Abstract
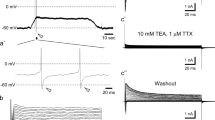
To unravel the functional implications of activity-dependent Cl− changes during early stages of neuronal development, we determined which changes in the GABA reversal potential (E GABA) and GABAergic rheobase shifts were induced by episodes of GABAA receptor activation using gramicidin-perforated patch-clamp recordings from Cajal–Retzius cells in tangential cortical slices of newborn mice. Under this condition, focal application of the GABAA agonist muscimol (10 μM) depolarized the membrane by 15 ± 0.8 mV (n = 35). Such subthreshold GABAergic depolarizations considerably reduced the rheobase, corresponding to an excitatory action. After repetitive focal muscimol applications (50 pulses at 0.5 Hz) a significant reduction of E GABA and an attenuation of the excitatory GABAergic rheobase shift were observed, while the GABAergic membrane conductance and the absolute value of the rheobase were unaltered after the muscimol pulses. Bath application of 100 μM carbachol induced bursts of spontaneous GABAergic postsynaptic potentials. Both, E GABA and the excitatory GABAergic rheobase shift was significantly reduced after such barrage of carbachol-induced GABAergic postsynaptic potentials, while neither the GABAergic membrane conductance nor the absolute value of the rheobase was affected under this condition. Both results indicate that GABAergic activity itself can limit the excitatory effects of GABAA receptor activation, which supports the hypothesis that the low capacity of the Cl− homeostasis in immature neurons could be a substrate for synaptic scaling and homeostatic plasticity.



Similar content being viewed by others
References
Achilles K, Okabe A, Ikeda M, Shimizu-Okabe C, Yamada J, Fukuda A et al (2007) Kinetic properties of Cl uptake mediated by Na+-dependent K+-2Cl cotransport in immature rat neocortical neurons. J Neurosci 27:8616–8627
Ben-Ari Y, Gaiarsa JL, Tyzio R, Khazipov R (2007) GABA: a pioneer transmitter that excites immature neurons and generates primitive oscillations. Physiol Rev 87:1215–1284
Cherubini E, Gaiarsa JL, Ben-Ari Y (1991) GABA: an excitatory transmitter in early postnatal life. Trends Neurosci 14:515–519
Chub N, Mentis GZ, O’Donovan MJ (2006) Chloride-sensitive MEQ fluorescence in chick embryo motoneurons following manipulations of chloride and during spontaneous network activity. J Neurophysiol 95:323–330
Chub N, O’Donovan MJ (2001) Post-episode depression of GABAergic transmission in spinal neurons of the chick embryo. J Neurophysiol 85:2166–2176
de Tassigny AD, Souktani R, Ghaleh B, Henry P, Berdeaux A (2003) Structure and pharmacology of swelling-sensitive chloride channels, I-Cl, I-swell. Fundam Clin Pharmacol 17:539–553
Defazio RA, Hablitz JJ (2001) Chloride accumulation and depletion during GABAA receptor activation in neocortex. Neuroreport 12:2537–2541
Dupont E, Hanganu IL, Kilb W, Hirsch S, Luhmann HJ (2006) Rapid developmental switch in the mechanisms driving early cortical columnar networks. Nature 439:79–83
Farrant M, Kaila K (2007) The cellular, molecular and ionic basis of GABA(A) receptor signalling. Prog Brain Res 160:59–87
Foehring RC, van Brederode JF, Kinney GA, Spain WJ (2002) Serotonergic modulation of supragranular neurons in rat sensorimotor cortex. J Neurosci 22:8238–8250
Hanganu IL, Ben Ari Y, Khazipov R (2006) Retinal waves trigger spindle bursts in the neonatal rat visual cortex. J Neurosci 26:6728–6736
Hanganu IL, Okabe A, Lessmann V, Luhmann HJ (2009) Cellular mechanisms of subplate-driven and cholinergic input-dependent network activity in the neonatal rat somatosensory cortex. Cereb Cortex 19:89–105
Hartmann K, Bruehl C, Golovko T, Draguhn A (2008) Fast homeostatic plasticity of inhibition via activity-dependent vesicular filling. PLoS ONE 3:e2979
Hubner CA, Stein V, Hermans-Borgmeyer I, Meyer T, Ballanyi K, Jentsch TJ (2001) Disruption of KCC2 reveals an essential role of K-Cl cotransport already in early synaptic inhibition. Neuron 30:515–524
Janusonis S, Gluncic V, Rakic P (2004) Early serotonergic projections to Cajal–Retzius cells: relevance for cortical development. J Neurosci 24:1652–1659
Jean-Xavier C, Mentis GZ, O’Donovan MJ, Cattaert D, Vinay L (2007) Dual personality of GABA/glycine-mediated depolarizations in immature spinal cord. Proc Natl Acad Sci USA 104:11477–11482
Kaila K, Voipio J (1987) Postsynaptic fall in intracellular pH induced by GABA-activated bicarbonate conductance. Nature 330:163–165
Khazipov R, Sirota A, Leinekugel X, Holmes GL, Ben-Ari Y, Buzsáki G (2004) Early motor activity drives spindle bursts in the developing somatosensory cortex. Nature 432:758–761
Kilb W, Ikeda M, Uchida K, Okabe A, Fukuda A, Luhmann HJ (2002) Depolarizing glycine responses in Cajal–Retzius cells of neonatal rat cerebral cortex. Neuroscience 112:299–307
Kilb W, Luhmann HJ (2001) Spontaneous GABAergic postsynaptic currents in Cajal–Retzius cells in neonatal rat cerebral cortex. Eur J Neurosci 13:1387–1390
Kilb W, Luhmann HJ (2003) Carbachol-induced network oscillations in the intact cerebral cortex of the newborn rat. Cereb Cortex 13:409–421
Kirmse K, Dvorzhak A, Henneberger C, Grantyn R, Kirischuk S (2007) Cajal Retzius cells in the mouse neocortex receive two types of pre- and postsynaptically distinct GABAergic inputs. J Physiol Lond 585:881–895
Lamsa K, Palva JM, Ruusuvuori E, Kaila K, Taira T (2000) Synaptic GABA(A) activation inhibits AMPA-kainate receptor-mediated bursting in the newborn (P0–P2) rat hippocampus. J Neurophysiol 83:359–366
Lee H, Chen CX, Liu YJ, Aizenman E, Kandler K (2005) KCC2 expression in immature rat cortical neurons is sufficient to switch the polarity of GABA responses. Eur J Neurosci 21:2593–2599
Mienville JM (1998) Persistent depolarizing action of GABA in rat Cajal–Retzius cells. J Physiol Lond 512:809–817
Pozas E, Paco S, Soriano E, Aguado F (2008) Cajal–Retzius cells fail to trigger the developmental expression of the Cl(−) extruding co-transporter KCC2. Brain Res 1239:85–91
Radnikow G, Feldmeyer D, Lubke J (2002) Axonal projection, input and output synapses, and synaptic physiology of Cajal–Retzius cells in the developing rat neocortex. J Neurosci 22:6908–6919
Rheims S, Minlebaev M, Ivanov A, Represa A, Khazipov R, Holmes GL et al (2008) Excitatory GABA in rodent developing neocortex in vitro. J Neurophysiol 100:609–619
Richter D, Luhmann HJ, Kilb W (2010) Intrinsic activation of GABA(A) receptors suppresses epileptiform activity in the cerebral cortex of immature mice. Epilepsia 51:1483–1492
Rivera C, Voipio J, Payne JA, Ruusuvuori E, Lahtinen H, Lamsa K et al (1999) The K+/Cl− co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature 397:251–255
Russell JM (2000) Sodium-potassium-chloride cotransport. Physiol Rev 80:211–276
Sava BA, David CS, Teissier A, Pierani A, Staiger JF, Luhmann HJ et al (2010) Electrophysiological and morphological properties of Cajal–Retzius cells with different ontogenetic origins. Neuroscience 167:724–734
Schwartz TH, Rabinowitz D, Unni V, Kumar VS, Smetters DK, Tsiola A et al (1998) Networks of coactive neurons in developing layer 1. Neuron 20:541–552
Sipila ST, Huttu K, Soltesz I, Voipio J, Kaila K (2005) Depolarizing GABA acts on intrinsically bursting pyramidal neurons to drive giant depolarizing potentials in the immature hippocampus. J Neurosci 25:5280–5289
Soda T, Nakashima R, Watanabe D, Nakajima K, Pastan I, Nakanishi S (2003) Segregation and coactivation of developing neocortical layer 1 neurons. J Neurosci 23:6272–6279
Staley KJ, Soldo BL, Proctor WR (1995) Ionic mechanisms of neuronal excitation by inhibitory GABAA receptors. Science 269:977–981
Stein V, Nicoll RA (2003) GABA generates excitement. Neuron 37:375–378
Sykova E, Nicholson C (2008) Diffusion in brain extracellular space. Physiol Rev 88:1277–1340
Vinay L, Jean-Xavier C (2008) Plasticity of spinal cord locomotor networks and contribution of cation-chloride cotransporters. Brain Res Rev 57:103–110
Wang DD, Kriegstein AR (2009) Defining the role of GABA in cortical development. J Physiol 587:1873–1879
Yamada J, Okabe A, Toyoda H, Kilb W, Luhmann HJ, Fukuda A (2004) Cl− uptake promoting depolarizing GABA actions in immature rat neocortical neurones is mediated by NKCC1. J Physiol 557:829–841
Acknowledgments
This work was supported by a grant from the Deutsche Forschungsgemeinschaft to WK (DFG Ki 835/2). The authors thank B. Krumm for excellent technical work and K. Achilles for her contribution to some experiments. All authors declare that they have no conflicts of interest and state that all experiments comply with the current laws of the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kolbaev, S.N., Luhmann, H.J. & Kilb, W. Activity-dependent scaling of GABAergic excitation by dynamic Cl− changes in Cajal–Retzius cells. Pflugers Arch - Eur J Physiol 461, 557–565 (2011). https://doi.org/10.1007/s00424-011-0935-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-011-0935-4