Abstract
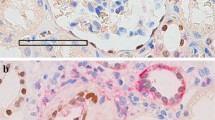
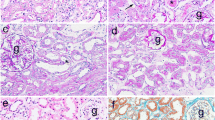
Tubulo-interstitial fibrosis in dogs may result from primary injury to the interstitium or develop secondary to other renal diseases. As in human renal pathology, tubular epithelial cells (TEC) are believed to actively participate in the mechanisms of renal fibrosis. In this study, we examined the changes in the tubular epithelial component in two specific canine diseases. Immunohistochemistry showed the expression of the epithelial marker cytokeratin, the smooth muscle marker α-SMA, the mesenchymal marker vimentin and PCNA in 20 dogs with membranous glomerulonephritis and membrano-proliferative glomerulonephritis. Results showed that the loss of the epithelial marker in TEC was directly correlated to the grade of tubulo-interstitial disease present and independent of the type of glomerulonephritis. Varying degrees of vimentin positivity were detected in tubular epithelium in areas of inflammation, and low numbers of scattered α-SMA-positive cells were also observed. Immunohistochemistry showed that epithelial tubular cells lose their cytokeratin staining characteristics and transdifferentiate into cells exhibiting key mesenchymal immunophenotypic feature of vimentin-positive staining in both diseases investigated. The integrity of the tubular basement membrane is likely to be fundamental in maintaining the epithelial phenotype of TEC. Animal models provide opportunities for investigating the pathogenesis of renal fibrosis in humans.



Similar content being viewed by others
References
Ben-Ze’ev A (1984) Differential control of cytokeratins and vimentin synthesis by cell-cell contact and cell spreading in cultured epithelial cells. J Cell Biol 99:1424–1433
Ben-Ze’ev A (1986) Tumor promoter-induced disruption of junctional complexes in cultured epithelial cells is followed by the inhibition of cytokeratin and desmoplakin synthesis. Exp Cell Res 164:335–352
Choi YJ, Chakraborty S, Nguyen V et al (2000) Peritubular capillary loss is associated with chronic tubulointerstitial injury in human kidney: altered expression of vascular endothelial growth factor. Hum Pathol 31:1491–1497
Eardley KS, Cockwell P (2005) Macrophages and progressive tubulointerstitial disease. Kidney Int 68:437–455
Eckardt KU, Rosenberger C, Jurgensen JS, Wiesener MS (2003) Role of hypoxia in the pathogenesis of renal disease. Blood Purif 21:253–257
Iwano M, Plieth D, Danoff TM, Xue C, Okada H, Neilson EG (2002) Evidence that fibroblasts derive from epithelium during tissue fibrosis. J Clin Invest 110:341–350
Kalluri R, Neilson EG (2003) Epithelial–mesenchymal transition and its implications for fibrosis. J Clin Invest 112:1776–1784
Kriz W, LeHir M (2005) Pathways to nephron loss starting from glomerular diseases-insights from animal models. Kidney Int 67:404–419
Kuusniemi AM, Lapatto R, Holmberg C, Karikoski R, Rapola J, Jalanko H (2005) Kidneys with heavy proteinuria show fibrosis, inflammation, and oxidative stress, but no tubular phenotypic change. Kidney Int 68:121–132
Liu Y (2004) Epithelial to mesenchymal transition in renal fibrogenesis: pathologic significance, molecular mechanism, and therapeutic intervention. J Am Soc Nephrol 15:1–12
Nangaku M (2006) Chronic hypoxia and tubulointerstitial injury: a final common pathway to end-stage renal failure. J Am Soc Nephrol 17:17–25
Nangaku M (2004) Mechanisms of tubulointerstitial injury in the kidney: final common pathways to end-stage renal failure. Intern Med 43:9–17
Rastaldi MP, Ferrario F, Giardino L et al (2002) Epithelial–mesenchymal transition of tubular epithelial cells in human renal biopsies. Kidney Int 62:137–146
Remuzzi G, Bertani T (1998) Pathophysiology of progressive nephropathies. N Engl J Med 339:1448–1456
Rodriguez-Iturbe B, Johnson RJ, Herrera-Acosta J (2005) Tubulointerstitial damage and progression of renal failure. Kidney Int Suppl 99:S82–S86
Scanziani E, Grieco V, Salvi S (1993) Expression of vimentin in the tubular epithelium of bovine kidneys with interstitial nephritis. Vet Pathol 30:298–293
Sinniah R, Khan TN (1999) Renal tubular basement membrane changes in tubulointerstitial damage in patients with glomerular diseases. Ultrastruct Pathol 23:359–368
Suzuki T, Kimura M, Asano M, Fujigaki Y, Hishida A (2001) Role of atrophic tubules in development of interstitial fibrosis in microembolism-induced renal failure in rat. Am J Pathol 158:75–85
Vilafranca M, Wohlsein P, Trautwein G (1995) Expression of class II major histocompatibility complex molecules in renal tubular epithelial cells of canine kidneys affected with tubulointerstitial nephritis. Res Vet Sci 59:114–117
Vongwiwatana A, Tasanarong A, Rayner DC, Melk A, Halloran PF (2005) Epithelial to mesenchymal transition during late deterioration of human kidney transplants: the role of tubular cells in fibrogenesis. Am J Transplant 5:1367–1374
Willis BC, duBois RM, Borok Z (2006) Epithelial origin of myofibroblasts during fibrosis in the lung. Proc Am Thorac Soc 3:377–382
Yamate J, Kuribayashi M, Kuwamura M, Kotani T, Ogihara K (2005) Differential immunoexpressions of cytoskeletons in renal epithelial and interstitial cells in rat and canine fibrotic kidneys, and in kidney-related cell lines under fibrogenic stimuli. Exp Toxicol Pathol 57:135–147
Yang B, Wagner BE, Savill J, El Nahas AM (1998) Cellular apoptosis and proliferation in experimental renal fibrosis. Nephrol Dial Transplant 13:2216–2226
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aresu, L., Rastaldi, M.P., Scanziani, E. et al. Epithelial–mesenchymal transition (EMT) of renal tubular cells in canine glomerulonephritis. Virchows Arch 451, 937–942 (2007). https://doi.org/10.1007/s00428-007-0482-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-007-0482-8