Abstract
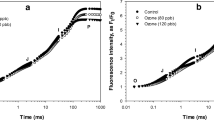
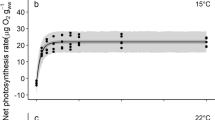
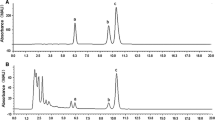
The emission of molecular iodine (I2) from the stipe, the meristematic area and the distal blade of the brown macroalga Laminaria digitata (Hudson) Lamouroux (Phaeophyceae) was monitored under low light and dark conditions. Photosynthetic parameters were determined to investigate both the extent of stress experienced by different thallus parts and the effects of emersion on photosynthesis. Immediately after air exposure, intense I2 emission was detectable from all thallus parts. I2 emission declined continuously over a period of 180 min following the initial burst, but was not affected by the light regime. The total number of mole of I2 emitted by stipes was approximately 10 times higher than those emitted from other thallus parts. Initial I2 emission rates (measured within 30 min of exposure to air) were highest for stipes (median values: 2,999 and 5,222 pmol g−1 dw min−1 in low light and dark, respectively) and lower, by one order of magnitude, for meristematic regions and distal blades. After exposure to air for between 60 and 180 min, I2 emission rates of all thallus parts were reduced by 70–80%. Air exposure resulted in a decrease of the maximum photosystem II (PSII) efficiency (F v/F m) by 3%, and in a 25–55% increase of the effective PSII quantum efficiency (\( \Updelta F/F^{\prime}_{\text{m}} \)); this was caused by a higher fraction of open reaction centres (qP), whereas the efficiency of the latter in capturing energy (\( F^{\prime}_{\text{v}} /F^{\prime}_{\text{m}} \)) remained constant. The results indicate the presence of an iodine pool which is easily volatilised and depleted due to air exposure, even under apparently low stress conditions.



Similar content being viewed by others
Abbreviations
- IBBCEAS:
-
Incoherent broadband cavity-enhanced absorption spectroscopy
- HWHM:
-
Half-width at half-maximum
- MBL:
-
Marine boundary layer
- NPQ:
-
Non-photochemical quenching
- PFD:
-
Photon flux density
- PAR:
-
Photosynthetic active radiation
- PS:
-
Photosystem
References
Amat MA, Srivastava LM (1985) Translocation of iodine in Laminaria saccharina (Phaeophyta). J Phycol 21:330–333
Ar Gall E, Küpper FC, Kloareg B (2004) A survey of iodine content in Laminaria digitata. Bot Mar 47:30–37
Bale CSE, Ingham T, Commane R, Heard DE, Bloss WJ (2008) Novel measurements of atmospheric iodine species by resonance fluorescence. J Atmos Chem 60:51–70
Ball SM, Hollingsworth SA, Humbles J, Leblanc C, Potin P, McFiggans G (2010) Spectroscopic studies of molecular iodine emitted into the gas phase by seaweed. Atmos Chem Phys 10:6237–6254
Bitter M, Ball SM, Povey IM, Jones RL (2005) A broadband cavity ringdown spectrometer for in situ measurements of atmospheric trace gases. Atmos Chem Phys 5:2547–2560
Büchel C, Wilhelm C (1993) In vivo analysis of slow fluorescence induction kinetics in algae: progress, problems and perspectives. J Photochem Photobiol 58:137–148
Carpenter LJ (2003) Iodine in the marine boundary layer. Chem Rev 103:4953–4962
Dethier MN, Williams SL (2009) Seasonal stresses shift optimal intertidal algal habitats. Mar Biol 156:555–567
Dixneuf S, Ruth AA, Vaughan S, Varma RM, Orphal J (2009) The time dependence of molecular iodine emission from Laminaria digitata. Atmos Chem Phys 9:823–829
Dring MJ, Brown FA (1982) Photosynthesis of intertidal brown algae during and after periods of emersion: a renewed search for physiological causes of zonation. Mar Ecol Prog Ser 8:301–308
Evans LV, Holligan MS (1972) Correlated light and electron microscope studies on brown algae. II. Physode production in Dictyota. New Phytol 71:1173–1180
Fiedler SE, Hese A, Ruth AA (2003) Incoherent broad-band cavity-enhanced absorption spectroscopy. Chem Phys Lett 371:284–294
Garbary DJ, Kim KY (2005) Anatomical differentiation and photosynthetic adaptation in brown algae. Algae 20:233–238
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron-transport and quenching of chlorophyll fluorescence. Biochem Biophys Acta 990:87–92
Gevaert F, Creach A, Davoult D, Holl AC, Seuront L, Lemoine Y (2002) Photo-inhibition and seasonal photosynthetic performance of the seaweed Laminaria saccharina during a simulated tidal cycle: chlorophyll fluorescence measurements and pigment analysis. Plant Cell Environ 25:859–872
Gómez I, López-Figueroa F, Ulloa N, Morales V, Lovengreen C, Huovinen P, Hess S (2004) Patterns of photosynthesis in 18 species of intertidal macroalgae from southern Chile. Mar Ecol Prog Ser 270:103–116
Harker M, Berkaloff C, Lemoine Y, Britton G, Young A, Duval JC, Rmiki N-E, Rousseau B (1999) Effects of high light and desiccation on the operation of the xanthophyll cycle in two marine brown algae. Eur J Phycol 34:35–42
Haug A, Jensen A (1954) Seasonal variations in the chemical composition of Alaria esculenta, Laminaria saccharina, Laminaria hyperborea and Laminaria digitata from northern Norway. Norwegian Institute of Seaweed Research, Report no. 84, p 34
Hou X, Hansena V, Aldahanb A, Possnert G, Lindd OC, Lujaniene G (2009) A review on speciation of iodine-129 in the environmental and biological samples. Anal Chim Acta 632:181–196
Huang RJ, Seitz K, Buxmann J, Pöhler D, Hornsby KE, Carpenter LJ, Platt U, Hoffmann T (2010) In situ measurements of molecular iodine in the marine boundary layer: the link to macroalgae and the implications for O3, IO, OIO and NO x . Atmos Chem Phys 10:4823–4833
Hunt LJH, Denny MW (2008) Desiccation protection and disruption: a trade-off for an intertidal marine alga. J Phycol 44:1164–1170
Johnson WS, Gigon A, Gulmon SL, Mooney HA (1974) Comparative photosynthetic capacities of intertidal algae under exposed and submerged conditions. Ecology 55:450–453
Kolb CE (2002) Iodine’s air of importance. Nature 417:597–598
Krause GH, Weis E (1991) Chlorophyll fluorescence and photosynthesis: the basics. Annu Rev Plant Physiol Plant Mol Biol 42:313–349
Kromkamp JC, Forster RM (2003) The use of variable fluorescence measurements in aquatic ecosystems: differences between multiple and single turnover measuring protocols and suggested terminology. Eur J Phycol 38:103–112
Küpper FC, Schweigert N, Ar Gall E, Legendre JM, Vilter H, Kloareg B (1998) Iodine uptake in Laminariales involves extracellular, haloperoxidase-mediated oxidation of iodide. Planta 207:163–171
Küpper FC, Kloareg B, Guern J, Potin P (2001) Oligoguluronates elicit an oxidative burst in the brown algal kelp Laminaria digitata. Plant Physiol 125:278–291
Küpper FC, Carpenter LJ, McFiggans GB, Palmer CJ, Waite TJ, Boneberg E-M, Woitsch S, Weiller M, Abela R, Grolimund D, Potin P, Butler A, Luther GW, Kroneck PMH, Meyer-Klaucke W, Feiters MC (2008) Iodide accumulation provides kelp with an inorganic antioxidant impacting atmospheric chemistry. Proc Natl Acad Sci USA 105:6954–6958
Kylin H (1929) Über das Vorkommen von Jodiden, Bromiden und Jodidoxidasen bei den Meeresalgen. Hoppe-Seylor’s Z Physiol Chem 186:50–84
Laturnus F, Svensson T, Wiencke C, Öberg G (2004) Ultraviolet radiation affects emission of ozone-depleting substances by marine macroalgae: results from a laboratory incubation study. Environ Sci Technol 38:6605–6609
Leblanc C, Colin C, Cosse A, Delage L, La Barre S, Morin P, Fiévet B, Voiseux C, Ambroise Y, Verhaeghe E, Amouroux D, Donard O, Tessier E, Potin P (2006) Iodine transfers in the coastal marine environment: the key role of brown algae and of their vanadium-dependent haloperoxidases. Biochimie 88:1773–1785
Linger P, Brüggemann W (1999) Correlations between chlorophyll fluorescence quenching parameters and photosynthesis in a segregating Lycopersicon esculentum × L. peruvianum population as measured under constant conditions. Photosynth Res 61:145–156
Lobban CS, Harrisson PJ (1994) Seaweed ecology and physiology. Cambridge University Press, Cambridge
Lüning K (1985) Meeresbotanik. Gustav Thieme Verlag, Stuttgart
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence − a practical guide. J Exp Bot 51:659–668
McFiggans G, Coe H, Burgess R, Allan J, Cubison M, Alfarra MR, Saunders R, Saiz-Lopez A, Plane JMC, Wevill DJ, Carpenter LJ, Rickard AR, Monks PS (2004) Direct evidence for coastal iodine particles from Laminaria macroalgae – linkage to emissions of molecular iodine. Atmos Chem Phys 4:701–713
McFiggans G, Bale CSE, Ball SM, Beames JM, Bloss WJ, Carpenter LJ, Dorsey J, Dunk R, Flynn MJ, Furneaux KL, Gallagher MW, Heard DE, Hollingsworth AM, Hornsby K, Ingham T, Jones CE, Jones RL, Kramer LJ, Langridge JM, Leblanc C, LeCrane JP, Lee JD, Leigh RJ, Longley I, Mahajan AS, Monks PS, Oetjen H, Orr-Ewing AJ, Plane JMC, Potin P, Shillings AJL, Thomas F, von Glasow R, Wada R, Whalley LK, Whitehead JD (2010) Iodine-mediated coastal particle formation: an overview of the Reactive Halogens in the Marine Boundary Layer (RHaMBLe) Roscoff coastal study. Atmos Chem Phys 10:2975–2999
O’Dowd CD, Hoffmann T (2005) Coastal new particle formation: a review of the current state-of-the-art. Environ Chem 2:245–255
O’Dowd CD, Jimenez JL, Bahreini R, Flagan RC, Seinfeld JH, Hameri K, Pirjola L, Kulmala M, Jennings SG, Hoffmann T (2002) Marine aerosol formation from biogenic iodine emissions. Nature 417:632–636
Palmer CJ, Anders TL, Carpenter LJ, Küpper FC, McFiggans GB (2005) Iodine and halocarbon response of Laminaria digitata to oxidative stress and links to atmospheric new particle production. Environ Chem 2:282–290
Pedersén M, Roomans GM (1983) Ultrastructural localization of bromine and iodine in the stipes of Laminaria digitata (Huds.) Lamour., Laminaria saccharina (L.) Lamour. and Laminaria hyperborea (Gunn.) Fosl. Bot Mar 26:113–118
Saenko GN, Kravtsova YY, Ivanenko VV, Sheludko SI (1978) Concentration of iodine and bromine by plants in seas of Japan and Okhotsk. Mar Biol 47:243–250
Saiz-Lopez A, Plane JMC (2004) Novel iodine chemistry in the marine boundary layer. Geophys Res Lett 31:L04112
Saiz-Lopez A, Saunders RW, Joseph DM, Ashworth SH, Plane JMC (2004) Absolute absorption cross-section and photolysis rate of I2. Atmos Chem Phys 4:1443–1450
Schonbeck M, Norton TA (1978) Factors controlling upper limits of fucoid algae on shore. J Exp Mar Biol Ecol 31:303–313
Schreiber U, Schliwa U, Bilger W (1986) Continuous recording of photochemical and nonphotochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. Photosynth Res 10:51–62
Seitz K, Buxmann J, Pöhler D, Sommer T, Tschritter J, Neary T, O’Dowd C, Platt U (2010) The spatial distribution of the reactive iodine species IO from simultaneous active and passive DOAS observations. Atmos Chem Phys 10:2117–2128
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research. Freeman, New York
van den Hoek C, Mann DG, Jahns HM (1995) Algae: an introduction to phycology. Cambridge University Press, Cambridge
Verhaeghe E, Fraysse A, Guerquin-Kern J-L, Wu T-D, Devès G, Mioskowski C, Leblanc C, Ortega R, Ambroise Y, Potin P (2008) Microchemical imaging of iodine distribution in the brown alga Laminaria digitata suggests a new mechanism for its accumulation. J Biol Inorg Chem 13:257–269
Wiltens J, Schreiber U, Vidaver W (1978) Chlorophyll fluorescence induction – indicator of photosynthetic activity in marine algae undergoing desiccation. Can J Bot Rev Can Bot 56:2787–2794
Acknowledgments
The authors thank Professor Colin O’Dowd (School of Physics and Ryan Institute, NUI Galway), Dr. Solène Connan (Botany and Plant Science and Ryan Institute, NUI Galway) and Dr. Stewart Vaughan (School of Chemistry, University of Leeds, UK) for comments on the experimental design and Dr. Jerome Sheahane (Statistics and Applied Mathematics, School of Mathematics, NUI Galway) for his help with statistical analyses. UN and SD gratefully appreciate financial support through the Irish Research Council for Science, Engineering and Technology (IRCSET ‘Embark Initiative’).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nitschke, U., Ruth, A.A., Dixneuf, S. et al. Molecular iodine emission rates and photosynthetic performance of different thallus parts of Laminaria digitata (Phaeophyceae) during emersion. Planta 233, 737–748 (2011). https://doi.org/10.1007/s00425-010-1334-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-010-1334-3