Abstract.
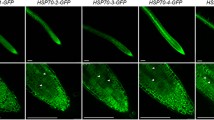
Similar to heat-stress transcription factors (HSFs) from non-plant sources, HSFA1 and HSFA2 from tomato (Lycopersicon esculentum Mill) contain two conserved clusters of basic amino acid residues (K/R1 and K/R2) which might serve as nuclear localization signal (NLS) motifs. Mutation of either one of them and functional testing of the corresponding proteins in a transient expression assay using tobacco (Nicotiana plumbaginifolia L.) protoplasts gave the following results. Whereas K/R1, positioned in all HSFs at the C-terminus of the DNA-binding domain, had no influence on nuclear import, the K/R1 mutants were impaired in their interaction with the DNA (band-shift assays). In contrast to this, mutants of the K/R2 motif, found 15–20 amino acid residues C-terminal of the oligomerization domain (HR-A/B region), had wild-type activity in DNA-binding but were defective in nuclear import. Thus, for the related tomato HSFA1 and HSFA2 the K/R2 cluster represents the only NLS motif, and in this function it cannot be replaced by K/R1.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 16 August 1996 / Accepted: 30 December 1996
Rights and permissions
About this article
Cite this article
Lyck, R., Harmening, U., Höhfeld, I. et al. Intracellular distribution and identification of the nuclear localization signals of two plant heat-stress transcription factors. Planta 202, 117–125 (1997). https://doi.org/10.1007/s004250050110
Issue Date:
DOI: https://doi.org/10.1007/s004250050110