Abstract
cyclo-Oxylipin-galactolipids (cGL) are mono- or digalactosyldiglycerides carrying a cyclo-oxylipin in the sn1- and/or sn2-position or esterified to the galactose moiety. These compounds were recently identified in Arabidopsis thaliana. We provide evidence that cGL are mainly, if not exclusively, part of the thylakoid and can be hydrolysed by lipolytic activities associated with photosynthesis-related protein complexes in vitro. Using HPLC/ESI–mass spectrometry, cGL are shown to be restricted in occurrence to the genus Arabidopsis, they do not occur in other plants tested. A. thaliana cGL are rapidly and transiently formed upon wounding with characteristic changes in composition of the cGL-fraction. While the biological role of cGL is not understood, the genus Arabidopsis may present a model-case of chemical evolution of a novel class of regulatory molecules.




Similar content being viewed by others
Abbreviations
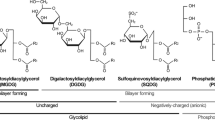
- DGDG:
-
Digalactosyldiacylglycerol
- cGL:
-
cyclo-Oxylipin-galactolipids
- JA:
-
Jasmonic acid
- LHCP:
-
Light harvesting complex protein
- OPDA:
-
12-Oxophytodienoic acid
- MGDG:
-
Monogalactosyldiacylglycerol
- PG:
-
Phosphatidylglycerol
- PSI (II):
-
Photosystem I (II)
- TPT:
-
Triose phosphate translocator
References
Andersson MX, Hamberg M, Kourtchenko O, Brunnström Å, McPhail KL, Gerwick WH, Göbel C, Feussner I, Ellerström M (2006) Oxylipin-profiling of the hypersensitive response in Arabidopsis thaliana: formation of a novel oxo-phytodienoic acid containing galactolipid, arabidopside E. J Biol Chem 281:31528–31537
Beckers GJM, Spoel SH (2005) Fine-tuning plant defence signalling: salicylate versus Jasmonate. Plant Biol 8:1–10
Blechert S, Brodschelm W, Hölder S, Kammerer L, Kutchan TM, Mueller MJ, Xia ZQ, Zenk MH (1995) The octadecanoic pathway: signal molecules for the regulation of secondary pathways. Proc Natl Acad Sci USA 92:4099–4105
Buseman CM, Tamura P, Sparks AA, Baughman EJ, Maatta S, Zhao J, Roth MR, Wynn Esch S, Shah J, Williams TD, Welti R (2006) Wounding stimulates the accumulation of glycerolipids containing oxophytodienoic and dinor-oxophytodienoic acid in Arabidopsis leaves. Plant Physiol 142:28–39
Delker C, Stenzel I, Hause B, Miersch O, Feussner I, Wasternack C (2006) Jasmonate biosynthesis in Arabidopsis thaliana—enzymes, products, regulation. Plant Biol 8:1–10
Demandre C, Tremolieres A, Justin AM, Mazliak P (1985) Analysis of molecular species of plant polar lipids by high-performance and gas liquid chromatography. Phytochemistry 24:481–485
Dörmann P, Hoffmann-Benning S, Balbo I, Benning C (1995) Isolation and characterization of an Arabidopsis mutant deficient in the thylakoid lipid digalactosyl diacylglycerol. Plant Cell 7:1801–1810
Ferro M, Salvi D, Brugière S, Miras S, Kowalski S, Louwagie M, Garin J, Joyard J, Rolland N (2003) Proteomics of the chloroplast envelope membranes from Arabidopsis thaliana. Mol Cell Proteomics 2:325–345
Feussner I, Kuhn H, Wasternack C (2001) Lipoxygenase-dependent degradation of storage lipids. Trends Plant Sci 6:268–273
Gounaris K, Whitford D, Barber J (1983) The effect of thylakoid lipids on an oxygen-evolving photosystem II preparation. FEBS Lett 163:230–234
Gundlach H, Zenk MH (1998) Biological activity and biosynthesis of pentacyclic oxylipins: the linoleic acid pathway. Phytochemistry 47:527–537
Guo J, Zhang Z, Bi Y, Yang W, Xu Y, Zhang L (2005) Decreased stability of photosystem I in dgd1 mutant of Arabidopsis thaliana. FEBS Lett 579:3619–3624
Härtel H, Dörmann P, Benning C (2000) DGD1-independent biosynthesis of extraplastidic galactolipids after phosphate deprivation in Arabidopsis. Proc Natl Acad Sci USA 97:10649–10654
Hisamatsu Y, Goto N, Hasegawa K, Shigemori H (2003) Arabidopsides A and B, two new oxylipins from Arabidopsis thaliana. Tetrahedron Lett 44:5553–5556
Hisamatsu Y, Goto N, Sekiguchi M, Hasegawa K, Shigemori H (2005) Oxylipins arabidopsides C and D from Arabidopsis thaliana. J Nat Prod 68:600–603
Hisamatsu Y, Goto N, Hasegawa K, Shigemori H (2006) Senescence-promoting effects of arabidopside A. Z Naturforsch 61:363–366
Ishiguro S, Kwai-Oda A, Ueda J, Nishida I, Okada K (2001) The defective in anther dehiscence1 gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation. Plant Cell 13:2191–2209
Jarvis P, Dörmann P, Charles AP, Jason L, Beninng C, Joanne C (2000) Galactolipid deficiency and abnormal chloroplast development in the Arabidopsis MGD synthase 1 mutant. Proc Natl Acad Sci USA 97:8175–8179
Jordan P, Fromme P, Witt HT, Klukas O, Saenger W, Krauß N (2001) Three-dimensional structure of cyanobacterial photosystem I at 2.5 Å resolution. Nature 411:909–917
Joyard J, Maréchal E, Block MA, Douce R (1996) Plant galactolipids and sulfolipid: structure, distribution and biosynthesis. In: Smallwood M, Knox P, Bowles DJ (ed) Membranes: specialized functions in plants. BIOS Scientific Publishers, Oxford, pp 179–194
Krupa Z, Baszynski T (1975) Requirement of galactolipids for photosystem I activity in lyophilized spinach chloroplasts. Biochim Biophys Acta 408:26–34
Kunst L (1998) Preparation of physiologically active chloroplasts from Arabidopsis. In: Martinez-Zapater JM, Salinas J (eds) Methods in molecular biology, Arabidopsis protocols, vol. 82. Humana Press, Totowa, pp 43–48
Kutchan TM (1993) 12-Oxo-phytodienoic acid induces accumulation of berberine bridge enzyme transcripts in a manner analogous to methyl jasmonate. J Plant Physiol 142:502–505
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Laudert D, Weiler EW (1998) Allene oxide synthase: a major control point in Arabidopsis thaliana octadecanoid signalling. Plant J 15:675–684
Laudert D, Pfannschmidt U, Lottspeich F, Holländer-Czytko H, Weiler EW (1996) Cloning, molecular and functional characterization of Arabidopsis thaliana allene oxide synthase (CYP 74), the first enzyme of the octadecanoid pathway to jasmonates. Plant Mol Biol 31:323–335
Li HM, Moore T, Keegstra K (1991) Targeting of proteins to the outer envelope membrane uses a different pathway than transport into chloroplasts. Plant Cell 3:709–717
Liu Z, Yan H, Wang K, Kuang T, Zhang J, Gui L, An X, Chang W (2004) Crystal structure of spinach major light-harvesting complex at 2.72 Å resolution. Nature 428:287–292
Loll B, Kern J, Saenger W, Zouni A, Biesiadka J (2005) Towards complete cofactor arrangement in the 3.0 Å resolution structure of photosystem II. Nature 438:1040–1044
Murata N, Fujimura Y, Higashi S (1990) Glycerolipids in various preparations of photosystem II from spinach chloroplasts. Biochim Biophys Acta 1019:261–268
Nussberger S, Dörr K, Wang DN, Kuhlbrandt W (1993) Lipid–protein interactions in crystals of plant light-harvesting complex. J Mol Biol 234:347–356
Pick U, Weiss M, Gounaris K, Barber J (1987) The role of different thylakoid glycolipids in the function of reconstituted chloroplasts ATP synthase. Biochim Biophys Acta 891:28–39
Schägger H, von Jagow G (1991) Blue-native electrophoresis for isolation of membrane protein complexes in enzymatically active form. Anal Biochem 199:223–231
Schaller F, Schaller A, Stinzi A (2005) Biosynthesis and metabolism of jasmonates. J Plant Growth Regul 23:179–199
Schleiff E, Klösgen RB (2001) Without a little help from my friends: direct insertion of proteins into chloroplast membranes? Biochim Biophys Acta 1541:22–33
Steffen R, Kelly AA, Huyer J, Dörmann P, Renger G (2005) Investigations on the reaction pattern of photosystem II in leaves from Arabidopsis thaliana wild type plants and mutants with genetically modified lipid content. Biochemistry 44:3134–3142
Stelmach BA, Müller A, Hennig P, Laudert D, Andert L, Weiler EW (1997) Quantitation of the octadecanoid 12-oxo-phytodienoic acid, a signalling compound in plant mechanotransduction. Phytochemistry 47:539–546
Stelmach BA, Müller A, Hennig P, Gebhardt S, Schubert-Zsilavecz M, Weiler EW (2001) A novel class of oxylipins, sn1-O-(12-oxophytodienoyl)-sn2-O-(hexadecatrienoyl)-monogalactosyl diglyceride from Arabidopsis thaliana. J Biol Chem 276:12832–12838
Stinzi A, Weber H, Reymond P, Browse J, Farmer EE (2001) Plant defense in the absence of jasmonic acid: the role of cyclopentenones. Proc Natl Acad Sci USA 98:12837–12842
Stroebel D, Choquet Y, Popot JL, Picot D (2003) An atypical haem in the cytochrome b6f complex. Nature 426:413–418
Taki N, Sasaki-Sekimoto Y, Obayashi T, Kikuta A, Kobayashi K, Ainai T, Yagi K, Sakurai N, Suzuki H, Masuda T, Takamiya K, Shibata D, Kobayashi Y, Ohta H (2005) 12-Oxo-phytodienoic acid triggers expression of a distinct set of genes and plays a role in wound-induced gene expression in Arabidopsis. Plant Physiol 139:1268–1283
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamid gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Turner JG, Ellis C, Devoto A (2002) The jasmonate signal pathway. Plant Cell Suppl S153-S164
Vidi PA, Kanwischer M, Baginsky S, Austin JR, Csucs G, Dörmann P, Kessler F, Bréhélin C (2006) Tocopherol cyclase (VTE1) localization and vitamin E accumulation in chloroplast plastoglobule lipoprotein particles. J Biol Chem 281:11225–11234
Weiler EW, Albrecht T, Groth B, Xia ZQ, Luxem M, Liß H, Andert L, Spengler P (1993) Evidence for the involvement of jasmonates and their octadecanoid precursors in the tendril coiling response of Bryonia dioica. Phytochemistry 32:591–600
Ytterberg AJ, Peltier JB, van Wijk KJ (2006) Protein profiling of plastoglobules in chloroplasts and chromoplasts. A surprising site for differential accumulation of metabolic enzymes. Plant Physiol 140:984–997
Zerbe P, Weiler EW, Schaller F (2007) Preparative enzymatic solid phase synthesis of cis(+)-12-oxo-phytodienoic acid–physical interaction of AOS and AOC is not necessary. Phytochemistry 68:229–236
Acknowledgments
We thank Jun-Prof. Danja Schünemann (Lehrstuhl für Allgemeine und Molekulare Botanik, Ruhr-Universität Bochum, Germany) for providing the LHCP and cpSRP43 antibodies as well as the secondary antibody from chicken. We also like to thank Prof. Ulf-Ingo Flügge (Lehrstuhl für Botanik II, Universität zu Köln, Germany) for the kind provision of the TPT antibody. Furthermore we thank Dr. Markus Piotrowski (Lehrstuhl für Pflanzenphysiologie, Ruhr-Universität Bochum, Germany) for the mass spectrometrical analyses of the thylakoid protein complexes. This work was funded by a grant of the Deutsche Forschungsgemeinschaft (SFB480, TP A8) to EWW.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Böttcher, C., Weiler, E.W. cyclo-Oxylipin-galactolipids in plants: occurrence and dynamics. Planta 226, 629–637 (2007). https://doi.org/10.1007/s00425-007-0511-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-007-0511-5