Abstract
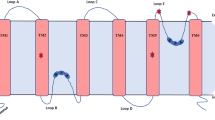
Plant has a large aquaporin family with more than 30 members which are divided into four subfamilies: plasma membrane intrinsic protein (PIP), tonoplast intrinsic protein (TIP), nodulin 26-like intrinsic proteins (NIP), and small and basic intrinsic proteins (SIP). Their primary structure, transport substrate, functional regulation, gene expression profile, protein amount, and intracellular localization are diversified. The SIP members have short N-terminal tails. Most aquaporins have two sets of common Asn–Pro–Ala (NPA) motif; however, the first motif of SIP1;1, SIP1;2, and SIP2;1 is changed to NPT, NPC, and NPL, respectively. SIP1;1 and SIP1;2, but not SIP2;1, have water transport activity. A recent study revealed that all three members of SIP are localized to the ER membrane and expressed in a cell specific manner in Arabidopsis thaliana. An overview is given on the main features of the SIP members in terms of their primary structure, ER membrane retention, homologues in mammals, and physiological function.




Similar content being viewed by others
References
Alexandersson E, Fraysse L, Sjovall-Larsen S, Gustavsson S, Fellert M, Karlsson M, Johanson U, Kjellbom P (2005) Whole gene family expression and drought stress regulation of aquaporins. Plant Mol Biol 59:469–484
Barlowe C (2003) Signals for COPII-dependent export from the ER: what’s the ticket out? Trends Cell Biol 13:295–300
Bienert GP, Møller ALB, Kristiansen KA, Schulz A, Møller IM, Schjoerring JK, Jahn TP (2007) Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J Biol Chem 282:1183–1192
Chaumont F, Barrieu F, Jung R, Chrispeels MJ (2000) Plasma membrane intrinsic proteins from maize cluster in two sequence subgroups with differential aquaporin activity. Plant Physiol 122:1025–1034
Chaumont F, Barrieu F, Wojcik E, Chrispeels MJ, Jung R (2001) Aquaporins constitute a large and highly divergent protein family in maize. Plant Physiol 125:1206–1215
Chaumont F, Moshelion M, Daniels MK (2005) Regulation of plant aquaporin activity. Biol Cell 97:749–764
Chen Y-F, Randlett MD, Findell JL, Schaller GE (2002) Localization of the ethylene receptor ETR1 to the endoplasmic reticulum of Arabidopsis. J Biol Chem 277:19861–19866
Dean RM, Rivers RL, Zeidel ML, Roberts DM (1999) Purification and functional reconstitution of soybean nodulin 26: an aquaporin with water and glycerol transport properties. Biochemistry 38:347–353
Flexas J, Ribas-Carbó M, Hanson DT, Bota J, Otto B, Cifre J, McDowell N, Medrano H, Kaldenhoff R (2006) Tobacco aquaporin NtAQP1 is involved in mesophyll conductance to CO2 in vivo. Plant J 48:427–439
Gonen T, Cheng Y, Sliz P, Hiroaki Y, Fujiyoshi Y, Harrison SC, Walz T (2005) Lipid–protein interactions in double-layered two-dimensional AQP0 crystals. Nature 438:633–638
Gorelick DA, Praetorius J, Tsunenari T, Nielsen S, Agre P (2006) Aquaporin-11: a channel protein lacking apparent transport function expressed in brain. BMC Biochem 7:14
Hanba YT, Shibasaka M, Hayashi Y, Hayakawa T, Kasamo K, Terashima I, Katsuhara M (2004) Overexpression of the barley aquaporin HvPIP2;1 increases internal CO2 conductance and CO2 assimilation in the leaves of transgenic rice plants. Plant Cell Physiol 45:521–529
Hachez C, Zelazny E, Chaumont F (2006) Modulating the expression of aquaporin genes in planta: a key to understand their physiological functions? Biochim Biophys Acta 1758:1142–1156
Hanton SL, Renna L, Bortolotti LE, Chatre L, Stefano G, Brandizzi F (2005) Diacidic motifs influence the export of transmembrane proteins from the endoplasmic reticulum in plant cells. Plant Cell 17:3081–3093
Ishikawa F, Suga S, Uemura T, Sato MH, Maeshima M (2005) Three SIP aquaporins of Arabidopsis are localized in the ER membrane and expressed in a tissue- and cell-specific manner. FEBS Lett 579:5814–5820
Ishibashi K (2006) Aquaporin subfamily with unusual NPA boxes. Biochem Biophys Acta 1758:989–993
Itoh T, Rai T, Kuwahara M, Ko SBH, Uchida S, Sasaki S, Ishibashi K (2005) Identification of a novel aquaporin, AQP12, expressed in pancreatic acinar cells. Biochem Biophys Res Commun 330:832–838
Jahn TP, MØller ALB, Zeuthen T, Holm LM, Klaerke DA, Mohsin B, Kuhlbratndt WK, Schjoerring JK (2004) Aquaporin homologues in plants and mammals transport ammonia. FEBS Lett 574:31–36
Jiang L, Phillips TE, Hamm CA, Drozdowicz YM, Rea PA, Maeshima M, Rogers SW, Rogers JC (2001) The protein storage vacuole: a unique compound organelle. J Biol Chem 155:991–1002
Johanson U, Karlsson M, Johansson I, Gustavsson S, Sjövall S, Fraysse L, Weig AR, Kjellbom P (2001) The complete set of genes encoding major intrinsic proteins in Arabidopsis provides a framework for new nomenclature for major intrinsic proteins in plants. Plant Physiol 126:1358–1369
Johanson U, Gustavsson S (2002) A new subfamily of major intrinsic proteins in plants. Mol Biol Evol 19:456–461
Kaldenhoff R, Fischer M (2006) Functional aquaporin diversity in plants. Biochem Biophys Acta 1758:1134–1141
Kobae Y, Mizutani M, Segami S, Maeshima M (2006) Immunochemical analysis of aquaporin isoforms in Arabidopsis suspension cultured cells. Biosci Biotech Biochem 70:980–987
Loque D, Ludewig U, Yuan L, von Wiren N (2005) Tonoplast intrinsic proteins AtTIP2;1 and AtTIP2;3 facilitate NH3 transport into the vacuole. Plant Physiol 137:671–680
Luu DT, Maurel C (2005) Aquaporins in a challenging environment: molecular gears for adjusting plant water status. Plant Cell Environ 28:85–96
Ma JF, Tamai K, Yamaji N, Mitani N, Konishi S, Ishiguro M, Katsuhara M, Murata Y, Yano M (2006) Silicon transporter in rice. Nature 440:688–691
Maeshima M, Hara-Nishimura I, Takeuchi Y, Nishimura M (1994) Accumulation of vacuolar H+-pyrophosphatase and H+-ATPase during reformation of the central vacuole in germinating pumpkin seeds. Plant Physiol 106:61–69
Maeshima M (2001) Tonoplast transporters: organization and function. Annu Rev Plant Physiol Plant Mol Biol 52:469–497
Maurel C (2007) Plant aquaporins: Novel functions and regulation properties. FEBS Lett 581:2227–2236
Mikosch M, Hurst AC, Hertel B, Homann U (2006) Diacidic motif is required for efficient transport of the K+ channel KAT1 to the plasma membrane. Plant Physiol 142:923–930
Morishita Y, Matsuzaki T, Hara-Chikuma M, Andoo A, Shimono M, Matsuki A, Kobayashi K, Ikeda M, Yamamoto T, Verkman A, Kusano E, Ookawara S, Takata K, Sasaki S, and Ishibashi K (2005) Disruption of aquaporin-11 produces polycystic kidneys following vacuolization of the proximal tubule. Mol Cell Biol 25:7770–7779
Mizutani M, Watanabe S, Nakagawa T, Maeshima M (2006) Aquaporin NIP2;1 is mainly localized to the ER membrane and shows root-specific accumulation in Arabidopsis thaliana. Plant Cell Physiol 47:1420–1426
Sakurai J, Ishikawa F, Yamaguchi T, Uemura M, Maeshima M (2005) Identification of 33 rice aquaporin genes and analysis of their expression and function. Plant Cell Physiol 46:1568–1577
Santoni V, Verdoucq L, Sommerer N, Vinh J, Pflieger D, Maurel C (2006) Methylation of aquaporins in plant plasma membrane. Biochem J 400:189–197
Suga S, Imagawa S, Maeshima M (2001) Specificity of the accumulation of mRNAs and proteins of the plasma membrane and tonoplast aquaporins in radish organs. Planta 212:294–304
Suga S, Komatsu S, Maeshima M (2002) Aquaporin isoforms responsive to salt and water stresses and phytohormones in radish seedlings. Plant Cell Physiol 43:1229–1237
Suga S, Maeshima M (2004) Water channel activity of radish plasma membrane aquaporins heterologously expressed in yeast and their modification by site-directed mutagenesis. Plant Cell Physiol 45:823–830
Takano J, Wada M, Ludewig U, Schaaf G, Wirén N, Fujiwara T (2006) The Arabidopsis major intrinsic protein NIP5;1 is essential for efficient boron uptake and plant development under boron limitation. Plant Cell 18:1498–1509
Tournaire-Roux C, Sutka M, Javot H, Gout E, Gerbeau P, Luu DFT, Bligny R, Maurel C (2003) Cytosolic pH regulates root water transport during anoxic stress through gating of aquaporins. Nature 425:393–397
Törnroth-Horsefield S, Wang Y, Hedfalk K, Johanson U, Karlsson M, Tajkhorshid E, Neutze R, Kjellbom P (2006) Structural mechanism of plant aquaporin gating. Nature 439:688–694
Uehlein N, Lovisolo C, Siefritz F, Kalenhoff R (2003) The tobacco aquaporin NtAQP1 is a membrane CO2 pore with physiological functions. Nature 425:734–737
Wallace IS, Choi WG, Roberts DM (2006) The structure, function and regulation of the nodulin 26-like intrinsic protein family of plant aquaglyceroporins. Biochem Biophys Acta 1758:1165–1175
Weig AR, Jakob C (2000) Functional identification of the glycerol permease activity of Arabidopsis thaliana NLM1 and NLM2 proteins by heterologous expression in Saccharomyces cerevisiae. FEBS Lett 481:293–298
Wilkinson JQ, Lanahan MB, Yen H-C, Giovannoni JJ, Klee HJ (1995) An ethylene-inducible component of signal transduction encoded by Never-ripe. Science 270:1809–1813
Yakata K, Hiroaki Y, Ishibashi K, Sohara E, Sasaki S, Mitsuoka K, Fujiyoshi Y (2007) Aquaporin-11 containing a divergent NPA motif has normal water channel activity. Biochem Biophys Acta 1768:688–693
Zimmermann P, Hennig L, Gruissem W (2005) Gene-expression analysis and network discovery using Genevestigator. Trends Plant Sci 10:407–409
Acknowledgments
We thank Sei Sasaki for the chance to write this review. Work in our laboratory was supported by the Program for Promotion of Basic Research Activities of Innovative Biosciences (PROBRAIN), RITE, Grants-in-Aid for Scientific Research (18380064 and 16085204) from the Ministry of Education, Sports, Culture, Science and Technology of Japan, and the Global Research Program of the Ministry of Science and Technology of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maeshima, M., Ishikawa, F. ER membrane aquaporins in plants. Pflugers Arch - Eur J Physiol 456, 709–716 (2008). https://doi.org/10.1007/s00424-007-0363-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-007-0363-7