaki, H., Hino, R. et al. SOX9 expression and its methylation status in gastric cancer. Virchows Arch 460, 271–279 (2012). https://doi.org/10.1007/s00428-012-1201-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-012-1201-7