Abstract
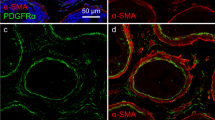
Here we investigate mechanisms underlying spontaneous phasic contractions (SPCs) and sympathetic control of contractility in the rat epididymis, a long tubular duct involved in transportation and maturation of sperm. Longitudinal contractions of short segments (~ 1.5 mm) of rat proximal and distal caudal epididymal duct were measured + / − nerve stimulation. The extent of sympathetic innervation of these duct regions was determined by immunohistochemistry. Proximal caudal duct segments (150–300 μm dia.) exhibited SPCs, while distal segments (350–500 μm) were quiescent in ~ 80% of preparations. SPC amplitude and frequency were reduced by the L-type voltage-dependent Ca2+ channel (LVDCC) blocker nifedipine (1 μM), with the T-type voltage-dependent Ca2+ channel (TVDCC) blocker ML218 (1 μM) specifically decreasing SPC frequency. SPCs were inhibited upon blockade of the SR/ER Ca2+-ATPase (CPA 10 μM). SPCs were also inhibited by caffeine (1 μM), 2-APB (100 μM), niflumic acid (100 μM), or by lowering extracellular [Cl−] from 134.4 to 12.4 mM but not by ryanodine (25 μM) or tetracaine (100 μM). Electrical field stimulation (EFS) at 2 Hz for 60 s caused a sustained α1-adrenoceptor-sensitive contraction in distal segments and enhanced and/or induced α2-adrenoceptor-sensitive oscillatory phasic contractions in proximal and distal segments, the latter mimicked by application of the α2-adrenoceptor agonist clonidine. We hypothesise that SPCs in the proximal cauda are triggered by pacemaker mechanisms involving rhythmic IP3 receptor–operated SR/ER store Ca2+ release and resultant activation of CaCC with TVDCCs and possibly LVDCCs subserving in this process. Sympathetic nerve-released noradrenaline induces α2-adrenoceptor-mediated phasic contractions in the proximal and distal cauda. These findings provide new pharmacological targets for male infertility and contraception.
Graphic Abstract









Similar content being viewed by others
Data availability
Data is available on demand from Professor Hikaru Hashitani in whose laboratory this research was undertaken.
Abbreviations
- 2-APB: :
-
2-Aminoethoxydiphenyl borate
- α-SMA:
-
α-Smooth muscle actin
- BSS:
-
Buffered saline solution
- CaCC:
-
Calcium-activated chloride channels
- CPA:
-
Cyclopiazonic acid
- DMSO:
-
Dimethyl sulphoxide
- EFS:
-
Electrical field stimulation
- ET-1:
-
Endothelin 1
- LVDCC:
-
L-type voltage-dependent Ca2+ channel
- IgG:
-
Immunoglobulin G
- SPCs:
-
Spontaneous phasic contractions
- SR/ER:
-
Sarcoendoplasmic reticulum
- TH:
-
Tyrosine hydroxylase
- TVDCC:
-
T-type voltage-dependent Ca2+ channel
References
Robaire B, Hinton BT, Orgebin-Crist M-C (2006) The epididymis. In: Neill JD (ed) Knobil and Neill’s physiology of reproduction-, 3rd edn. Elsevier, United States, pp 1071–1148
Takano H, Abe K, Ito T (1981) Changes in the mouse epididymis after ligation of the ductuli efferentes or proximal epididymal duct: qualitative and quantitative histological studies (author’s transl). Kaibogaku Zasshi 56:79–90
Turner TT, Gleavy JL, Harris JM (1990) Fluid movement in the lumen of the rat epididymis: effect of vasectomy and subsequent vasovasostomy. J Androl 11:422–428
Von L, Neuhaeuser G (1964) Morphometric analysis of the human epididymis. Z Anat Entwicklungsgesch 124:126–152
Dacheux JL, Dacheux F (2014) New insights into epididymal function in relation to sperm maturation. Reproduction 147:R27-42. https://doi.org/10.1530/REP-13-0420
Skerget S, Rosenow MA, Petritis K, Karr TL (2015) Sperm proteome maturation in the mouse epididymis. PLoS ONE 10:e0140650. https://doi.org/10.1371/journal.pone.0140650
Crabo B (1965) Studies on the composition of epididymal content in bulls and boars. Acta Vet Scand 22:1–94
Elfgen V, Mietens A, Mewe M, Hau T, Middendorff R (2018) Contractility of the epididymal duct: function, regulation and potential drug effects. Reproduction 156:R125–R141. https://doi.org/10.1530/REP-17-0754
Johnson AL, Howards SS (1975) Intratubular hydrostatic pressure in testis and epididymis before and after vasectomy. Am J Physiol 228:556–564. https://doi.org/10.1152/ajplegacy.1975.228.2.556
Mewe M, Bauer CK, Muller D, Middendorff R (2006) Regulation of spontaneous contractile activity in the bovine epididymal duct by cyclic guanosine 5’-monophosphate-dependent pathways. Endocrinology 147:2051–2062. https://doi.org/10.1210/en.2005-1324
Baumgarten HG, Falck B, Holstein AF, Owman C, Owman T (1968) Adrenergic innervation of the human testis, epididymis, ductus deferens and prostate: a fluorescence microscopic and fluorimetric study. Z Zellforsch Mikrosk Anat 90:81–95
Holstein AF (1969) Morphologische studien am nebenhoden des menschen. Zwanglose Abhandlungen aus dem Gebiet der Normalen und Pathologischen Anatomie 20:1-91
Jaakkola UM (1983) Regional variations in transport of the luminal contents of the rat epididymis in vivo. J Reprod Fertil 68:465–470
Mewe M, Bauer CK, Schwarz JR, Middendorff R (2006) Mechanisms regulating spontaneous contractions in the bovine epididymal duct. Biol Reprod 75:651–659. https://doi.org/10.1095/biolreprod.106.054577
Talo A, Jaakkola UM, Markkula-Viitanen M (1979) Spontaneous electrical activity of the rat epididymis in vitro. J Reprod Fertil 57:423–429
Hodson N (1970) The nerves of the testis, epididymis and scrotum. In: Gomes WR, Vandemark NL (eds) Johnson AD. Academic Press, New York, New York, pp 47–99
Mitchell GA (1935) The innervation of the kidney, ureter, testicle and epididymis. J Anat 70(10–32):15
Ricker DD (1998) The autonomic innervation of the epididymis: its effects on epididymal function and fertility. J Androl 19:1–4
Hib J (1976) Effects of autonomic drugs on epididymal contractions. Fertil Steril 27:951–956
El-Badawi A, Schenk EA (1967) The distribution of cholinergic and adrenergic nerves in the mammalian epididymis: a comparative histochemical study. Am J Anat 121:1–14. https://doi.org/10.1002/aja.1001210102
Grundy D (2015) Principles and standards for reporting animal experiments in the journal of physiology and experimental physiology. J Physiol 593:2547–2549. https://doi.org/10.1113/JP270818
Neild TO (1989) Measurement of arteriole diameter changes by analysis of television images. Blood Vessels 26:48–52
Hashitani H, Nguyen MJ, Noda H, Mitsui R, Higashi R, Ohta K, Nakamura KI, Lang RJ (2017) Interstitial cell modulation of pyeloureteric peristalsis in the mouse renal pelvis examined using FIBSEM tomography and calcium indicators. Pflugers Arch 469:797–813. https://doi.org/10.1007/s00424-016-1930-6
Brooks DE (1973) Epididymal and testicular temperature in the unrestrained conscious rat. JReprod Fertil 35:157–160. https://doi.org/10.1530/jrf.0.0350157
Bedford JM (1991) Effects of elevated temperature on the epididymis and testis: experimental studies. Adv Exp Med Biol 286:19–32. https://doi.org/10.1007/978-1-4684-5913-5_3
Acierno LJ, Worrell LT (2004) Albrecht Fleckenstein: father of calcium antagonism. Clin Cardiol 27:710–711. https://doi.org/10.1002/clc.4960271213
Fleckenstein A (1983) History of calcium antagonists. Circ Res 52:I3-16
Xiang Z, Thompson AD, Brogan JT, Schulte ML, Melancon BJ, Mi D, Lewis LM, Zou B, Yang L, Morrison R, Santomango T, Byers F, Brewer K, Aldrich JS, Yu H, Dawson ES, Li M, McManus O, Jones CK, Daniels JS, Hopkins CR, Xie XS, Conn PJ, Weaver CD, Lindsley CW (2011) The discovery and characterization of ML218: a novel, centrally active T-type calcium channel inhibitor with robust effects in STN neurons and in a rodent model of Parkinson’s disease. ACS Chem Neurosci 2:730–742. https://doi.org/10.1021/cn200090z
Hashitani H, van Helden DF, Suzuki H (1996) Properties of spontaneous depolarizations in circular smooth muscle cells of rabbit urethra. Br J Pharmacol 118:1627–1632
Nilsson H, Aalkjaer C (2003) Vasomotion: mechanisms and physiological importance. Mol Interv 3:79–89
Van Helden DF (1993) Pacemaker potentials in lymphatic smooth muscle of the guinea-pig mesentery. J Physiol 471:465–479
van Helden DF, Imtiaz MS, Nurgaliyeva K, von der Weid P, Dosen PJ (2000) Role of calcium stores and membrane voltage in the generation of slow wave action potentials in guinea-pig gastric pylorus. J Physiol 524:245–265
Seidler NW, Jona I, Vegh M, Martonosi A (1989) Cyclopiazonic acid is a specific inhibitor of the Ca2+-ATPase of sarcoplasmic reticulum. J Biol Chem 264:17816–17823
Drumm BT, Large RJ, Hollywood MA, Thornbury KD, Baker SA, Harvey BJ, McHale NG, Sergeant GP (2015) The role of Ca(2+) influx in spontaneous Ca(2+) wave propagation in interstitial cells of Cajal from the rabbit urethra. J Physiol 593:3333–3350. https://doi.org/10.1113/JP270883
Parker I, Ivorra I (1991) Caffeine inhibits inositol trisphosphate-mediated liberation of intracellular calcium in Xenopus oocytes. J Physiol 433:229–240. https://doi.org/10.1113/jphysiol.1991.sp018423
Maruyama T, Kanaji T, Nakade S, Kanno T, Mikoshiba K (1997) 2APB, 2-aminoethoxydiphenyl borate, a membrane-penetrable modulator of Ins(1,4,5)P3-induced Ca2+ release. J Biochem 122:498–505. https://doi.org/10.1093/oxfordjournals.jbchem.a021780
Huang F, Rock JR, Harfe BD, Cheng T, Huang X, Jan YN, Jan LY (2009) Studies on expression and function of the TMEM16A calcium-activated chloride channel. Proc Natl Acad Sci U S A 106:21413–21418. https://doi.org/10.1073/pnas.0911935106
Sanders KM, Zhu MH, Britton F, Koh SD, Ward SM (2012) Anoctamins and gastrointestinal smooth muscle excitability. Exp Physiol 97:200–206. https://doi.org/10.1113/expphysiol.2011.058248
da Gao Y, Zhang BL, Leung MC, Au SC, Wong PY, Shum WW (2016) Coupling of TRPV6 and TMEM16A in epithelial principal cells of the rat epididymis. J Gen Physiol 148:161–182. https://doi.org/10.1085/jgp.201611626
Evans RJ, Surprenant A (1992) Vasoconstriction of guinea-pig submucosal arterioles following sympathetic nerve stimulation is mediated by the release of ATP. Br J Pharmacol 106:242–249. https://doi.org/10.1111/j.1476-5381.1992.tb14323.x
Venkova K, Krier J (1993) Stimulation of lumbar sympathetic nerves evokes contractions of cat colon circular muscle mediated by ATP and noradrenaline. Br J Pharmacol 110:1260–1270. https://doi.org/10.1111/j.1476-5381.1993.tb13951.x
Ventura S, Pennefather JN (1991) Sympathetic co-transmission to the cauda epididymis of the rat: characterization of postjunctional adrenoceptors and purinoceptors. Br J Pharmacol 102:540–544. https://doi.org/10.1111/j.1476-5381.1991.tb12207.x
Wiszniewska B, Kurzawa R, Ciechanowicz A, Machalinski B (1997) Inducible nitric oxide synthase in the epithelial epididymal cells of the rat. Reprod Fertil Dev 9:789–794. https://doi.org/10.1071/r97063
von der Weid PY, Zhao J, Van Helden DF (2001) Nitric oxide decreases pacemaker activity in lymphatic vessels of guinea pig mesentery. Am J Physiol Heart Circ Physiol 280:H2707-2716. https://doi.org/10.1152/ajpheart.2001.280.6.H2707
Zhang H, Walcott GP, Rogers JM (2018) Effects of gadolinium on cardiac mechanosensitivity in whole isolated swine hearts. Sci Rep 8:10506. https://doi.org/10.1038/s41598-018-28743-w
von der Weid P-Y (2019) Lymphatic vessel pumping. In: Hashitani H, Lang RJ (eds) Smooth muscle spontaneous activity, vol 1. Advances in experimental medicine and biology. Springer, Singapore, pp 357–377
Warner TD, Giuliano F, Vojnovic I, Bukasa A, Mitchell JA, Vane JR (1999) Nonsteroid drug selectivities for cyclo-oxygenase-1 rather than cyclo-oxygenase-2 are associated with human gastrointestinal toxicity: a full in vitro analysis. Proc Natl Acad Sci U S A 96:7563–7568. https://doi.org/10.1073/pnas.96.13.7563
Liu LW, Thuneberg L, Huizinga JD (1995) Cyclopiazonic acid, inhibiting the endoplasmic reticulum calcium pump, reduces the canine colonic pacemaker frequency. J Pharmacol Exp Ther 275:1058–1068
Caputo A, Caci E, Ferrera L, Pedemonte N, Barsanti C, Sondo E, Pfeffer U, Ravazzolo R, Zegarra-Moran O, Galietta LJ (2008) TMEM16A, a membrane protein associated with calcium-dependent chloride channel activity. Science 322:590–594. https://doi.org/10.1126/science.1163518
Schroeder BC, Cheng T, Jan YN, Jan LY (2008) Expression cloning of TMEM16A as a calcium-activated chloride channel subunit. Cell 134:1019–1029. https://doi.org/10.1016/j.cell.2008.09.003
Yang YD, Cho H, Koo JY, Tak MH, Cho Y, Shim WS, Park SP, Lee J, Lee B, Kim BM, Raouf R, Shin YK, Oh U (2008) TMEM16A confers receptor-activated calcium-dependent chloride conductance. Nature 455:1210–1215. https://doi.org/10.1038/nature07313
Gomez-Pinilla PJ, Gibbons SJ, Bardsley MR, Lorincz A, Pozo MJ, Pasricha PJ, Van de Rijn M, West RB, Sarr MG, Kendrick ML, Cima RR, Dozois EJ, Larson DW, Ordog T, Farrugia G (2009) Ano1 is a selective marker of interstitial cells of Cajal in the human and mouse gastrointestinal tract. Am J Physiol Gastrointest Liver Physiol 296:G1370-1381. https://doi.org/10.1152/ajpgi.00074.2009
Hwang SJ, Blair PJ, Britton FC, O’Driscoll KE, Hennig G, Bayguinov YR, Rock JR, Harfe BD, Sanders KM, Ward SM (2009) Expression of anoctamin 1/TMEM16A by interstitial cells of Cajal is fundamental for slow wave activity in gastrointestinal muscles. J Physiol 587:4887–4904. https://doi.org/10.1113/jphysiol.2009.176198
Zawieja SD, Castorena JA, Gui P, Li M, Bulley SA, Jaggar JH, Rock JR, Davis MJ (2019) Ano1 mediates pressure-sensitive contraction frequency changes in mouse lymphatic collecting vessels. J Gen Physiol 151:532–554. https://doi.org/10.1085/jgp.201812294
Matchkov VV, Aalkjaer C, Nilsson H (2004) A cyclic GMP-dependent calcium-activated chloride current in smooth-muscle cells from rat mesenteric resistance arteries. J Gen Physiol 123:121–134. https://doi.org/10.1085/jgp.200308972
Ganapathi SB, Wei SG, Zaremba A, Lamb FS, Shears SB (2013) Functional regulation of ClC-3 in the migration of vascular smooth muscle cells. Hypertension 61:174–179. https://doi.org/10.1161/HYPERTENSIONAHA.112.194209
Yamada S, Suzuki Y, Bernotiene E, Giles WR, Imaizumi Y, Yamamura H (2021) Swelling-activated ClC-3 activity regulates prostaglandin E2 release in human OUMS-27 chondrocytes. Biochem Biophys Res Commun 537:29–35. https://doi.org/10.1016/j.bbrc.2020.12.068
Strogatz SH, Stewart I (1993) Coupled oscillators and biological synchronization. Scientific Am 269:102–109
Imtiaz MS, Zhao J, Hosaka K, von der Weid PY, Crowe M, van Helden DF (2007) Pacemaking through Ca2+ stores interacting as coupled oscillators via membrane depolarization. Biophys J 92:3843–3861
van Helden DF, Imtiaz MS (2003) Ca2+ phase waves: a basis for cellular pacemaking and long-range synchronicity in the guinea-pig gastric pylorus. J Physiol 548(1):271–296
Jaakkola UM, Talo A (1983) Movements of the luminal contents in two different regions of the caput epididymidis of the rat in vitro. J Physiol 336:453–463
Chaturapanich G, Maythaarttaphong S, Verawatnapakul V, Pholpramool C (2002) Mediation of contraction in rat cauda epididymidis by alpha-adrenoceptors. Reproduction 124:887–892
Pacini ESA, Castilho ACS, Hebeler-Barbosa F, Pupo AS, Kiguti LRA (2018) Contraction of rat cauda epididymis smooth muscle to alpha1-adrenoceptor activation is mediated by alpha1A-adrenoceptors. J Pharmacol Exp Ther 366:21–28. https://doi.org/10.1124/jpet.117.246710
Queiroz DB, Mendes FR, Porto CS, Avellar MC (2002) Alpha1-adrenoceptor subtypes in rat epididymis and the effects of sexual maturation. Biol Reprod 66:508–515. https://doi.org/10.1095/biolreprod66.2.508
da Silva Junior ED, de Souza BP, Vilela VV, Rodrigues JQ, Nichi M, de Agostini Losano JD, Dalmazzo A, Barnabe VH, Jurkiewicz A, Jurkiewicz NH (2014) Epididymal contraction and sperm parameters are affected by clonidine. Andrology 2:955–966. https://doi.org/10.1111/andr.283
Ventura S, Pennefather JN (1994) Alpha 2-adrenoceptor binding sites vary along the length of the male reproductive tract: a possible basis for the regional variation in response to field stimulation. Eur J Pharmacol 254:167–173. https://doi.org/10.1016/0014-2999(94)90384-0
Gesek FA (1996) Alpha 2-adrenergic receptors activate phospholipase C in renal epithelial cells. Mol Pharmacol 50:407–414
Haynes JM, Hill SJ (1996) Alpha-adrenoceptor mediated responses of the cauda epididymis of the guinea-pig. Br J Pharmacol 119:1203–1210. https://doi.org/10.1111/j.1476-5381.1996.tb16023.x
Berridge MJ, Irvine RF (1984) Inositol trisphosphate, a novel second messenger in cellular signal transduction. Nature 312:315–321. https://doi.org/10.1038/312315a0
Funding
The present study was partly supported by Grant-in-Aid for Scientific Research (C) (No. 19K08426) from Japan Society for Promotion of the Science (JSPS) to R.M.
Author information
Authors and Affiliations
Contributions
Conceptualization: HH, DVH; methodology: HH, RL, RM, DVH; formal analysis and investigation: HH, RM, DVH; writing—review and editing: HH, RL, RM, DVH.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethics approval
Experiments were carried out in accordance with the Care and Use of Animals in the Field of Physiological Sciences set out by the Physiological Society of Japan (2015).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mitsui, R., Hashitani, H., Lang, R.J. et al. Mechanisms underlying spontaneous phasic contractions and sympathetic control of smooth muscle in the rat caudal epididymis. Pflugers Arch - Eur J Physiol 473, 1925–1938 (2021). https://doi.org/10.1007/s00424-021-02609-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-021-02609-z