Abstract
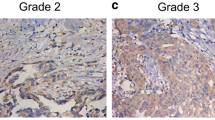
Recent studies have indicated that Aurora B expression is related to cell proliferation and prognosis in many cancers, but its association with epithelial ovarian carcinoma is not fully understood. Therefore, we examined the Aurora B kinase expression in epithelial ovarian cancer patients. Using immunohistochemistry, the expression levels of Aurora B and phosphohistone H3 (Ser10) (mitosis-specific marker) were measured in 156 patients with epithelial ovarian cancer. The expression levels of Aurora B at the protein and messenger RNA levels were examined using Western blotting and reverse transcriptase polymerase chain reaction. In total, 53 tumorous ovarian samples (34.0%) showed Aurora B overexpression, which was significantly higher than that found in the 15 normal ovarian tissue samples (0%, p = 0.006). The overexpression of Aurora B was also significantly higher in cases showing phosphohistone H3 (Ser10) overexpression (44.3% vs. 27.4%, p = 0.03). In addition, the expression of Aurora B in poorly and moderately differentiated carcinomas of the ovary was significantly higher than in well-differentiated carcinomas (53.6% vs. 28.2% vs.10.0%, respectively, p = 0.02). The overexpression of Aurora B was significantly higher in cases with lymph node metastasis (p = 0.01) and a positive ascites cytology (p = 0.008). Overall, the Aurora B overexpression group demonstrated a significantly shorter progression-free survival (p = 0.001) and overall survival (p = 0.023) than the Aurora B low expression group using univariate analysis (log-rank statistic). Aurora B is an effective predictor of aggressive epithelial ovarian carcinoma in terms of differentiation, metastasis, and prognosis.



Similar content being viewed by others
References
NCI Surveillance, E.a.E.R.S.P.a.t.N.C.f.H.S. National Cancer Institute—a snapshot of ovarian cancer: incidence and mortality rate trends 2006 Sep. http://planning.cancer.gov/disease/ovarian-snapshot.pdf
Hoskins WJ, Perez CA, Young RC (2000) Principles and practice of gynecologic oncology. Lippincott Williams and Wilkins, Philadelphia, pp 1005–1007
Adams RR, Carmena M, Earnshaw WC (2001) Chromosomal passengers and the (Aurora) ABCs of mitosis. Trends Cell Biol 11:49–54
Tatsuka M, Katayama H, Ota T et al (1998) Multinuclearity and increased ploidy caused by overexpression of the aurora- and Ipl1-like midbody-associated protein mitotic kinase in human cancer cells. Cancer Res 58:4811–4816
Kaitna S, Pasierbek P, Jantsch M et al (2002) The aurora B kinase AIR-2 regulates kinetochores during mitosis and is required for separation of homologous chromosomes during meiosis. Curr Biol 12:798–812
Ota T, Suto S, Katayama H et al (2002) Increased mitotic phosphorylation of histone H3 attributable to AIM-1/Aurora-B overexpression contributes to chromosome number instability. Cancer Res 62:5168–5177
Murata-Hori M, Wang YL (2002) Both midzone and astral microtubules are involved in the delivery of cytokinesis signals: insights from the mobility of aurora B. J Cell Biol 159:45–53
Sorrentino R, Libertini S, Pallante PL et al (2005) Aurora B overexpression associates with the thyroid carcinoma undifferentiated phenotype and is required for thyroid carcinoma cell proliferation. J Clin Endocrinol Metab 90:928–935
Katayama H, Ota T, Jisaki F et al (1999) Mitotic kinase expression and colorectal cancer progression. J Natl Cancer Inst 91:1160–1162
Sistayanarain A, Tsuneyama K, Zheng H et al (2006) Expression of Aurora-B kinase and phosphorylated histone H3 in hepatocellular carcinoma. Anticancer Res 26:3585–3593
Ulisse S, Delcros JG, Baldini E et al (2006) Expression of Aurora kinases in human thyroid carcinoma cell lines and tissues. Int J Cancer 119:275–282
Hayama S, Daigo Y, Yamabuki T et al (2007) Phosphorylation and activation of cell division cycle associated 8 by aurora kinase B plays a significant role in human lung carcinogenesis. Cancer Res 67:4113–4122
Remmele W, Schicketanz KH (1993) Immunohistochemical determination of estrogen and progesterone receptor content in human breast cancer. Computer-assisted image analysis (QIC score) vs subjective grading (IRS). Pathol Res Pract 8:227–245
Chieffi P, Cozzolino L, Kisslinger A et al (2006) Aurora B expression directly correlates with prostate cancer malignancy and influence prostate cell proliferation. Prostate 66:326–333
Kurai M, Shiozawa T, Shih HC et al (2005) Expression of Aurora kinases A and B in normal, hyperplastic and malignant human endometrium: Aurora B as a predictor for poor prognosis in endometrial carcinoma. Hum Pathol 36:1281–1288
Kulkarni AA, Loddo M, Leo E et al (2007) DNA replication licensing factors and aurora kinases are linked to aneuploidy and clinical outcome in epithelial ovarian carcinoma. Clin Cancer Res 13:6153–6161
Hendzel MJ, Wei Y, Mancini MA et al (1997) Mitosis-specific phosphorylation of histone H3 initiates primarily within pericentromeric heterochromatin during G2 and spreads in an ordered fashion coincident with mitotic chromosome condensation. Chromosoma 106:348–360
Kim YJ, Ketter R, Steudel WI et al (2007) Prognostic significance of the mitotic index using the mitosis marker anti-phosphohistone H3 in meningiomas. Am J Clin Pathol 128:118–125
Rosai J (2004) Ackerman’s surgical pathology, 9th edn. Mosby, St. Louis
Köbel M, Kalloger SE, Boyd N et al (2008) Ovarian carcinoma subtypes are different diseases: implications for biomarker studies. PLoS Med 5:e232
Soslow RA (2008) Histologic subtypes of ovarian carcinoma: an overview. Int J Gynecol Pathol 27:161–174
Lee EC, Frolov A, Li R et al (2006) Targeting Aurora kinases for the treatment of prostate cancer. Cancer Res 66:4996–5002
Remmele W, Schicketanz KH (1993) Immunohistochemical determination of estrogen and progesterone receptor content in human breast cancer. Computer-assisted image analysis (QIC score) vs. subjective grading (IRS). Pathol Res Pract 189:862–866
Twu NF, Yuan CC, Yen MS et al (2009) Expression of aurora kinase A and B in normal and malignant cervical tissue: high Aurora A kinase expression in squamous cervical cancer. Eur J Obstet Gynecol Reprod Biol 142:57–63
Chen CM, Behringer RR (2004) Ovca1 regulates cell proliferation, embryonic development, and tumorigenesis. Genes Dev 18:320–332
Kaplan E, Meyer P (1958) Non parametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Altman DG, Lausen B, Sauerbrei W et al (1994) Dangers of using “Optimal” cut points in the evaluation of prognostic factors. J Natl Cancer Inst 86:829–835
Cox DR (1970) Analysis of binary data. Methuen, London
Andrews PD, Knatko E, Moore WJ et al (2003) Mitotic mechanics: the Auroras come into view. Curr Opin Cell Biol 15:672–683
Ruchaud S, Carmena M, Earnshaw WC (2007) The chromosomal passenger complex: one for all and all for one. Cell 131:230–231
Hsu JY, Sun ZW, Li X et al (2000) Mitotic phosphorylation of histone H3 is governed by Ipl1/aurora kinase and Glc7/PP1 phosphatase in budding yeast and nematodes. Cell 102:279–291
Hendzel MJ, Wei Y, Mancini MA et al (1997) Mitosis-specific phosphorylation of histone H3 initiates primarily within pericentromeric heterochromatin during G2 and spreads in an ordered fashion coincident with mitotic chromosome condensation. Chromosoma 106:348–360
Wei Y, Yu L, Bowen J et al (1999) Phosphorylation of histone H3 is required for proper chromosome condensation and segregation. Cell 97:99–109
Wheatley SP, Barrett RM, Andrews PD et al (2007) Phosphorylation by aurora-B negatively regulates survivin function during mitosis. Cell Cycle 6:1220–1230
Jacobberger JW, Frisa PS, Sramkoski RM et al (2008) A new biomarker for mitotic cells. Cytometry A 73:5–15
Kim YJ, Ketter R, Steudel WI et al (2007) Prognostic significance of the mitotic index using the mitosis marker anti-phosphohistone H3 in meningiomas. Am J Clin Pathol 128:118–125
Qi G, Ogawa I, Kudo Y et al (2007) Aurora-B expression and its correlation with cell proliferation and metastasis in oral cancer. Virchows Arch 450:297–302
Acknowledgments
We thank Ching-Fen Yang for her evaluation of the immunohistochemical score. We appreciate Professor Ralphy Kirby’s help in improving the style of written English. We thank Dr. Muh-Hwa Yang for his involvement in revising the manuscript. We are grateful for the assistance of Miss Tsui-Ying Huang in completing the RT-PCR and Western blots. This research was supported, in part, by grant 96-VGH-B1-010 from Veterans General Hospital and by a grant from the Ministry of Education “Aim for the Top University” Plan. This study was also supported by research grants from Yen-Tjing-Ling Medical Foundation (CI-97-6).
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
Yi-Jen Chen carried out immunohistochemical staining, performed the statistical analysis, and drafted the manuscript. Chun-Ming Chen carried out the RT-PCR, Western blot and drafted the manuscript. Chiung-Ru Lai assessed the pathological specimens and evaluated the immunohistochemical score. Nae-Fang Twu, Ming-Shyen Yen, Hua-Hsi Wu, and Peng-Hui Wang participated in the design of the study and provided patients’ data. Chiou-Chung Yuan conceived of the study and participated in its design and coordination. All authors read and approved the final manuscript.
C.-M. Chen and C.-C. Yuan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, YJ., Chen, CM., Twu, NF. et al. Overexpression of Aurora B is associated with poor prognosis in epithelial ovarian cancer patients. Virchows Arch 455, 431–440 (2009). https://doi.org/10.1007/s00428-009-0838-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-009-0838-3