Abstract
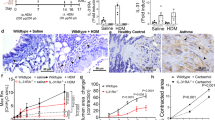
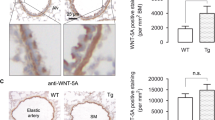
Airway smooth muscle (ASM) is a key target cell in allergen-induced asthma known to contribute to airway hyperresponsiveness (AHR) and chronic airway remodeling. Changes in ASM calcium homeostasis have been shown to contribute to AHR although the mechanisms and Ca2+ signal effectors are incompletely understood. In the present study, we tested the function of ASM multifunctional protein kinase Ca2+/calmodulin-dependent kinase II (CaMKII) isoforms CaMKIIδ and CaMKIIγ in allergen-induced AHR and airway remodeling in vivo. Using a murine model of atopic asthma, we demonstrate that CaMKIIδ protein is upregulated in ASM derived from ovalbumin (OVA)-treated animals compared to controls. A genetic approach to conditionally knock out smooth muscle CaMKIIδ and CaMKIIγ in separate Cre-loxp systems was validated, and using this loss-of-function approach, the function of these CaMKII isoforms was tested in ovalbumin (OVA)-induced airway remodeling and AHR. OVA treatment in control mice had no effect on ASM remodeling in this model of AHR, and CaMKIIδ knockouts had no independent effects on ASM content. However, at 1 day post-final OVA challenge, OVA-induced AHR was eliminated in the CaMKIIδ knockouts. OVA-induced peribronchial inflammation and bronchoalveolar lavage fluid (BALF) levels of the Th2 cytokine IL-13 were significantly decreased in the CaMKIIδ knockouts. Unexpectedly, we found increased peribronchial eosinophils in the smooth muscle CaMKIIδ knockouts compared to control animals at 1 day post-final challenge, suggesting that lack of ASM CaMKIIδ delays the progression of AHR rather than inhibiting it. Indeed, when AHR was determined at 7 days post-final OVA challenge, CaMKIIδ knockouts showed robust AHR while AHR was fully resolved in OVA-challenged control mice. These in vivo studies demonstrate a role for smooth muscle CaMKIIδ in promoting airway inflammation and AHR and suggest a complex signaling role for CaMKIIδ in regulating ASM function. These studies confirm the diverse roles of ASM cells as immune effectors that control AHR and call for further studies into CaMKIIδ-mediated signaling in ASM cells during disease.








Similar content being viewed by others
References
Ammit AJ, Moir LM, Oliver BG, Hughes JM, Alkhouri H, Ge Q, Burgess JK, Black JL, Roth M (2007) Effect of IL-6 trans-signaling on the pro-remodeling phenotype of airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 292:L199–L206
Amrani Y, Lazaar AL, Hoffman R, Amin K, Ousmer S, Panettieri RA Jr (2000) Activation of p55 tumor necrosis factor-alpha receptor-1 coupled to tumor necrosis factor receptor-associated factor 2 stimulates intercellular adhesion molecule-1 expression by modulating a thapsigargin-sensitive pathway in human tracheal smooth muscle cells. Mol Pharmacol 58:237–245
Amrani Y, Panettieri RA (2003) Airway smooth muscle: contraction and beyond. Int J Biochem Cell Biol 35:272–276
An SS, Bai TR, Bates JH, Black JL, Brown RH, Brusasco V, Chitano P, Deng L, Dowell M, Eidelman DH, Fabry B et al (2007) Airway smooth muscle dynamics: a common pathway of airway obstruction in asthma. Eur Respir J 29:834–860
Backs J, Backs T, Neef S, Kreusser MM, Lehmann LH, Patrick DM, Grueter CE, Qi X, Richardson JA, Hill JA, Katus HA et al (2009) The delta isoform of CaM kinase II is required for pathological cardiac hypertrophy and remodeling after pressure overload. Proc Natl Acad Sci U S A 106:2342–2347
Backs J, Stein P, Backs T, Duncan FE, Grueter CE, McAnally J, Qi X, Schultz RM, Olson EN (2010) The gamma isoform of CaM kinase II controls mouse egg activation by regulating cell cycle resumption. Proc Natl Acad Sci U S A 107:81–86
Berkman N, Krishnan VL, Gilbey T, Newton R, O’Connor B, Barnes PJ, Chung KF (1996) Expression of RANTES mRNA and protein in airways of patients with mild asthma. Am J Respir Crit Care Med 154:1804–1811
Camoretti-Mercado B, Forsythe SM, LeBeau MM, Espinosa R III, Vieira JE, Halayko AJ, Willadsen S, Kurtz B, Ober C, Evans GA, Thweatt R et al (1998) Expression and cytogenetic localization of the human SM22 gene (TAGLN). Genomics 49:452–457
Catley MC, Sukkar MB, Chung KF, Jaffee B, Liao SM, Coyle AJ, Haddad E, Barnes PJ, Newton R (2006) Validation of the anti-inflammatory properties of small-molecule IkappaB Kinase (IKK)-2 inhibitors by comparison with adenoviral-mediated delivery of dominant-negative IKK1 and IKK2 in human airways smooth muscle. Mol Pharmacol 70:697–705
Cazzola M, Page CP, Rogliani P, Matera MG (2013) beta2-agonist therapy in lung disease. Am J Respir Crit Care Med 187:690–696
Cox G, Thomson NC, Rubin AS, Niven RM, Corris PA, Siersted HC, Olivenstein R, Pavord ID, McCormack D, Chaudhuri R, Miller JD et al (2007) Asthma control during the year after bronchial thermoplasty. N Engl J Med 356:1327–1337
Del Prete GF, De CM, D’Elios MM, Maestrelli P, Ricci M, Fabbri L, Romagnani S (1993) Allergen exposure induces the activation of allergen-specific Th2 cells in the airway mucosa of patients with allergic respiratory disorders. Eur J Immunol 23:1445–1449
Doherty TA, Soroosh P, Broide DH, Croft M (2009) CD4+ cells are required for chronic eosinophilic lung inflammation but not airway remodeling. Am J Physiol Lung Cell Mol Physiol 296:L229–L235
Elias JA, Wu Y, Zheng T, Panettieri R (1997) Cytokine- and virus-stimulated airway smooth muscle cells produce IL-11 and other IL-6-type cytokines. Am J Physiol 273:L648–L655
Erjefalt JS, Persson CG (2000) New aspects of degranulation and fates of airway mucosal eosinophils. Am J Respir Crit Care Med 161:2074–2085
Frischauf I, Schindl R, Derler I, Bergsmann J, Fahrner M, Romanin C (2008) The STIM/Orai coupling machinery. Channels (Austin) 2:261–268
Gunst SJ, Panettieri RA Jr (2012) Last Word on Point: alterations in airway smooth muscle phenotype do cause airway hyperresponsiveness in asthma. J Appl Physiol 113:847
Hakonarson H, Maskeri N, Carter C, Grunstein MM (1999) Regulation of TH1- and TH2-type cytokine expression and action in atopic asthmatic sensitized airway smooth muscle. J Clin Invest 103:1077–1087
Halayko AJ, Solway J (2001) Molecular mechanisms of phenotypic plasticity in smooth muscle cells. J Appl Physiol 90:358–368
House SJ, Ginnan RG, Armstrong SE, Singer HA (2007) Calcium/calmodulin-dependent protein kinase II-delta isoform regulation of vascular smooth muscle cell proliferation. Am J Physiol Cell Physiol 292:C2276–C2287
House SJ, Singer HA (2008) CaMKII-delta isoform regulation of neointima formation after vascular injury. Arterioscler Thromb Vasc Biol 28:441–447
Hudmon A, Schulman H (2002) Structure-function of the multifunctional Ca2+/calmodulin-dependent protein kinase II. Biochem J 364:593–611
Jarai G, Sukkar M, Garrett S, Duroudier N, Westwick J, Adcock I, Chung KF (2004) Effects of interleukin-1beta, interleukin-13 and transforming growth factor-beta on gene expression in human airway smooth muscle using gene microarrays. Eur J Pharmacol 497:255–265
Kips JC (2003) The relation between morphologic and functional airway changes in bronchial asthma. Verh K Acad Geneeskd Belg 65:247–265
Lamkhioued B, Renzi PM, Bi-Younes S, Garcia-Zepada EA, Allakhverdi Z, Ghaffar O, Rothenberg MD, Luster AD, Hamid Q (1997) Increased expression of eotaxin in bronchoalveolar lavage and airways of asthmatics contributes to the chemotaxis of eosinophils to the site of inflammation. J Immunol 159:4593–4601
Lepore JJ, Cheng L, Min LM, Mericko PA, Morrisey EE, Parmacek MS (2005) High-efficiency somatic mutagenesis in smooth muscle cells and cardiac myocytes in SM22alpha-Cre transgenic mice. Genesis 41:179–184
Li W, Li H, Sanders PN, Mohler PJ, Backs J, Olson EN, Anderson ME, Grumbach IM (2011) The multifunctional Ca2+/calmodulin-dependent kinase II delta (CaMKIIdelta) controls neointima formation after carotid ligation and vascular smooth muscle cell proliferation through cell cycle regulation by p21. J Biol Chem 286:7990–7999
Li L, Miano JM, Mercer B, Olson EN (1996) Expression of the SM22alpha promoter in transgenic mice provides evidence for distinct transcriptional regulatory programs in vascular and visceral smooth muscle cells. J Cell Biol 132:849–859
Ling H, Gray CB, Zambon AC, Grimm M, Gu Y, Dalton N, Purcell NH, Peterson K, Brown JH (2013) Ca2+/Calmodulin-dependent protein kinase II delta mediates myocardial ischemia/reperfusion injury through nuclear factor-kappaB. Circ Res 112:935–944
Luo SF, Chang CC, Lee IT, Lee CW, Lin WN, Lin CC, Yang CM (2009) Activation of ROS/NF-kappaB and Ca2+/CaM kinase II are necessary for VCAM-1 induction in IL-1beta-treated human tracheal smooth muscle cells. Toxicol Appl Pharmacol 237:8–21
Mahn K, Hirst SJ, Ying S, Holt MR, Lavender P, Ojo OO, Siew L, Simcock DE, McVicker CG, Kanabar V, Snetkov VA et al (2009) Diminished sarco/endoplasmic reticulum Ca2+ ATPase (SERCA) expression contributes to airway remodelling in bronchial asthma. Proc Natl Acad Sci U S A 106:10775–10780
Malm-Erjefalt M, Greiff L, Ankerst J, Andersson M, Wallengren J, Cardell LO, Rak S, Persson CG, Erjefalt JS (2005) Circulating eosinophils in asthma, allergic rhinitis, and atopic dermatitis lack morphological signs of degranulation. Clin Exp Allergy 35:1334–1340
Mercure MZ, Ginnan R, Singer HA (2008) CaM kinase II delta2-dependent regulation of vascular smooth muscle cell polarization and migration. Am J Physiol Cell Physiol 294:C1465–C1475
Moessler H, Mericskay M, Li Z, Nagl S, Paulin D, Small JV (1996) The SM 22 promoter directs tissue-specific expression in arterial but not in venous or visceral smooth muscle cells in transgenic mice. Development 122:2415–2425
Nials AT, Uddin S (2008) Mouse models of allergic asthma: acute and chronic allergen challenge. Dis Model Mech 1:213–220
Odaka M, Matsukura S, Kuga H, Kokubu F, Kasama T, Kurokawa M, Kawaguchi M, Ieki K, Suzuki S, Watanabe S, Homma T et al (2007) Differential regulation of chemokine expression by Th1 and Th2 cytokines and mechanisms of eotaxin/CCL-11 expression in human airway smooth muscle cells. Int Arch Allergy Immunol 143(Suppl 1):84–88
Owens GK (1998) Molecular control of vascular smooth muscle cell differentiation. Acta Physiol Scand 164:623–635
Owens GK, Kumar MS, Wamhoff BR (2004) Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol Rev 84:767–801
Pare PD, Mitzner W (2012) Last Word on Counterpoint: alterations in airway smooth muscle phenotype do not cause airway hyperresponsiveness in asthma. J Appl Physiol 113:848
Pepe C, Foley S, Shannon J, Lemiere C, Olivenstein R, Ernst P, Ludwig MS, Martin JG, Hamid Q (2005) Differences in airway remodeling between subjects with severe and moderate asthma. J Allergy Clin Immunol 116:544–549
Prefontaine D, Lajoie-Kadoch S, Foley S, Audusseau S, Olivenstein R, Halayko AJ, Lemiere C, Martin JG, Hamid Q (2009) Increased expression of IL-33 in severe asthma: evidence of expression by airway smooth muscle cells. J Immunol 183:5094–5103
Ramos-Barbon D, Fraga-Iriso R, Brienza NS, Montero-Martinez C, Verea-Hernando H, Olivenstein R, Lemiere C, Ernst P, Hamid QA, Martin JG (2010) T Cells localize with proliferating smooth muscle alpha-actin + cell compartments in asthma. Am J Respir Crit Care Med 182:317–324
Robins S, Roussel L, Schachter A, Risse PA, Mogas AK, Olivenstein R, Martin JG, Hamid Q, Rousseau S (2011) Steroid-insensitive ERK1/2 activity drives CXCL8 synthesis and neutrophilia by airway smooth muscle. Am J Respir Cell Mol Biol 45:984–990
Sanders PN, Koval OM, Jaffer OA, Prasad AM, Businga TR, Scott JA, Hayden PJ, Luczak ED, Dickey DD, Allamargot C, Olivier AK et al (2013) CaMKII is essential for the proasthmatic effects of oxidation. Sci Transl Med 5:195ra97
Saunders MA, Mitchell JA, Seldon PM, Yacoub MH, Barnes PJ, Giembycz MA, Belvisi MG (1997) Release of granulocyte-macrophage colony stimulating factor by human cultured airway smooth muscle cells: suppression by dexamethasone. Br J Pharmacol 120:545–546
Schmitz J, Owyang A, Oldham E, Song Y, Murphy E, McClanahan TK, Zurawski G, Moshrefi M, Qin J, Li X, Gorman DM et al (2005) IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23:479–490
Schramm CM, Puddington L, Wu C, Guernsey L, Gharaee-Kermani M, Phan SH, Thrall RS (2004) Chronic inhaled ovalbumin exposure induces antigen-dependent but not antigen-specific inhalational tolerance in a murine model of allergic airway disease. Am J Pathol 164:295–304
Schworer CM, Rothblum LI, Thekkumkara TJ, Singer HA (1993) Identification of novel isoforms of the delta subunit of Ca2+/calmodulin-dependent protein kinase II. Differential expression in rat brain and aorta. J Biol Chem 268:14443–14449
Shan L, Redhu NS, Saleh A, Halayko AJ, Chakir J, Gounni AS (2010) Thymic stromal lymphopoietin receptor-mediated IL-6 and CC/CXC chemokines expression in human airway smooth muscle cells: role of MAPKs (ERK1/2, p38, and JNK) and STAT3 pathways. J Immunol 184:7134–7143
Shi HZ, Humbles A, Gerard C, Jin Z, Weller PF (2000) Lymph node trafficking and antigen presentation by endobronchial eosinophils. J Clin Invest 105:945–953
Spinelli AM, Gonzalez-Cobos JC, Zhang X, Motiani RK, Rowan S, Zhang W, Garrett J, Vincent PA, Matrougui K, Singer HA, Trebak M (2012) Airway smooth muscle STIM1 and Orai1 are upregulated in asthmatic mice and mediate PDGF-activated SOCE, CRAC currents, proliferation, and migration. Pflugers Arch 464:481–492
Sumi M, Kiuchi K, Ishikawa T, Ishii A, Hagiwara M, Nagatsu T, Hidaka H (1991) The newly synthesized selective Ca2+/calmodulin dependent protein kinase II inhibitor KN-93 reduces dopamine contents in PC12h cells. Biochem Biophys Res Commun 181:968–975
Tan X, Alrashdan YA, Alkhouri H, Oliver BG, Armour CL, Hughes JM (2013) Airway smooth muscle CXCR3 ligand production: regulation by JAK-STAT1 and intracellular Ca(2)(+). Am J Physiol Lung Cell Mol Physiol 304:L790–L802
Tombes RM, Faison MO, Turbeville JM (2003) Organization and evolution of multifunctional Ca(2+)/CaM-dependent protein kinase genes. Gene 322:17–31
Trian T, Benard G, Begueret H, Rossignol R, Girodet PO, Ghosh D, Ousova O, Vernejoux JM, Marthan R, Tunon-de-Lara JM, Berger P (2007) Bronchial smooth muscle remodeling involves calcium-dependent enhanced mitochondrial biogenesis in asthma. J Exp Med 204:3173–3181
Venge P (2010) The eosinophil and airway remodelling in asthma. Clin Respir J 4(Suppl 1):15–19
Walker C, Bode E, Boer L, Hansel TT, Blaser K, Virchow JC Jr (1992) Allergic and nonallergic asthmatics have distinct patterns of T-cell activation and cytokine production in peripheral blood and bronchoalveolar lavage. Am Rev Respir Dis 146:109–115
Wright DB, Trian T, Siddiqui S, Pascoe CD, Johnson JR, Dekkers BG, Dakshinamurti S, Bagchi R, Burgess JK, Kanabar V, Ojo OO (2013) Phenotype modulation of airway smooth muscle in asthma. Pulm Pharmacol Ther 26:42–49
Zuyderduyn S, Sukkar MB, Fust A, Dhaliwal S, Burgess JK (2008) Treating asthma means treating airway smooth muscle cells. Eur Respir J 32:265–274
Acknowledgments
The authors greatly acknowledge the following research support: AMC’s Candice Weir Fund, NIH grant HL097111 (to MT), NIH grant HL049426 (to HAS), and Albany Medical Center fellowship and stipend support (AMS). We would also like to acknowledge receipt of generous gifts including founder mice carry floxed CaMKIIδ and CaMKIIγ alleles which were kindly provided by Dr. Johannes Backs (Heidelberg) and Dr. Eric Olson (UTSW) and the eosinophil-specific MPB antibody, provided by Dr. Jamie Lee (Mayo Clinic Scottsdale, Arizona). We also thank Xiaolan Ding, Miao Jiang, and Diane Singer for their general animal and cell culture support, Dr. Margarida Barroso for her assistance and insight into immunohistochemical and microscopy techniques, and the Singer, Trebak, and Jourd’heuil lab for the exchange of ideas and experiences on the first floor of the ME building at AMC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Spinelli, A.M., Liu, Y., Sun, LY. et al. Smooth muscle CaMKIIδ promotes allergen-induced airway hyperresponsiveness and inflammation. Pflugers Arch - Eur J Physiol 467, 2541–2554 (2015). https://doi.org/10.1007/s00424-015-1713-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-015-1713-5