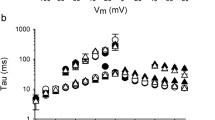
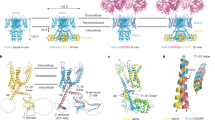
Abstract
Voltage-gated potassium (Kv) channels are tetrameric assemblies of transmembrane Kv proteins with cytosolic N- and C-termini. The N-terminal domain of Kv1 proteins binds to β-subunits, but the role of the C-terminus is less clear. Therefore, we studied the role of the C-terminus in regulating Kv1.5 channel and its interactions with Kvβ-subunits. When expressed in COS-7 cells, deletion of the C-terminal domain of Kv1.5 did not affect channel gating or kinetics. Coexpression of Kv1.5 with Kvβ3 increased current inactivation, whereas Kvβ2 caused a hyperpolarizing shift in the voltage dependence of current activation. Inclusion of NADPH in the patch pipette solution accelerated the inactivation of Kv1.5-Kvβ3 currents. In contrast, NADP+ decreased the rate and the extent of Kvβ3-induced inactivation and reversed the hyperpolarizing shift in the voltage dependence of activation induced by Kvβ2. Currents generated by Kv1.5ΔC+Kvβ3 or Kv1.5ΔC+Kvβ2 complexes did not respond to changes in intracellular pyridine nucleotide concentration, indicating that the C-terminus is required for pyridine nucleotide-dependent interactions between Kvβ and Kv1.5. A glutathione-S-transferase (GST) fusion protein containing the C-terminal peptide of Kv1.5 did not bind to apoKvβ2, but displayed higher affinity for Kvβ2:NADPH than Kvβ2:NADP+. The GST fusion protein also precipitated Kvβ proteins from mouse brain lysates. Pull-down experiments, structural analysis and electrophysiological data indicated that a specific region of the C-terminus (Arg543-Val583) is required for Kvβ binding. These results suggest that the C-terminal domain of Kv1.5 interacts with β-subunits and that this interaction is essential for the differential regulation of Kv currents by oxidized and reduced nucleotides.











Similar content being viewed by others
Abbreviations
- Kvβ:
-
β-Subunit of the voltage-gated potassium channel
- Kv:
-
Voltage-gated potassium channel
- Kv∆C:
-
Deletion of the last C-terminal amino acids of Kv channel
- GST:
-
Glutathione-S-transferase fusion protein
- V h act:
-
Voltage at which half of the channels are activated
- V h inact:
-
Voltage at which half of the channels are inactivated
References
Barski OA, Tipparaju SM, Bhatnagar A (2008) The aldo-keto reductase superfamily and its role in drug metabolism and detoxification. Drug Metab Rev 40(4):553–624
Ceconi C, Bernocchi P, Boraso A, Cargnoni A, Pepi P, Curello S, Ferrari R (2000) New insights on myocardial pyridine nucleotides and thiol redox state in ischemia and reperfusion damage. Cardiovasc Res 47(3):586–594
Cheng Y, Oldfield CJ, Meng J, Romero P, Uversky VN, Dunker AK (2007) Mining alpha-helix-forming molecular recognition features with cross species sequence alignments. Biochemistry 46(47):13468–13477
Cole C, Barber JD, Barton GJ (2008) The Jpred 3 secondary structure prediction server. Nucleic Acids Res 36(Web Server issue):W197–W201
Deal KK, England SK, Tamkun MM (1996) Molecular physiology of cardiac potassium channels. Physiol Rev 76(1):49–67
Dubois JM, Rouzaire-Dubois B (1993) Role of potassium channels in mitogenesis. Prog Biophys Mol Biol 59(1):1–21
Ekhterae D, Platoshyn O, Zhang S, Remillard CV, Yuan JX (2003) Apoptosis repressor with caspase domain inhibits cardiomyocyte apoptosis by reducing K+ currents. Am J Physiol Cell Physiol 284(6):C1405–C1410
Eldstrom J, Doerksen KW, Steele DF, Fedida D (2002) N-terminal PDZ-binding domain in Kv1 potassium channels. FEBS Lett 531(3):529–537
Gulbis JM, Zhou M, Mann S, MacKinnon R (2000) Structure of the cytoplasmic beta subunit-T1 assembly of voltage-dependent K+ channels. Science 289(5476):123–127
Houtkooper RH, Canto C, Wanders RJ, Auwerx J (2010) The secret life of NAD+: an old metabolite controlling new metabolic signaling pathways. Endocr Rev 31(2):194–223
Klaidman LK, Leung AC, Adams JD Jr (1995) High-performance liquid chromatography analysis of oxidized and reduced pyridine dinucleotides in specific brain regions. Anal Biochem 228(2):312–317
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23(21):2947–2948
Lee TE, Philipson LH, Kuznetsov A, Nelson DJ (1994) Structural determinant for assembly of mammalian K+ channels. Biophys J 66(3 Pt 1):667–673
Leicher T, Bahring R, Isbrandt D, Pongs O (1998) Coexpression of the KCNA3B gene product with Kv1.5 leads to a novel A-type potassium channel. J Biol Chem 273(52):35095–35101
Li M, Jan YN, Jan LY (1992) Specification of subunit assembly by the hydrophilic amino-terminal domain of the Shaker potassium channel. Science 257(5074):1225–1230
Liu SQ, Jin H, Zacarias A, Srivastava S, Bhatnagar A (2001) Binding of pyridine nucleotide coenzymes to the beta-subunit of the voltage-sensitive K+ channel. J Biol Chem 276(15):11812–11820
Long SB, Campbell EB, Mackinnon R (2005) Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 309(5736):897–903
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Magidovich E, Fleishman SJ, Yifrach O (2006) Intrinsically disordered C-terminal segments of voltage-activated potassium channels: a possible fishing rod-like mechanism for channel binding to scaffold proteins. Bioinformatics 22(13):1546–1550
Magidovich E, Orr I, Fass D, Abdu U, Yifrach O (2007) Intrinsic disorder in the C-terminal domain of the Shaker voltage-activated K+ channel modulates its interaction with scaffold proteins. Proc Natl Acad Sci U S A 104(32):13022–13027
McCormack T, McCormack K (1994) Shaker K+ channel beta subunits belong to an NAD(P)H-dependent oxidoreductase superfamily. Cell 79(7):1133–1135
Meszaros B, Simon I, Dosztanyi Z (2009) Prediction of protein binding regions in disordered proteins. PLoS Comput Biol 5(5):e1000376
Nagaya N, Papazian DM (1997) Potassium channel alpha and beta subunits assemble in the endoplasmic reticulum. J Biol Chem 272(5):3022–3027
Oldfield CJ, Cheng Y, Cortese MS, Romero P, Uversky VN, Dunker AK (2005) Coupled folding and binding with alpha-helix-forming molecular recognition elements. Biochemistry 44(37):12454–12470
Pan Y, Weng J, Cao Y, Bhosle RC, Zhou M (2008) Functional coupling between the Kv1.1 channel and aldoketoreductase Kvbeta1. J Biol Chem 283(13):8634–8642
Pongs O, Schwarz JR (2010) Ancillary subunits associated with voltage-dependent K+ channels. Physiol Rev 90(2):755–796
Romero P, Obradovic Z, Li X, Garner EC, Brown CJ, Dunker AK (2001) Sequence complexity of disordered protein. Proteins 42(1):38–48
Sewing S, Roeper J, Pongs O (1996) Kv beta 1 subunit binding specific for shaker-related potassium channel alpha subunits. Neuron 16(2):455–463
Shen NV, Chen X, Boyer MM, Pfaffinger PJ (1993) Deletion analysis of K+ channel assembly. Neuron 11(1):67–76
Sokolova O, Accardi A, Gutierrez D, Lau A, Rigney M, Grigorieff N (2003) Conformational changes in the C terminus of Shaker K+ channel bound to the rat Kvbeta2-subunit. Proc Natl Acad Sci U S A 100(22):12607–12612
Tipparaju SM, Barski OA, Srivastava S, Bhatnagar A (2008) Catalytic mechanism and substrate specificity of the beta-subunit of the voltage-gated potassium channel. Biochemistry 47(34):8840–8854. doi:10.1021/bi800301b
Tipparaju SM, Liu SQ, Barski OA, Bhatnagar A (2007) NADPH binding to beta-subunit regulates inactivation of voltage-gated K(+) channels. Biochem Biophys Res Commun 359(2):269–276
Tipparaju SM, Saxena N, Liu SQ, Kumar R, Bhatnagar A (2005) Differential regulation of voltage-gated K+ channels by oxidized and reduced pyridine nucleotide coenzymes. Am J Physiol Cell Physiol 288(2):C366–C376
Tristani-Firouzi M, Chen J, Mitcheson JS, Sanguinetti MC (2001) Molecular biology of K(+) channels and their role in cardiac arrhythmias. Am J Med 110(1):50–59
Uebele VN, England SK, Gallagher DJ, Snyders DJ, Bennett PB, Tamkun MM (1998) Distinct domains of the voltage-gated K+ channel Kv beta 1.3 beta-subunit affect voltage-dependent gating. Am J Physiol 274(6 Pt 1):C1485–C1495
Uebele VN, Yeola SW, Snyders DJ, Tamkun MM (1994) Deletion of highly conserved C-terminal sequences in the Kv1 K+ channel sub-family does not prevent expression of currents with wild-type characteristics. FEBS Lett 340(1–2):104–108
VanDongen AM, Frech GC, Drewe JA, Joho RH, Brown AM (1990) Alteration and restoration of K+ channel function by deletions at the N- and C-termini. Neuron 5(4):433–443
Weir EK, Lopez-Barneo J, Buckler KJ, Archer SL (2005) Acute oxygen-sensing mechanisms. N Engl J Med 353(19):2042–2055
Weng J, Cao Y, Moss N, Zhou M (2006) Modulation of voltage-dependent Shaker family potassium channels by an aldo-keto reductase. J Biol Chem 281(22):15194–15200
Xue B, Dunbrack RL, Williams RW, Dunker AK, Uversky VN (2010) PONDR-FIT: a meta-predictor of intrinsically disordered amino acids. Biochim Biophys Acta 1804(4):996–1010
Yu W, Xu J, Li M (1996) NAB domain is essential for the subunit assembly of both alpha–alpha and alpha–beta complexes of shaker-like potassium channels. Neuron 16(2):441–453
Acknowledgments
The authors thank Dr. Maiying Kong for her advice on statistical analysis, and Joshua Salabei and Ermin Villa for their help in cloning experiments. This work was partly supported by NIH grants RR024489 HL-55477, HL-59378 (to A.B.), HL-089372 (to O.A.B.), 0865466D American Heart Beginning-grant-in-aid, HL-102171, Deans Research Funding USF-COP (to S.M.T), and the Program of the Russian Academy of Sciences for the “Molecular and Cellular Biology” (to V.N.U).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tipparaju, S.M., Li, XP., Kilfoil, P.J. et al. Interactions between the C-terminus of Kv1.5 and Kvβ regulate pyridine nucleotide-dependent changes in channel gating. Pflugers Arch - Eur J Physiol 463, 799–818 (2012). https://doi.org/10.1007/s00424-012-1093-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-012-1093-z