Abstract
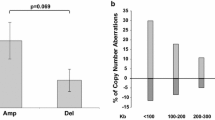
The susceptibility of thyroid epithelium to radiation-induced carcinogenesis is well recognised. In this context, thyroid carcinogenesis is associated with specific somatic ret/papillary thyroid carcinoma (PTC) rearrangements and morphologically with the papillary phenotype. Previous studies have demonstrated the possibility of inducing ret rearrangements in vitro using X-rays. The purpose of our study was to assess whether gamma (γ) radiation using a Caesium 137 source can induce specific ret rearrangements in a human thyroid epithelial cell culture model. We further hypothesised that if radiation-induced thyroid carcinogenesis is associated with non-random rearrangement events, then DNA copy gain and loss induced by irradiation may also occur in a non-random manner. We irradiated SV40-immortalised human thyroid epithelial cells with incremental doses of γ-radiation and, using TaqMan reverse-transcription polymerase chain reaction, looked for the presence of the common ret rearrangements. Cohorts showing evidence of ret/PTC chimeric transcripts were further analysed using microarray comparative genomic hybridisation (CGH) to detect copy gain and loss associated with radiation. Four Grays of γ-radiation was sufficient to induce ret/PTC-3. In this model, transcripts of ret/PTC-1 were not detected, and we suggest that the type of radiation may influence the resulting rearrangement that occurs. Using array CGH, we have demonstrated a predominant pattern of subtelomeric deletions occurring in association with this radiation cohort and raise the possibility that chromosome 10 may be a hotspot for radiation-induced damage for as yet unknown reasons.


Similar content being viewed by others
References
Rubino C, Cailleux AF, De Valthaire F, Schlumberger M (2002) Thyroid cancer after radiation exposure. Eur J Cancer 38:645–647
Ron E, Lubin JH, Shore RE, Mabuchi K, Modan B, Pottern LM, Schneider AB, Tucker MA, Boice JD Jr (1995) Thyroid cancer after exposure to external radiation: a pooled analysis of seven studies. Radiat Res 141:259–277
Lundell M, Hakulinen T, Holm LE (1994) Thyroid cancer after radiotherapy for skin hemangioma in infancy. Radiat Res 140:334–339
De Vathaire F, Hardiman C, Shamsaldin A, Campbell S, Grimaud E, Hawkins M, Raquin M, Oberlin O, Diallo I, Zucker JM, Panis X, Lagrange JL, Daly-Schveitzer N, Lemerle J, Chavaudra J, Schlumberger M, Bonaiti C (1999) Thyroid carcinomas after irradiation for a first cancer during childhood. Arch Int Med 159:2713–2720
Hamilton TE, Van Belle G, LoGerfo JP (1987) Thyroid carcinoma in Marshall Islanders exposed to nuclear fallout. JAMA 258:629–636
Kazakov VS, Demidchik EP, Astakhova LN (1992) Thyroid cancer after Chernobyl. Nature 359:21–22
Shibata Y, Yamashita S, Masyakin VB, Panasyuk GD, Nagataki S (2001) 15 years after Chernobyl: new evidence of thyroid cancer. Lancet 358:1965–1966
Santoro M, Carlomagno F, Hay ID, Herrmann MA, Grieco M, Melillo R, Pierotti MA, Bongarzone I, Della-Porta G, Berger N, Paulin C, Fabien N, Vecchio G, Jenkins RB, Fusco A (1992) Ret oncogene activation in human thyroid neoplasms is restricted to the papillary cancer subtype A. J Clin Invest 89:1517–1522
Kimura ET, Nikiforova MN, Zhaowen Z, Knauf J, Nikiforov YE, Fagin JA (2003) High prevalence of BRAF mutations in thyroid carcinoma: genetic evidence for constitutive activation of the RET/PTC-RASBRAF signaling pathway in papillary thyroid carcinoma. Cancer Res 63:1454–1457
Soares P, Trovisco V, Rocha AS, Lima J, Castro P, Preto A, Maimo V, Botelho T, Seruca R, Sobrinho-Simoes M (2003) BRAF mutations and RET/PTC rearrangements are alternative events in the etiopathogenesis of PTC. Oncogene 22:4578–4580
Xu X, Quiros RM, Gattuso P, Ain KB, Prinz RA (2003) High prevalence of BRAF gene mutations in papillary thyroid carcinoma and thyroid tumour cell lines. Cancer Res 63:4561–4567
Xing M, Vasko V, Tallini G, Larin A, Wu G, Udelsman R, Ringel MD, Ladenson PW, Sidransky D (2004) BRAF T1796A transversion mutation in various thyroid neoplasms. J Clin Endocrinol Metab 89:1365–1368
Tallini G, Asa SL (2001) Ret oncogene activation in papillary thyroid carcinoma. Adv Anat Pathol 8:345–354
Thomas GA, H. Bunnell, Cook HA, Williams ED, Nerovnya A, Cherstvoy ED, Tronko ND, Bogdanova TI, Chiappetta G, Viglietto G, Pentimalli F, Salvatore G, Fusco A, Santoro M, Vecchio G (2001) High prevalence of RET/PTC rearrangements in Ukrainian and Belarusian post-Chernobyl thyroid papillary carcinomas: a strong correlation between RET/PTC3 and the solid-follicular variant. J Clin Endocrinol Metab 84:4232–4238
Kallioniemi A, Kallioniemi OP, Sudar D, Rutovitz D, Gray JW, Waldman F, Pinkel D (1992) Comparative genomic hybridization for molecular cytogenetic analysis of solid tumours. Science 258:818–821
Hui AB, Lo KW, Yin XL, Poon WS, Ng HK (2001) Detection of multiple gene amplifications in glioblastoma multiforme using array based comparative genomic hybridization. Lab Invest 81:717–723
Pollack JR, Sørlie T, Perou CM, Rees CA, Jeffrey SS, Lonning PE, Tibshirani R, Botstein D, Børresen-Dale AL, Brown PO (2002) Microarray analysis revels a major direct role of DNA copy number alteration in the transcriptional program of human breast tumours. PNAS 99:12963–12968
Wilhelm M, Veltman JA, Osshen AB, Jain AN, Moore DH, Presti JC, Kocacs G, Waldman FM (2002) Array-based comparative genomic hybridization for the differential diagnosis of renal cell cancer. Cancer Res 62:957–960
Weiss M, Hermsen MAJA, Meijer GA, van Grieken NCT, Baak JPA, Kuipers EJ, van Diest PJ (1999) Demystified...comparative genomic hybridization. J Clin Pathol 52:243–251
Riches AC, Herceg Z, Bryant PE, Wynford-Thomas D (1994) Radiation-induced transformation of SV40-immortalized human thyroid epithelial cells by single and fractionated exposure to γ-irradiation in vitro. Int J Radiat Biol 66:757–765
Zitelsberger H, Bruch J, Smida J, Hieber L, Peddie CM, Bryant PE, Riches AC, Fung J, Weier HU, Bauchinger M (2001) Clonal chromosomal aberrations in Simian virus 40-transfected thyroid cells and in tumors developed after in vitro irradiation. Int J Cancer 96:166–177
Ito T, Seyama T, Iwamoto KS, Hayashi T, Mizuno T, Tsuyama N, Dohi K, Nakamura N, Akiyama M (1993) In vitro irradiation is able to cause RET oncogene rearrangement. Cancer Res 53:2940–2943
Mizuno T, Iwamoto KS, Kyoizumi S, Nagamura H, Shinohara T, Koyama K, Seyama T, Hamatani K (2000) Preferential induction of RET/PTC1 rearrangement by X-ray irradiation. Oncogene 19:438–443
Williams ED (2002) Cancer after nuclear fallout: lessons from the Chernobyl accident. Nature 2:543–549
Gavrilin YI, Khrouch VT, Shinkarev SM, Krysenko NA, Skryabin AM, Bouville A, Anspaugh LR (1999) Chernobyl accident: reconstruction of thyroid dose for inhabitants of the Republic of Belarus. Health Phys 76:105–119
Lemoine NR, Mayall ES, Jones T, Sheer D, McDermid S, Kendall-Taylor P, Wynford-Thomas D (1989) Characteristics of human thyroid epithelial cells immortalized in vitro by simian virus 40 DNA transfection. Br J Cancer 60:897–903
Pacini F, Vivaldi A, Santoro M, Fedele M, Fusco A, Romei C, Basolo F, Pinchera A (1998) Simian virus 40-like DNA sequences in human papillary thyroid carcinomas. Oncogene 16:665–669
Finn SP, Smyth P, O’Leary JJ, Sweeney EC, Sheils OM (2003) Ret/PTC chimeric transcripts in an Irish cohort of sporadic papillary thyroid carcinoma. J Clin Endocrinol Metab 88:938–941
Wright EG (1999) Inherited and inducible chromosomal instability: a fragile bridge between genome integrity mechanisms and tumourigenesis. J Pathol 187:19–27
Riches A, Peddie C, Rendell S, Bryant P, Zitzelsberger H, Bruch J, Smida J, Hieber L, Bauchinger M (2001) Neoplastic transformation and cytogenetic changes after Gamma irradiation of human epithelial cells expressing telomerase. Radiat Res 155:222–229
Smida J, Salassidis K, Hieber L, Zitzelsberger H, Kellerer AM, Demidchik EP, Negele T, Spelsberg F, Lengfelder E, Werner M, Bauchinger M (1999) Distinct frequency of ret rearrangements in papillary thyroid carcinomas of children and adults from Belarus. Int J Cancer 80:32–38
Bounacer A, Wicker R, Caillou B, Cailleux AF, Sarasin A, Schlumberger M, Suarez HG (1997) High prevalence of activating ret proto-oncogene rearrangements, in thyroid tumors from patients who had received external radiation. Oncogene 15:1263–1273
Sarasin A, Bounacer A, Lepage F, Schlumberger M, Suarez HG (1999) Mechanisms of mutagenesis in mammalian cells. Application to human thyroid tumours. C R Acad Sci III 322:143–149
Latre L, Tusell L, Martin M, Miro R, Egozcue J, Blasco MA, Genesca A (2003) Shortened telomeres join to DNA breaks interfering with their correct repair. Exp Cell Res 287:282–288
Sabatier L, Lebeau J, Dutrillaux B (1995) Radiation-induced carcinogenesis: individual sensitivity and genomic instability. Radiat Environ Biophys 34:229–232
Hemmer S, Wesenius VM, Knuutila S, Franssila K, Joensuu H (1999) DNA copy number changes in thyroid carcinoma. Am J Pathol 154:1539–1547
Kjellman P, Lagercrantz S, Hoog A, Wallin G, Larsson C, Zedenius J (2001) Gain of 1q and loss of 9q21.3-q32 are associated with a less favorable prognosis in papillary thyroid carcinoma. Genes Chromosomes Cancer 32:43–49
Zitelsberger H, Lehmann L, Hieber L, Weier HG, Janish C, Fung J, Negele T, Spelsberg F, Lengfelder E, Demidchik E, Salassidis K, Kellerer AM, Werner M, Bauchinger M (1999) Cytogenetic changes in radiation induced tumors of the thyroid. Cancer Res 59:135–140
Acknowledgements
This work was supported by a grant from the Health Research Board of Ireland. Thanks to Professor Sissy Jhiang for providing ret/PTC-3 plasmid used for positive control. Thanks to Professor Kingston Mills for providing irradiation facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Finn, S.P., Smyth, P., O’Regan, E. et al. Array comparative genomic hybridisation analysis of gamma-irradiated human thyrocytes. Virchows Arch 445, 396–404 (2004). https://doi.org/10.1007/s00428-004-1070-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-004-1070-9