Abstract
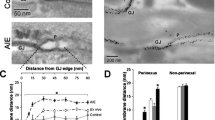
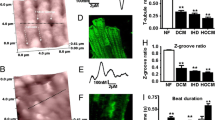
It was recently demonstrated that cardiac sodium channels (Nav1.5) localized at the perinexus, an intercalated disc (ID) nanodomain associated with gap junctions (GJ), may contribute to electrical coupling between cardiac myocytes via an ephaptic mechanism. Impairment of ephaptic coupling by acute interstitial edema (AIE)-induced swelling of the perinexus was associated with arrhythmogenic, anisotropic conduction slowing. Given that Kir2.1 has also recently been reported to localize at intercalated discs, we hypothesized that Kir2.1 channels may reside within the perinexus and that inhibiting them may mitigate arrhythmogenic conduction slowing observed during AIE. Using gated stimulated emission depletion (gSTED) and stochastic optical reconstruction microscopy (STORM) super-resolution microscopy, we indeed find that a significant proportion of Kir2.1 channels resides within the perinexus. Moreover, whereas Nav1.5 inhibition during AIE exacerbated arrhythmogenic conduction slowing, inhibiting Kir2.1 channels during AIE preferentially increased transverse conduction velocity—decreasing anisotropy and ameliorating arrhythmia risk compared to AIE alone. Comparison of our results with a nanodomain computer model identified enrichment of both Nav1.5 and Kir2.1 at intercalated discs as key factors underlying the experimental observations. We demonstrate that Kir2.1 channels are localized within the perinexus alongside Nav1.5 channels. Further, targeting Kir2.1 modulates intercellular coupling between cardiac myocytes, anisotropy of conduction, and arrhythmia propensity in a manner consistent with a role for ephaptic coupling in cardiac conduction. For over half a century, electrical excitation in the heart has been thought to occur exclusively via gap junction-mediated ionic current flow between cells. Further, excitation was thought to depend almost exclusively on sodium channels with potassium channels being involved mainly in returning the cell to rest. Here, we demonstrate that sodium and potassium channels co-reside within nanoscale domains at cell-to-cell contact sites. Experimental and computer modeling results suggest a role for these channels in electrical coupling between cardiac muscle cells via an ephaptic mechanism working in tandem with gap junctions. This new insight into the mechanism of cardiac electrical excitation could pave the way for novel therapies against cardiac rhythm disturbances.








Similar content being viewed by others
References
Agullo-Pascual E, Lin X, Leo-Macias A, Zhang M, Liang FX, Li Z, Pfenniger A, Lubkemeier I, Keegan S, Fenyo D, Willecke K, Rothenberg E, and Delmar M. Super-resolution imaging reveals that loss of the C-terminus of connexin43 limits microtubule plus-end capture and NaV1.5 localization at the intercalated disc. Cardiovascular research 2014.
Clausen Mathias P, Galiani S, de la Serna Jorge B, Fritzsche M, Chojnacki J, Gehmlich K, Lagerholm BC, and Eggeling C. Pathways to optical STED microscopy. In: NanoBioImaging 2013, p. 1.
Dhein S, Seidel T, Salameh A, Jozwiak J, Hagen A, Kostelka M, Hindricks G, Mohr FW (2014) Remodeling of cardiac passive electrical properties and susceptibility to ventricular and atrial arrhythmias. Front Physiol 5:424
Ferreira G, Yi J, Rios E, Shirokov R (1997) Ion-dependent inactivation of barium current through L-type calcium channels. J Gen Physiol 109:449–461
Geisler SB, Green KJ, Isom LL, Meshinchi S, Martens JR, Delmar M, Russell MW (2010) Ordered assembly of the adhesive and electrochemical connections within newly formed intercalated disks in primary cultures of adult rat cardiomyocytes. J Biomed Biotechnol 2010:624719
George SA, Sciuto KJ, Lin J, Salama ME, Keener JP, Gourdie RG, and Poelzing S. Extracellular sodium and potassium levels modulate cardiac conduction in mice heterozygous null for the Connexin43 gene. Pflugers Arch 2015.
Girouard SD, Laurita KR, Rosenbaum DS (1996) Unique properties of cardiac action potentials recorded with voltage-sensitive dyes. J Cardiovasc Electrophysiol 7:1024–1038
Green CR, Severs NJ (1984) Gap junction connexon configuration in rapidly frozen myocardium and isolated intercalated disks. The Journal of cell biology 99:453–463
Hong M, Bao L, Kefaloyianni E, Agullo-Pascual E, Chkourko H, Foster M, Taskin E, Zhandre M, Reid DA, Rothenberg E, Delmar M, Coetzee WA (2012) Heterogeneity of ATP-sensitive K+ channels in cardiac myocytes: enrichment at the intercalated disk. J Biol Chem 287:41258–41267
Hoyt RH, Cohen ML, Saffitz JE (1989) Distribution and three-dimensional structure of intercellular junctions in canine myocardium. Circ Res 64:563–574
Kleber AG, Rudy Y (2004) Basic mechanisms of cardiac impulse propagation and associated arrhythmias. Physiol Rev 84:431–488
Kucera JP, Kleber AG, Rohr S (1998) Slow conduction in cardiac tissue. II: effects of branching tissue geometry Circulation research 83:795–805
Kucera JP, Rohr S, Rudy Y (2002) Localization of sodium channels in intercalated disks modulates cardiac conduction. Circ Res 91:1176–1182
Larsen AP, Olesen SP, Grunnet M, Poelzing S (2010) Pharmacological activation of IKr impairs conduction in guinea pig hearts. J Cardiovasc Electrophysiol 21:923–929
Leo-Macias A, Liang FX, Delmar M (2015) Ultrastructure of the intercellular space in adult murine ventricle revealed by quantitative tomographic electron microscopy. Cardiovasc Res 107:442–452
Lin J, Keener JP (2013) Ephaptic coupling in cardiac myocytes. IEEE Trans Biomed Eng 60:576–582
Lin J, Keener JP (2014) Microdomain effects on transverse cardiac propagation. Biophys J 106:925–931
Matthes S, Zhang M, Taffet S, Delmar M (2010) Desmosomes and gap junctions in epicardium-derived cells a possible role in arrhythmogenic right ventricular cardiomyopathy? Heart Rhythm 7:1715–1716
Mays DJ, Foose JM, Philipson LH, Tamkun MM (1995) Localization of the Kv15 K+ channel protein in explanted cardiac tissue. J Clin Invest 96:282–292
Milstein ML, Musa H, Balbuena DP, Anumonwo JM, Auerbach DS, Furspan PB, Hou L, Hu B, Schumacher SM, Vaidyanathan R, Martens JR, Jalife J (2012) Dynamic reciprocity of sodium and potassium channel expression in a macromolecular complex controls cardiac excitability and arrhythmia. Proc Natl Acad Sci U S A 109:E2134–E2143
Mori Y, Fishman GI, Peskin CS (2008) Ephaptic conduction in a cardiac strand model with 3D electrodiffusion. Proc Natl Acad Sci U S A 105:6463–6468
Poelzing S, Veeraraghavan R (2007) Heterogeneous ventricular chamber response to hypokalemia and inward rectifier potassium channel blockade underlies bifurcated T wave in guinea pig. Am J Phys Heart Circ Phys 292:H3043–H3051
Rhett JM, Jourdan J, Gourdie RG (2011) Connexin 43 connexon to gap junction transition is regulated by zonula occludens-1. Mol Biol Cell 22:1516–1528
Rhett JM, Ongstad EL, Jourdan J, Gourdie RG (2012) Cx43 associates with Na(v)15 in the cardiomyocyte perinexus. J Membr Biol 245:411–422
Schoonderwoert V, Dijkstra R, Luckinavicius G, Kobler O, der Voort H Huygens STED v (2013) Deconvolution increases signal-to-noise and image resolution towards 22 nm. Micros Today 21:38–44
Skibsbye L, Wang X, Axelsen LN, Bomholtz SH, Nielsen MS, Grunnet M, Bentzen BH, Jespersen T (2015) Antiarrhythmic mechanisms of SK channel inhibition in the rat atrium. J Cardiovasc Pharmacol 66:165–176
Slawsky MT, Castle NA (1994) K+ channel blocking actions of flecainide compared with those of propafenone and quinidine in adult rat ventricular myocytes. J Pharmacol Exp Ther 269:66–74
Sperelakis N, McConnell K (2002) Electric field interactions between closely abutting excitable cells. IEEE Eng Med Biol Mag 21:77–89
Stein M, Boulaksil M, Engelen MA, van Veen TA, Hauer RN, de Bakker JM, van Rijen HV (2006) Conduction reserve and arrhythmias. Neth Heart J 14:113–116
Stein M, van Veen TA, Remme CA, Boulaksil M, Noorman M, van Stuijvenberg L, van der Nagel R, Bezzina CR, Hauer RN, de Bakker JM, van Rijen HV (2009) Combined reduction of intercellular coupling and membrane excitability differentially affects transverse and longitudinal cardiac conduction. Cardiovasc Res 83:52–60
Tran TN, Drab K, Daszykowski M (2013) Revised DBSCAN algorithm to cluster data with dense adjacent clusters. Chemom Intell Lab Syst 120:92–96
Veeraraghavan R, and Gourdie R Stochastic optical reconstruction microscopy-based relative localization analysis (STORM-RLA) for quantitative nanoscale assessment of spatial protein organization Mol Biol Cell 2016
Veeraraghavan R, Larsen AP, Torres NS, Grunnet M, Poelzing S (2013) Potassium channel activators differentially modulate the effect of sodium channel blockade on cardiac conduction. Acta Physiol (Oxf) 207:280–289
Veeraraghavan R, Lin J, Hoeker GS, Keener JP, Gourdie RG, and Poelzing S Sodium channels in the Cx43 gap junction perinexus may constitute a cardiac ephapse: an experimental and modeling study Pflugers Arch 2015
Veeraraghavan R, Poelzing S (2008) Mechanisms underlying increased right ventricular conduction sensitivity to flecainide challenge. Cardiovasc Res 77:749–756
Veeraraghavan R, Salama ME, and Poelzing S Interstitial volume modulates the conduction velocity—gap junction relationship American journal of physiology Heart and circulatory physiology 2012
Acknowledgments
The authors would like to thank Dr. James Smyth for the assistance with the STORM microscopy. Thanks also to Dr. Gregory Hoeker and Michael Entz for the assistance with optical mapping experiments and to Mrs. Jane Jourdan for the assistance with NRVM experiments.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The investigation was conducted in conformation with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication no. 85-23, revised 1996). All animal study protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at the Virginia Polytechnic University.
Conflicts of interest
The authors declare that they have no conflict of interest.
Funding sources
This work was supported by an NIH R01 grant awarded to Dr. Poelzing (R01-HL102298-01A1), by an NIH R01 grant awarded to Dr. Gourdie (RO1 HL56728-14A2), and by an American Heart Association post-doctoral fellowship awarded to Dr. Veeraraghavan (2013–15, completed).
Disclosures
None
Rights and permissions
About this article
Cite this article
Veeraraghavan, R., Lin, J., Keener, J.P. et al. Potassium channels in the Cx43 gap junction perinexus modulate ephaptic coupling: an experimental and modeling study. Pflugers Arch - Eur J Physiol 468, 1651–1661 (2016). https://doi.org/10.1007/s00424-016-1861-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-016-1861-2