Abstract
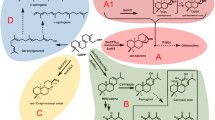
Catharanthus roseus is an important medicinal plant and the sole commercial source of monoterpenoid indole alkaloids (MIA), anticancer compounds. Recently, triterpenoids like ursolic acid and oleanolic acid have also been found in considerable amounts in C. roseus leaf cuticular wax layer. These simple pentacyclic triterpenoids exhibit various pharmacological activities such as anti-inflammatory, anti-tumor and anti-microbial properties. Using the EST collection from C. roseus leaf epidermome (http://www.ncbi.nlm.nih.gov/dbEST), we have successfully isolated a cDNA (CrAS) encoding 2,3-oxidosqualene cyclase (OSC) and a cDNA (CrAO) encoding amyrin C-28 oxidase from the leaves of C. roseus. The functions of CrAS and CrAO were analyzed in yeast (Saccharomyces cerevisiae) systems. CrAS was characterized as a novel multifunctional OSC producing α- and β-amyrin in a ratio of 2.5:1, whereas CrAO was a multifunctional C-28 oxidase converting α-amyrin, β-amyrin and lupeol to ursolic-, oleanolic- and betulinic acids, respectively, via a successive oxidation at the C-28 position of the substrates. In yeast co-expressing CrAO and CrAS, ursolic- and oleanolic acids were detected in the yeast cell extracts, while the yeast cells co-expressing CrAO and AtLUP1 from Arabidopsis thaliana produced betulinic acid. Both CrAS and CrAO genes show a high expression level in the leaf, which was consistent with the accumulation patterns of ursolic- and oleanolic acids in C. roseus. These results suggest that CrAS and CrAO are involved in the pentacyclic triterpene biosynthesis in C. roseus.






Similar content being viewed by others
Abbreviations
- CrAS:
-
Catharanthus roseus mixed amyrin synthase
- CrAO:
-
Catharanthus roseus amyrin oxidase
- EST:
-
Expressed sequence tag
- ORF:
-
Open reading frame
- LC–MS:
-
Liquid chromatogram mass spectrum
- P450:
-
Cytochrome P450 monoxygenase
- QRT-PCR:
-
Quantitative reverse transcription polymerase chain reaction
- RACE:
-
Rapid amplification of cDNA ends
- TAIL-PCR:
-
Thermal asymmetric interlaced polymerase chain reaction
References
Abe I, Rohmer M, Prestwich GD (1993) Enzymatic cyclization of squalene and oxidosqualene to sterols and triterpenes. Chem Rev 93:2189–2206
Augustin JM, Kuzina V, Andersen SB, Bak S (2011) Molecular activities, biosynthesis and evolution of triterpenoid saponins. Phytochemistry 72:435–457
Basyuni M, Oku H, Inafuku M, Baba S, Iwasaki H, Oshiro K, Okabe T, Shibuya M, Ebizuka Y (2006) Molecular cloning and functional expression of a multifunctional triterpene synthase cDNA from a mangrove species Kandelia candel (L.) Druce. Phytochemistry 67:2517–2524
Bohlmann J, Keeling CI (2008) Terpenoid biomaterials. Plant J 54:656–669
Brendolise C, Yauk Y-K, Eberhard ED, Wang M, Chagne D, Andre C, Greenwood DR, Beuning LL (2011) An unusual plant triterpene synthase with predominant α-amyrin producing activity identified by characterising oxidosqualene cyclases from Malus × domestica. FEBS J 278:2485–2499
Bringe K, Schumacher CFA, Schmitz-Eiberger M, Steiner U, Oerke E-C (2006) Ontogenetic variation in chemical and physical characteristics of adaxial apple leaf surfaces. Phytochemistry 67:161–170
Carelli M, Biazzi E, Panara F, Tava A, Scaramelli L, Porceddu A, Graham N, Odoardi M, Piano E, Arcioni S, May S, Scotti C, Calderini O (2011) Medicago truncatula CYP716A12 is a multifunctional oxidase involved in the biosynthesis of hemolytic saponins. Plant Cell 23:3070–3081
El-Sayed M, Verpoorte R (2007) Catharanthus terpenoid indole alkaloids: biosynthesis and regulation. Phytochem Rev 6:277–305
Farina C, Pinza M, Pifferi G (1998) Synthesis and anti-ulcer activity of new derivatives of glycyrrhetic, oleanolic and ursolic acids. Il Farmaco 53:22–32
Fazio GC, Xu R, Matsuda SP (2004) Genome mining to identify new plant triterpenoids. J Am Chem Soc 126:5678–5679
Fukushima EO, Seki H, Ohyama K, Ono E, Umemoto N, Mizutani M, Saito K, Muranaka T (2011) CYP716A subfamily members are multifunctional oxidases in triterpenoid biosynthesis. Plant Cell Physiol 52:2050–2061
Fulda S (2008) Betulinic acid for cancer treatment and prevention. Int J Mol Sci 9:1096–1107
Gietz RD, Woods RA (2002) Transformation of yeast by lithium acetate/single-stranded carrier DNA/polyethylene glycol method. Methods Enzymol 350:87–96
Grncarevic M, Radler F (1971) A review of the surface lipids of grapes and their importance in the drying process. AM J Enol Vitic 20:80–86
Guhling O, Hobl B, Yeats T, Jetter R (2006) Cloning and characterization of a lupeol synthase involved in the synthesis of epicuticular wax crystals on stem and hypocotyl surfaces of Ricinus communis. Arch Biochem Biophys 448:60–72
Hayashi H, Hiraoka N, Ikeshiro Y, Kushiro T, Morita M, Shibuya M, Ebizuka Y (2000) Molecular cloning and characterization of a cDNA for Glycyrrhiza glabra cycloartenol synthase. Biol Pharm Bull 23:231–234
Hayashi H, Huang P, Inoue K, Hiraoka N, Ikeshiro Y, Yazaki K, Tanaka S, Kushiro T, Shibuya M, Ebizuka Y (2001) Molecular cloning and characterization of isomultiflorenol synthase, a new triterpene synthase from Luffa cylindrica, involved in biosynthesis of bryonolic acid. Eur J Biochem 268:6311–6317
Helliwell CA, Poole A, Peacock WJ, Dennis ES (1999) Arabidopsis ent-kaurene oxidase catalyzes three steps of gibberellin biosynthesis. Plant Physiol 119:507–510
Herrera JB, Bartel B, Wilson WK, Matsuda SP (1998) Cloning and characterization of the Arabidopsis thaliana lupeol synthase gene. Phytochemistry 49:1905–1911
Husselstein-Muller T, Schaller H, Benveniste P (2001) Molecular cloning and expression in yeast of 2,3-oxidosqualene-triterpenoid cyclases from Arabidopsis thaliana. Plant Mol Biol 45:75–92
Kajikawa M, Yamato KT, Fukuzawa H, Sakai Y, Uchida H, Ohyama K (2005) Cloning and characterization of a cDNA encoding beta-amyrin synthase from petroleum plant Euphorbia tirucalli L. Phytochemistry 66:1759–1766
Kirby J, Romanini DW, Paradise EM, Keasling JD (2008) Engineering triterpene production in Saccharomyces cerevisiae-beta-amyrin synthase from Artemisia annua. FEBS J 275:1852–1859
Kushiro T, Shibuya M, Ebizuka Y (1998) β-Amyrin synthase cloning of oxidosqualene cyclase that catalyzes the formation of the most popular triterpene among higher plants. Eur J Biochem 256:238–244
Liu J (1995) Pharmacology of oleanolic acid and ursolic acid. J Ethnopharmacol 49:57–68
Liu J (2005) Oleanolic acid and ursolic acid: research perspectives. J Ethnopharmacol 100:92–94
Liu YG, Whittier RF (1995) Thermal asymmetric interlaced PCR: automatable amplification and sequencing of insert end fragments from P1 and YAC clones for chromosome walking. Genomics 25:674–681
Morita M, Shibuya M, Kushiro T, Masuda K, Ebizuka Y (2000) Molecular cloning and functional expression of triterpene synthases from pea. Eur J Biochem 267:3453–3460
Murata J, Roepke J, Gordon H, De Luca V (2008) The leaf epidermome of Catharanthus roseus reveals its biochemical specialization. Plant Cell 20:524–542
O’Connor SE, Maresh JJ (2006) Chemistry and biology of monoterpene indole alkaloid biosynthesis. Nat Prod Rep 23:532–547
Pereira SI, Freire CSR, Pascoal Neto C, Silvestre AJD, Silva AMS (2005) Chemical composition of the epicuticular wax from the fruits of Eucalyptus globulus. Phytochem Anal 16:364–369
Phillips DR, Rasbery JM, Bartel B, Matsuda SPT (2006) Biosynthetic diversity in plant triterpene cyclization. Curr Opin Plant Biol 9:305–314
Pompon D, Louerat B, Bronine A, Urban P (1996) Yeast expression of animal and plant P450s in optimized redox environments. Methods Enzymol 272:51–64
Qi X, Bakht S, Qin B, Leggett M, Hemmings A, Mellon F, Eagles J, Werck-Reichhart D, Schaller H, Lesot A, Melton R, Osbourn A (2006) A different function for a member of an ancient and highly conserved cytochrome P450 family: from essential sterols to plant defense. Proc Natl Acad Sci USA 103:18848–18853
Reiling KK, Yoshikuni Y, Martin VJJ, Newman J, Bohlmann J, Keasling JD (2004) Mono and diterpene production in Escherichia coli. Biotechnol Bioeng 87:200–212
Ro D, Arimura G, Lau S, Piers E, Bohlmann J (2005) Loblolly pine abietadienol/abietadienal oxidase PtAO (CYP720B1) is a multifunctional, multisubstrate cytochrome P450 monooxygenase. Proc Natl Acad Sci USA 102:8060–8065
Ro D-K, Paradise EM, Ouellet M, Fisher KJ, Newman KL, Ndungu JM, Ho KA, Eachus RA, Ham TS, Kirby J, Chang MCY, Withers ST, Shiba Y, Sarpong R, Keasling JD (2006) Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440:940–943
Seki H, Ohyama K, Sawai S, Mizutani M, Ohnishi T, Sudo H, Akashi T, Aoki T, Saito K, Muranaka T (2008) Licorice β-amyrin 11-oxidase, a cytochrome P450 with a key role in the biosynthesis of the triterpene sweetener glycyrrhizin. Proc Natl Acad Sci USA 105:14204–14209
Shibuya M (2004) Cucurbitadienol synthase, the first committed enzyme for cucurbitacin biosynthesis, is a distinct enzyme from cycloartenol synthase for phytosterol biosynthesis. Tetrahedron 60:6995–7003
Shibuya M, Zhang H, Endo A, Shishikura K, Kushiro T, Ebizuka Y (1999) Two branches of the lupeol synthase gene in the molecular evolution of plant oxidosqualene cyclases. Eur J Biochem 266:302–307
Shibuya M, Hoshino M, Katsube Y, Hayashi H, Kushiro T, Ebizuka Y (2006) Identification of beta-amyrin and sophoradiol 24-hydroxylase by expressed sequence tag mining and functional expression assay. FEBS J 273:948–959
Shibuya M, Katsube Y, Otsuka M, Zhang H, Tansakul P, Xiang T, Ebizuka Y (2009) Identification of a product specific beta-amyrin synthase from Arabidopsis thaliana. Plant Physiol Biochem 47:26–30
Smimaru H, Orihara Y, Tansakul P, Kang YH, Shibuya MYE (2007) Production of triterpene acids by cell suspension cultures of Olea europaea. Chem Pharm Bull 55:784–788
Teoh KH, Polichuk DR, Reed DW, Nowak G, Covello PS (2006) Artemisia annua L. (Asteraceae) trichome-specific cDNAs reveal CYP71AV1, a cytochrome P450 with a key role in the biosynthesis of the antimalarial sesquiterpene lactone artemisinin. FEBS Lett 580:1411–1416
Usia T, Watabe T, Kadota S, Tezuka Y (2005) Cytochrome P450 2D6 (CYP2D6) inhibitory constituents of Catharanthus roseus. Biol Pharm Bull 28:1021–1024
van der Heijden R, Jacobs DI, Snoeijer W, Hallared D, Verpoorte R (2004) The Catharanthus alkaloids: pharmacognosy and biotechnology. Curr Med Chem 11:607–628
Wang Z, Guhling O, Yao R, Li F, Yeats TH, Rose JKC, Jetter R (2010) Two oxidosqualene cyclases responsible for biosynthesis of tomato fruit cuticular triterpenoids. Plant Physiol 155:540–552
Xiong Q, Wilson WK, Matsuda SP (2006) An Arabidopsis oxidosqualene cyclase catalyzes iridal skeleton formation by Grob fragmentation. Angew Chem Int Ed Engl 45:1285–1288
Yu MML, Konorov SO, Schulze HG, Blades MW, Turner RFB, Jetter R (2007) In situ analysis by microspectroscopy reveals triterpenoid compositional patterns within leaf cuticles of Prunus laurocerasus. Planta 227:823–834
Acknowledgments
This project was partially supported by the Grant for One Hundred Talents Program of the Chinese Academy of Sciences, China (Project No. Y129441R01), the National Science and Technology Program of China during the Twelveth Five-Year Plan period and the National Natural Science Foundation of China (Project No. 91733A1001).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, L., Li, J., Ye, H. et al. Molecular characterization of the pentacyclic triterpenoid biosynthetic pathway in Catharanthus roseus . Planta 236, 1571–1581 (2012). https://doi.org/10.1007/s00425-012-1712-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-012-1712-0