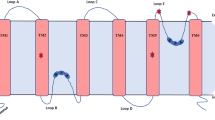
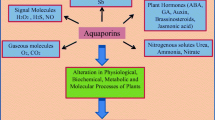
Abstract
We identified barley aquaporins and demonstrated that one, HvPIP2;1, transports water and CO2. Regarding water homeostasis in plants, regulations of aquaporin expression were observed in many plants under several environmental stresses. Under salt stress, a number of plasma membrane-type aquaporins were down-regulated, which can prevent continuous dehydration resulting in cell death. The leaves of transgenic rice plants that expressed the largest amount of HvPIP2;1 showed a 40% increase in internal CO2 conductance compared with leaves of wild-type rice plants. The rate of CO2 assimilation also increased in the transgenic plants. The goal of our plant aquaporin research is to determine the key aquaporin species responsible for water and CO2 transport, and to improve plant water relations, stress tolerance, CO2 uptake or assimilation, and plant productivity via molecular breeding of aquaporins.

Similar content being viewed by others
References
Aharon R, Shahak Y, Wininger S, Bendov R, Kapulnik Y, Galili G (2003) Overexpression of a plasma membrane aquaporin in transgenic tobacco improves plant vigor under favorable growth conditions but not under drought or salt stress. Plant Cell 15:439–447 DOI 10.1105/tpc.009225
Bienert GP, Møller ALB, Kristiansen KA, Schulz A, Møller IM, Schjoerring JK, Jahn TP (2007) Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J Biol Chem 282:1183–1192 DOI 10.1074/jbc.M603761200
Chaumont F, Barrieu F, Jung R, Chrispeels MJ (2000) Plasma membrane intrinsic proteins from maize cluster in two sequence subgroups with differential aquaporin activity. Plant Physiology 122:1025–1034
Clarkson DT, Carvajal M, Henzler T, Waterhouse RN, Smyth AJ, Cooke DT, Steudle E (2000) Root hydraulic conductance: diurnal aquaporin expression and the effects of nutrient stress. J Exp Bot 51:61–70
Cooper GJ, Boron WF (1998) Effect of PCMBS on CO2 permeability of Xenopus oocytes expressing aquaporin 1 or its C189S mutant. Am J Physiol Cell Physiol 275:1481–1486
Fetter K, Wider VV, Moshelion M, Chaumont F (2004) Interactions between plasma membrane aquaporins modulate their water channel activity. Plant Cell 16:215–228 DOI 10.1105/tpc.017194
Flexas J, Ribas-Carbó M, Hanson DT, Bota J, Otto B, Cifre J, McDowell N, Medrano H, Kaldenhoff R (2006) Tobacco aquaporin NtAQP1 is involved in mesophyll conductance to CO2 in vivo. Plant J 48:427–439 DOI 10.1111/j.1365-313X.2006.02879.x
Hanba YT, Shibasaka M, Hayashi Y, Hayakawa T, Kasamo K, Terashima I, Katsuhara M (2004) Overexpression of the barley aquaporin HvPIP2;1 increases internal CO2 conductance and CO2 assimilation in the leaves of transgenic rice plants. Plant Cell Physiol 45:521–529
Jahn TP, MØller ALB, Zeuthen T, Holm LM, Klaerke DA, Mohsin B, Kuhlbratndt WK, Schjoerring JK (2004) Aquaporin homologues in plants and mammals transport ammonia. FEBS Letters 574:31 – 36 DOI 10.1016/j.febslet.2004.08.004
Jang JY, Kim DG, Kim YO, Kim JS, Kang H (2004) An expression analysis of a gene family encoding plasma membrane aquaporins in response to abiotic stresses in Arabidopsis thaliana. Plant Mol Biol 54:713–725
Johanson U, Karlsson M, Johansson I, Gustavsson S, Sjövall S, Fraysse L, Weig AR, Kjellbom P (2001) The complete set of genes encoding major intrinsic proteins in Arabidopsis provides a framework for new nomenclature for major intrinsic proteins in plants. Plant Physiol. 126:1358–1369
Katsuhara M, Akiyama Y, Koshio K, Shibasaka M, Kasamo K (2002) Functional analysis of water channels in barley roots. Plant Cell Physiol 43:885–893
Katsuhara M, Chung GC, Sakurai J, Murai M, Izumi Y, Tsumuki H (2007) Low temperature and aquaporins, a molecular mechanism of water transport. Cryobiol Cryotech 53:21–32
Katsuhara M, Koshio K, Shibasaka M, Hayashi Y, Hayakawa T, Kasamo K (2003) Over-expression of a barely aquaporin increased the shoot/root ratio and raised salt sensitivity in transgenic rice plants. Plant Cell Physiol 44:1378–1383
Katsuhara M, Shibasaka M (2007) Barley root hydraulic conductivity and aquaporins expression in relation to salt tolerance. Soil Sci Plant Nutr 53:466–470
Kjellbom P, Larsson C, Johansson I, Karlsson M, Johanson U (1999) Aquaporins and water homeostasis in plants. Trend Plant Sci 4:308–314
Liu LH, Ludewig U, Gassert B, Frommer WB, von Wiren N (2003) Urea transport by nitrogen-regulated tonoplast intrinsic proteins in Arabidopsis. Plant Physiol 133:1220–1228
Luu DT, Maurel C (2005) Aquaporins in a challenging environment: molecular gears for adjusting plant water status. Plant Cell Environ 28:85–96 DOI 10.1111/j.1365-3040.2004.01295.x
Ma JF, Tamai K, Yamaji N, Mitani N, Konishi S, Ishiguro M, Katsuhara M, Murata Y, Yano M (2006) Silicon transporter in rice. Nature 440:688–691 DOI 10.1038/nature04590
Nakhoul NL, Bruce AD, Romero MF, Boron WF (1998) Effect of expressing the aquaporin aquaporin-1 on the CO2 permeability of Xenopus oocytes. Am J Physiol Cell Physiol 274:543–548
Sakurai J, Ishikawa F, Yamaguchi T, Uemura M, Maeshima M (2005) Identification of 33 rice aquaporin genes and analysis of their expression and function. Plant Cell Physiol 46:1568–1577 DOI 10.1093/pcp/pci172
Sato-Nara K, Nagasaka A, Yamashita H, Ishida J, Enju A, Seki M, Shinozaki K, Suzuki H (2004) Identification of genes regulated by dark adaptation and far-red light illumination in roots of Arabidopsis thaliana. Plant Cell Environ 27:1387–1394
Suga S, Komatsu S, Maeshima M (2002) Aquaporin isoforms responsive to salt and water stresses and phytohormones in radish seedlings. Plant Cell Physiol 43:1229–1237
Takano J, Wada M, Ludewig U, Schaaf G, Wirén N, Fujiwara T (2006) The Arabidopsis major intrinsic protein NIP5;1 is essential for efficient boron uptake and plant development under boron limitation. Plant Cell 18:1498–1509
Terashima I, Ono K (2002) Effects of HgCl2 on CO2 dependence of leaf photosynthesis: evidence indicating involvement of aquaporins in CO2 diffusion across the plasma membrane. Plant Cell Physiol 43:70–78 DOI 10.1105/tpc.106.041640
Törnroth-Horsefield S, Wang Y, Hedfalk K, Johanson U, Karlsson M, Tajkhorshid E, Neutze R, Kjellbom P (2006) Structural mechanism of plant aquaporin gating. Nature 439:688–659 DOI 10.1038/nature04316
Tyerman SD, Bohnert H, Maurel C, Steudle E, Smith JAC (1999) Plant aquaporins: their molecular biology, biophysics and significance for plant water relations. J Exp Bot 50:1055–1071
Uehlein N, Lovisolo C, Siefritz F, Kalenhoff R (2003) The tobacco aquaporin NtAQP1 is a membrane CO2 pore with physiological functions. Nature 425:734–737
Vandeleur R, Niemietz C, Tilbrook J, Tyerman SD (2005) Roles of aquaporins in root responses to irrigation. Plant Soil 274:141–161 DOI 10.007/s11104-004-8070-z
Yang X, Fukuda N, van Hoek A, Matthay MA, Ma TH, Verkman AS (2000) Carbon dioxide permeability of aquaporin-1 measured in erythrocytes and lung of aquaporin-1 null mice and in reconstituted proteoliposomes. J Bio Chem 25:2686–2692
Zelazny E, Borst JW, Muylaert M, Batoko H, Hemminga MA, Chaumont F (2007) FRET imaging in living maize cells reveals that plasma membrane aquaporins interact to regulate their subcellular localization. Pro Nat Acad Sci 104:12359–12364 DOI 10.1073/pnas.0701180104
Zhu C, Schraut D, Hartung W, Schäffner AR (2005) Differential responses of maize MIP genes to salt stress and ABA. J Exp Bot 56:2971–2981 DOI 10.1093/jxb/eri294
Acknowledgements
The authors are grateful to Dr. Shibasaka and graduate students as collaborators. Transgenic rice studies involved collaboration with Dr. Hayashi and Dr. Hayakawa (Plantech Research Institute). This manuscript summarized work supported by the Bio Design Program, Core Research for Evolutional Science and Technology of the Japanese Science and Technology Corporation, and the Program for the Promotion of Basic Research Activities for Innovative Biosciences to MK.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katsuhara, M., Hanba, Y.T. Barley plasma membrane intrinsic proteins (PIP Aquaporins) as water and CO2 transporters. Pflugers Arch - Eur J Physiol 456, 687–691 (2008). https://doi.org/10.1007/s00424-007-0434-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-007-0434-9