Abstract
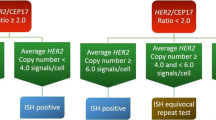
Overexpression of Her-2/neu-oncoprotein is used as marker for Herceptin® therapy. To investigate the sensitivity and specificity of automatic immunohistochemistry (Benchmark, Ventana), we compared the results to the manual testing (Dako) in 130 breast carcinomas and validated the results by fluorescence in situ hybridization (FISH). Manual and automatic immunohistochemistry of Her-2/neu-oncoprotein using two different antibodies (HercepTest™, Her-2/neuTest 4B5) was analyzed. FISH was performed in all cases with uncertain or strong overexpression in either immunohistochemical stainings or with different immunohistochemical results. Same immunohistochemical results were seen in 73.8%. Two cases with overexpression, detected with Her-2/neuTest 4B5 and confirmed by FISH, showed no overexpression using HercepTest™. From 21 cases with 2+ by Her-2/neuTest 4B5, 15 cases had no gene amplification (two of them with 3+ HercepTest™); three cases showed a gene amplification (one of them with failing overexpression by HercepTest™); two other cases were polysomic; one could not be analyzed. Ventana immunohistochemistry seems to be of same reliability like Dako with a little better concordance to FISH in our study.



Similar content being viewed by others
References
Berchuck A, Kamel A, Whitaker R et al (1990) Overexpression of HER-2/neu is associated with poor survival in advanced epithelial ovarian cancer. Cancer Res 50:4087–4091
Press MF, Pike MC, Hung G et al (1994) Amplification and overexpression of HER 2/neu in carcinomas of the salivary gland: correlation with poor prognosis. Cancer Res 54:5675–5682
Raspollini MR, Amunni G, Villanucci A et al (2006) HER-2/neu and bcl-2 in ovarian carcinoma: clinicopathologic, immunohistochemical, and molecular study in patients with shorter and longer survival. Appl Immunohistochem Mol Morphol 14:181–186
Formenti SC, Spicer D, Skinner K et al (2002) Low HER2/neu gene expression is associated with pathological response to concurrent paclitaxel and radiation therapy in locally advanced breast cancer. Int J Radiat Oncol Biol Phys 52:397–405
Gasparini G, Longo R, Torino F et al (2005) Therapy of breast cancer with molecular targeting agents. Ann Oncol 16(Suppl 4):iv28–iv36
Gusterson BA, Gelber RD, Goldhirsch A et al (1992) Prognostic importance of c erbB-2 expression in breast cancer. International (Ludwig) Breast Cancer Study Group. J Clin Oncol 10:1049–1056
Pegram MD, Pauletti G, Slamon DJ (1998) HER-2/neu as a predictive marker of response to breast cancer therapy. Breast Cancer Res Treat 52:65–77
Paik S, Bryant J, Tan-Chiu E et al (2002) Real-world performance of HER2 testing—National Surgical Adjuvant Breast and Bowel Project experience. J Natl Cancer Inst 94:852–854
Rhodes A, Jasani B, Anderson E et al (2002) Evaluation of HER-2/neu immunohistochemical assay sensitivity and scoring on formalin-fixed and paraffin-processed cell lines and breast tumors: a comparative study involving results from laboratories in 21 countries. Am J Clin Pathol 118:408–417
Rhodes A, Jasani B, Couturier J et al (2002) A formalin-fixed, paraffin-processed cell line standard for quality control of immunohistochemical assay of HER-2/neu expression in breast cancer. Am J Clin Pathol 117:81–89
. DAKO A/S, F. Hoffmann-La Roche Ltd (eds) (2002) Atlas of interpretation of HercepTestTM staining. (2000)
Wolff AC, Hammond ME, Schwartz JM et al (2007) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25:118–145
Mayr D, Diebold J (2000) Grading of ovarian carcinomas. Int J Gynecol Pathol 19:348–353
Pauletti G, Godolphin W, Press MF et al (1996) Detection and quantitation of HER-2/neu gene amplification in human breast cancer archival material using fluorescence in situ hybridization. Oncogene 13:63–72
Bankfalvi A, Simon R, Brandt B et al (2000) Comparative methodological analysis of erbB-2/HER-2 gene dosage, chromosomal copy number and protein overexpression in breast carcinoma tissues for diagnostic use. Histopathology 37:411–419
Press MF, Slamon DJ, Flom KJ et al (2002) Evaluation of HER-2/neu gene amplification and overexpression: comparison of frequently used assay methods in a molecularly characterized cohort of breast cancer specimens. J Clin Oncol 20:3095–3105
Tubbs RR, Pettay JD, Roche PC et al (2001) Discrepancies in clinical laboratory testing of eligibility for trastuzumab therapy: apparent immunohistochemical false positives do not get the message. J Clin Oncol 19:2714–2721
Dowsett M, Hanby AM, Laing R et al (2007) HER2 testing in the UK: consensus from a national consultation. J Clin Pathol 60:685–689
Gouvea AP, Milanezi F, Olson SJ et al (2006) Selecting antibodies to detect Her2 overexpression by immunohistochemistry in invasive mammary carcinomas. Appl Immunohistochem Mol Morphol 14:103–108
Powell WC, Hicks DG, Prescott N et al (2007) A new rabbit monoclonal antibody (4B5) for immunohistochemical (IHC) determination of Her2 status in breast cancer: comparison with CB11, fluorescence in situ hybridization (FISH), and interlaboratory reproducibility. Appl Immunohistochem Mol Morphol 15:94–102
Ricardo SAV, Milanezi F, Carvalho ST et al (2008) Her2 evaluation using the novel rabbit monoclonal antibody SP3 and CISH in tissue microarrays of invasive breast. J Clin Path 60:1001–1005
Brunelli M, Manfrin E, Martignoni G et al (2008) Her-2/neu assessment in breast cancer using the original FDA and new ASCO/CAP guideline recommendations: impact on selecting patients for Herceptin therapy. Am J Clin Pathol 129:907–911
Moeder CB, Giltnane JM, Harigopal M et al (2007) Quantitative justification of the change from 10% to 30% for human epidermal growth factor receptor 2 scoring in the American Society of Clinical Oncology/College of American Pathologists guidelines: tumor heterogeneity in breast cancer and its implications for tissue microarray-based assessment of outcome. J Clin Oncol 25:5418–5425
Acknowledgments
The authors thank Gabi Charell, Gabi Janssen, and Annegret Schäfer for their outstanding technical contribution, Simone Fenn for critically reading the manuscript, and Jutta Engel for statistical analysis.
Conflict of interest statement
All authors declare that no conflict exists for devices or material used in the current study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mayr, D., Heim, S., Werhan, C. et al. Comprehensive immunohistochemical analysis of Her-2/neu oncoprotein overexpression in breast cancer: HercepTest™ (Dako) for manual testing and Her-2/neuTest 4B5 (Ventana) for Ventana BenchMark automatic staining system with correlation to results of fluorescence in situ hybridization (FISH). Virchows Arch 454, 241–248 (2009). https://doi.org/10.1007/s00428-009-0728-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-009-0728-8