Abstract
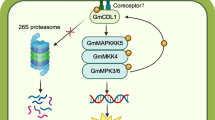
Plants recognize microbial pathogens by discriminating pathogen-associated molecular patterns from self-structures. We study the non-host disease resistance of soybean (Glycine max L.) to the oomycete, Phytophthora sojae. Soybean senses a specific molecular pattern consisting of a branched heptaglucoside that is present in the oomycetal cell walls. Recognition of this elicitor may be achieved through a β-glucan-binding protein, which forms part of a proposed receptor complex. Subsequently, soybean mounts a complex defense response, which includes the increase of the cytosolic calcium concentration, the production of reactive oxygen species, and the activation of genes responsible for the synthesis of phytoalexins. We now report the identification of two mitogen-activated protein kinases (MAPKs) and one MAPK kinase (MAPKK) that may function as signaling elements in triggering the resistance response. The use of specific antisera enabled the identification of GmMPKs 3 and 6 whose activity is enhanced within the signaling pathway leading to defense reactions. Elicitor specificity of MAPK activation as well as the sensitivity against inhibitors suggested these kinases as part of the β-glucan signal transduction pathway. An upstream GmMKK1 was identified based on sequence similarity to other plant MAPKKs and its interaction with the MAPKs was analyzed. Recombinant GmMKK1 interacted predominantly with GmMPK6, with concomitant phosphorylation of the MAPK protein. Moreover, a preferential physical interaction between GmMKK1 and GmMPK6 was demonstrated in yeast. These results suggest a role of a MAPK cascade in mediating β-glucan signal transduction in soybean, similar to other triggers that activate MAPKs during innate immune responses in plants.



Similar content being viewed by others
Abbreviations
- A9C:
-
Anthracene-9-carboxylate
- DP:
-
Degree of polymerization
- DPI:
-
Diphenyleneiodonium
- EGTA:
-
Ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid
- EST:
-
Expressed sequence tag
- IMAC:
-
Immobilized metal chelate affinity chromatography
- MAPK:
-
(MPK) mitogen-activated protein kinase
- MAPKK:
-
(MKK) MAPK kinase
- MBP:
-
Myelin basic protein
- NPPB:
-
5-Nitro-2-(3-phenylpropylamino)-benzoate
- PAGE:
-
Polyacrylamide gelelectrophoresis
- SA:
-
Salicylic acid
- SSC:
-
Standard saline citrate
References
Asai T, Tena G, Plotnikova J, Willmann MR, Chiu W-L, Gomez-Gomez L, Boller T, Ausubel FM, Sheen J (2002) MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415:977–983
Bögre L, Ligterink W, Meskiene I, Barker PJ, Heberle-Bors E, Huskisson NS, Hirt H (1997) Wounding induces the rapid and transient activation of a specific MAP kinase pathway. Plant Cell 9:75–83
Boyle WJ, van der Geer P, Hunter T (1991) Phosphopeptide mapping and phosphoamino acid analysis by two-dimensional separation on thin-layer cellulose plates. Methods Enzymol 201:110–149
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 282:152–160
Brunner F, Rosahl S, Lee J, Rudd JJ, Geiler C, Kauppinen S, Rasmussen G, Scheel D, Nürnberger T (2002) Pep-13, a plant defense-inducing pathogen-associated pattern from Phytophthora transglutaminases. EMBO J 21:6681–6688
Cardinale F, Jonak C, Ligterink W, Niehaus K, Boller T, Hirt H (2000) Differential activation of four specific MAPK pathways by distinct elicitors. J Biol Chem 275:36734–36740
Cardinale F, Meskiene I, Ouaked F, Hirt H (2002) Convergence and divergence of stress-induced mitogen-activated protein kinase signaling pathways at the level of two distinct mitogen-activated protein kinase kinases. Plant Cell 14:703–711
Chinchilla D, Bauer Z, Regenass M, Boller T, Felix G (2006) The Arabidopsis receptor kinase FLS2 binds flg22 and determines the specificity of flagellin perception. Plant Cell 18:465–476
Contor L, Lamy F, Lecocq RE (1987) Use of electroblotting to detect and analyze phosphotyrosine containing peptides separated by two-dimensional gel electrophoresis. Anal Biochem 160:414–420
Corpet F (1988) Multiple sequence alignment with hierarchical clustering. Nucl Acids Res 16:10881–10890
Cosio EG, Frey T, Verduyn R, van Boom J, Ebel J (1990) High-affinity binding of a synthetic heptaglucoside and fungal glucan phytoalexin elicitors to soybean membranes. FEBS Lett 271:223–226
del Pozo O, Pedley KF, Martin GB (2004) MAPKKKα is a positive regulator of cell death associated with both plant immunity and disease. EMBO J 23:3072–3082
Ebel J, Mithöfer A (1998) Early events in the elicitation of plant defense. Planta 206:335–348
Felix G, Duran JD, Volko S, Boller T (1999) Plants have a sensitive perception system for the most conserved domain of bacterial flagellin. Plant J 18:265–276
Fliegmann J, Mithöfer A, Wanner G, Ebel J (2004) An ancient enzyme domain hidden in the putative β-glucan elicitor receptor of soybean may play an active part in the perception of pathogen-associated molecular patterns during broad host resistance. J Biol Chem 279:1132–1140
Fliegmann J, Schüler G, Boland W, Ebel J, Mithöfer A (2003) The role of octadecanoids and functional mimics in soybean defense responses. Biol Chem 384:437–446
Hall TA (1999) BioEdit. A user-friendly biological sequence alignment and analysis program for Windows 95/98/NT. Nucl Acid Symp Ser 41:95–98
He P, Shan L, Lin N-C, Martin GB, Kemmerling B, Nürnberger T, Sheen J (2006) Specific bacterial suppressors of MAMP signaling upstream of MAPKKK in Arabidopsis innate immunity. Cell 125:563–575
Hille A, Purwin C, Ebel J (1982) Induction of enzymes of phytoalexin synthesis in cultured soybean cells by an elicitor from Phytophthora megasperma f. sp. glycinea. Plant Cell Rep 1:123–127
Huang Y, Li H, Gupta R, Morris PC, Luan S, Kieber JJ (2000) AtMPK4, an Arabidopsis homolog of mitogen-activated protein kinase, is activated in vitro by AtMEK1 through threonine phosphorylation. Plant Physiol 122:1301–1310
Janeway CA Jr, Medzhitov R (2002) Innate immune recognition. Annu Rev Immunol 20:197–216
Jonak C, Kiegerl S, Ligterink W, Barker PJ, Huskisson NS, Hirt H (1996) Stress signaling in plants: a mitogen-activated protein kinase pathway is activated by cold and drought. Proc Natl Acad Sci USA 93:11274–11279
Jonak C, Pay A, Bögre L, Hirt H, Heberle-Bors E (1993) The plant homologue of MAP kinase is expressed in a cell cycle-dependent and organ-specific manner. Plant J 3:611–617
Kiegerl S, Cardinale F, Siligan C, Gross A, Baudouin E, Liwosz A, Eklöf S, Till S, Bögre L, Hirt H, Meskiene I (2000) SIMKK, a mitogen-activated protein kinase (MAPK) kinase, is a specific activator of the salt stress-induced MAPK, SIMK. Plant Cell 12:2247–2258
Kroj T, Rudd JJ, Nürnberger T, Gäbler Y, Lee J, Scheel D (2003) Mitogen-activated protein kinases play an essential role in oxidative burst-independent expression of pathogenesis-related genes in parsley. J Biol Chem 278:2256–2264
Lebrun-Garcia A, Ouaked F, Chiltz A, Pugin A (1998) Activation of MAPK homologues by elicitors in tobacco cells. Plant J 15:773–781
Lee J, Klessig DF, Nürnberger T (2001b) A harpin binding site in tobacco plasma membranes mediates activation of the pathogenesis-related gene HIN1 independent of extracellular calcium but dependent on mitogen-activated protein kinase activity. Plant Cell 13:1079–1093
Lee J, Rudd JJ, Macioszek VK, Scheel D (2004) Dynamic changes in the localization of MAPK cascade components controlling pathogenesis-related (PR) gene expression during innate immunity in parsley. J Biol Chem 279:22440–22448
Lee S, Hirt H, Lee Y (2001a) Phosphatidic acid activates a wound-activated MAPK in Glycine max. Plant J 26:479–486
Liu YH, Zhang SQ, Klessig DF (2000) Molecular cloning and characterization of a tobacco MAP kinase kinase that interacts with SIPK. Mol Plant-Microbe Interact 13:118–124
MAPK Group (2002) Mitogen-activated protein kinase cascades in plants: a new nomenclature. Trends Plant Sci 7:301–308
Medzhitov R, Janeway CA Jr (1997) Innate immunity: the virtues of a nonclonal system of recognition. Cell 91:295–298
Mithöfer A, Bhagwat AA, Feger M, Ebel J (1996) Suppression of fungal β-glucan-induced plant defence in soybean (Glycine max L.) by cyclic 1,3–1,6-β-glucans from the symbiont Bradyrhizobium japonicum. Planta 199:270–275
Mithöfer A, Daxberger A, Fromhold-Treu D, Ebel J (1997) Involvement of an NAD(P)H oxidase in the elicitor-inducible oxidative burst of soybean. Phytochemistry 45:1101–1107
Mithöfer A, Ebel J, Bhagwat AA, Boller T, Neuhaus-Url G (1999) Transgenic aequorin monitors cytosolic calcium transients in soybean cells challenged with β-glucan or chitin elicitors. Planta 207:566–574
Mithöfer A, Ebel J, Felle H (2005) Cation fluxes cause plasma membrane depolarization involved in β-glucan elicitor-signaling in soybean roots. Mol Plant-Microbe Interact 18:983–990
Mithöfer A, Fliegmann J, Daxberger A, Ebel C, Neuhaus U, Bhagwat AA, Keister DL, Ebel J (2001) Induction of H2O2 synthesis by β-glucan elicitors in soybean is independent on cytosolic calcium transients. FEBS Lett 508:191–195
Mithöfer A, Fliegmann J, Neuhaus-Url G, Schwarz H, Ebel J (2000) The hepta-β-glucoside elicitor-binding proteins from legumes represent a putative receptor family. Biol Chem 381:705–713
Mizoguchi T, Hayashida N, Yamaguchi-Shinozaki K, Kamada H, Shinozaki K (1993) AtMPKs: a gene family of plant MAP kinases in Arabidopsis thaliana. FEBS Lett 336:440–444
Munnik T, Ligterink W, Meskiene I, Calderini O, Beyerly J, Musgrave A, Hirt H (1999) Distinct osmo-sensing protein kinase pathways are involved in signalling moderate and severe hyper-osmotic stress. Plant J 20:381–388
Nakagami H, Pitzschke A, Hirt H (2005) Emerging MAPK pathways in plant stress signaling. Trends Plant Sci 10:339–346
Nühse TS, Peck SC, Hirt H, Boller T (2000) Microbial elicitors induce activation and dual phosphorylation of the Arabidopsis thaliana MAPK6. J Biol Chem 275:7521–7526
Nürnberger T, Scheel D (2001) Signal transmission in the plant immune response. Trends Plant Sci 6:372–379
Nürnberger T, Brunner F, Kemmerling B, Piater L (2004) Innate immunity in plants and animals: similarities and obvious differences. Immunol Rev 198:249–266
Pedley KF, Martin GB (2004) Identification of MAPKs and their possible MAPKK activators involved in the Pto-mediated defense response of tomato. J Biol Chem 279:49229–49235
Pedley KF, Martin GB (2005) Role of mitogen-activated protein kinases in plant immunity. Curr Opin Plant Biol 8:541–547
Ren D, Yang H, Zhang S (2002) Cell death mediated by MAPK is associated with hydrogen peroxide production in Arabidopsis. J Biol Chem 277:559–565
Romeis T (2001) Protein kinases in the plant defence response. Curr Opin Plant Biol 4:407–414
Seo S, Okamoto M, Seto H, Ishizuka K, Sano H, Ohashi Y (1995) Tobacco MAP kinase: a possible mediator in wound signal transduction pathways. Science 270:1988–1992
Sharp JK, McNeil M, Albersheim P (1984b) The primary structures of one elicitor-active and seven elicitor-inactive hexa(β-D-glucopyranosyl)-D-glucitols isolated from the mycelial walls of Phytophthora megasperma f. sp. glycinea. J Biol Chem 259:11321–11336
Sharp JK, Valent B, Albersheim P (1984a) Purification and partial characterization of a beta-glucan fragment that elicits phytoalexin accumulation in soybean. J Biol Chem 259:11312–11320
Stäb MR, Ebel J (1987) Effects of Ca2+ on phytoalexin induction by fungal elicitor in soybean cells. Arch Biochem Biophys 257:416–423
Stratmann JW, Ryan CA (1997) Myelin basic protein kinase activity in tomato leaves is induced systemically by wounding and increases in response to systemin and oligosaccharide elicitors. Proc Natl Acad Sci USA 94:11085–11089
Suzuki K, Shinshi H (1995) Transient activation and tyrosine phosphorylation of a protein kinase in tobacco cells treated with a fungal elicitor. Plant Cell 7:639–647
Taylor ATS, Kim J, Low PS (2001) Involvement of mitogen-activated protein kinase activation in the signal-transduction pathways of the soya bean oxidative burst. Biochem J 355:795–803
Tena G, Asai T, Chiu W-L, Sheen J (2001) Plant mitogen-activated protein kinase signaling cascades. Curr Opin Plant Biol 4:392–400
Yang K-Y, Liu Y, Zhang S (2001) Activation of a mitogen-activated protein kinase pathway is involved in disease resistance in tobacco. Proc Natl Acad Sci USA 98:741–746
Zhang S, Klessig DF (1997) Salicylic acid activates a 48-kD MAP kinase in tobacco. Plant Cell 9:809–824
Zhang S, Klessig DF (2001) MAPK cascades in plant defense signaling. Trends Plant Sci 6:520–527
Acknowledgment
This work was supported by the Deutsche Forschungsgemeinschaft (SFB369).
Author information
Authors and Affiliations
Corresponding author
Additional information
The nucleotide sequences encoding the MAPKs and MAPKK1 from soybean can be accessed through the GenBank database under GenBank accession numbers AF104247, AF329506, and AY070230.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Daxberger, A., Nemak, A., Mithöfer, A. et al. Activation of members of a MAPK module in β-glucan elicitor-mediated non-host resistance of soybean. Planta 225, 1559–1571 (2007). https://doi.org/10.1007/s00425-006-0442-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-006-0442-6