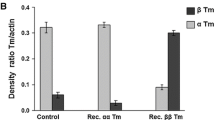
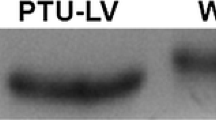
Abstract
Cross-bridge kinetics underlying stretch-induced force transients was studied in cardiac muscle strips with different myosin heavy chain (MHC) and myosin light chain (MLC) isoforms. The force transients were induced by stepwise stretches of maximally Ca2+-activated skinned muscle strips. The MHC and MLC isoforms were analyzed by electrophoreses after the mechanical experiments. Muscle strips of euthyroid rats and mice exclusively containing α-MHC were used. In addition, muscle strips of hyper- and hypothyroid rats containing different combinations of MHC and MLC isoforms were used. The thyroid hormone is known to alter the expression of MHC but not of MLC isoforms. In muscle strips containing exclusively α-MHC, atrial MLC isoforms (all atria of rats and mice) were associated with about 30% faster kinetics than ventricular MLC isoforms (ventricles of hyperthyroid rats and some muscle strips of ventricles of euthyroid rats and mice). On the other hand, in muscle strips containing exclusively ventricular MLC isoforms, α-MHC (ventricles of hyperthyroid rats) was associated with about 2.6 times faster kinetics than β-MHC (ventricles of hypothyroid rats). We conclude that the MLC isoforms fine-tune cross-bridge kinetics, which underlies stretch-induced force transients, whereas the MHC isoforms mainly determine this kinetics. The effect of MLC isoforms on the cross-bridge kinetics may partially contribute to the faster twitch contraction in atria than in ventricles. Furthermore, it may play a role in various cardiomyopathies where atrial MLC isoforms are partially expressed in ventricles or ventricular MLC isoforms are partially expressed in atria.



Similar content being viewed by others
References
Abdelaziz AI, Pagel I, Schlegel WP, Kott M, Monti J, Haase H, Morano I (2005) Human atrial myosin light chain 1 expression attenuates heart failure. Adv Exp Med Biol 565:283–292
Alpert NR, Brosseau C, Federico A, Krenz M, Robbins J, Warshaw DM (2002) Molecular mechanics of mouse cardiac myosin isoforms. Am J Physiol 283:H1446–H1454
Andruchov O, Andruchova O, Wang Y, Galler S (2004) Kinetic properties of myosin heavy chain isoforms in mouse skeletal muscle: comparison with rat, rabbit and human and correlation with amino acid sequence. Am J Physiol 287:C1725–C1732
Andruchov O, Wang Y, Andruchova O, Galler S (2004) Functional properties of skinned rabbit skeletal and cardiac muscle preparations containing α-cardiac myosin heavy chain. Pflugers Arch 448:44–53
Blanchard E, Seidman C, Seidman JG, LeWinter M, Maughan D (1999) Altered crossbridge kinetics in the α-MHC403/+ mouse model of familial hypertrophic cardiomyopathy. Circ Res 84:475–483
Bottinelli R, Canepari M, Cappelli V, Reggiani C (1995) Maximum speed of shortening and ATPase activity in atrial and ventricular myocardia of hyperthyroid rats. Am J Physiol 269:C785–C790
Cappelli V, Moggio R, Polla B, Bottinelli R, Pogessi C, Reggiani C (1988) The dual effect of thyroid hormones on contractile properties of rat myocardium. Pflügers Arch 411:620–627
Davis JS, Satorius CL, Epstein ND (2002) Kinetic effects of myosin regulatory light chain phosphorylation on skeletal muscle contraction. Biophys J 83:359–370
Fewell JG, Hewet TE, Sanbe A, Klevitsky R, Hayes E, Warshaw D, Maughan D, Robbins J (1998) Functional significance of cardiac myosin essential light chain isoform switching in transgenic mice. J Clin Invest 101:2630–2639
Galler S, Hilber K (1994) Unloaded shortening of skinned mammalian skeletal muscle fibres. Effects of the experimental approach and passive force. J Muscle Res Cell Motil 15:400–412
Galler S, Hilber K, Pette D (1996) Force responses following stepwise length changes of rat skeletal muscle fibre types. J Physiol 493:219–227
Galler S, Puchert E, Gohlsch B, Schmid D, Pette D (2002) Kinetic properties of cardiac myosin heavy chain isoforms in rat. Pflugers Arch 445:218–223
Galler S, Schmitt T, Pette D (1994) Stretch activation, unloaded shortening velocity, and myosin heavy chain isoforms of rat skeletal muscle fibres. J Physiol 478:513–521
Galler S, Wang G, Kawai M (2005) Elementary steps of the cross-bridge cycle in fast-twitch fiber types from rabbit skeletal muscles. Biophys J 89:3248–3260
Galler S (1999) Ca2+, Sr2+ force relationships and kinetic properties of fast-twitch rat leg muscle fibre subtypes. Acta Physiol Scand 167:131–141
Heinl P, Kuhn HJ, Rüegg JC (1974) Tension responses to quick length changes of glycerinated skeletal muscle fibres from frog and tortoise. J Physiol 237:243–258
Huxley AF, Simmons RM (1971) Proposed mechanism of force generation in striated muscle. Nature 233:533–538
Kaasik A, Minajeva A, Paju K, Eimre M, Seppet EK (1997) Thyroid hormones differentially affect sarcoplasmic reticulum function in rat atria and ventricles. Mol Cell Biochem 176:119–126
Kawai M, Brandt PW (1980) Sinusoidal analysis: a high resolution method for correlating biochemical reactions with physiological processes in activated skeletal muscles of rabbit, frog and crayfish. J Muscle Res Cell Motil 1:279–303
Kawai M, Saeki Y, Zhao Y (1993) Crossbridge scheme and the kinetic constants of elementary steps deduced from chemically skinned papillary and trabecular muscles of the ferret. Circ Res 73:35–50
Khalina YN, Bartsch H, Petzhold D, Haase H, Podlubnaya ZA, Shpagina MD, Morano I (2005) Reconstitution of ventricular myosin with atrial light chains 1 improves its functional properties. Acta Biochim Pol 52:443–448
Lowey S, Risby D (1971) Light chains from fast and slow muscle myosins. Nature 234:81–85
Lüss I, Boknik P, Jones LR, Kirchhefer U, Knapp J, Linck B, Luss H, Meissner A, Muller FU, Schmitz W, Vahlensieck U, Neumann J (1999) Expression of cardiac calcium regulatory proteins in atrium v ventricle in different species. J Mol Cell Cardiol 31:1299–1314
Maughan D, Moore J, Vigoreaux J, Barnes B, Mulieri LA (1998) Work production and work absorption in muscle strips from vertebrate cardiac and insect flight muscle fibers. Adv Exp Med Biol 453:471–480
Mercadier JJ, Bouveret P, Gorza L, Schiaffino S, Clark WA, Zak R, Swynghedauw B, Schwartz K (1983) Myosin isoenzymes in normal and hypertrophied human ventricular myocardium. Circ Res 53:52–62
Metzger JM, Wahr PA, Michelle DE, Albayya F, Westfall MV (1999) Effects of myosin heavy chain isoform switching on Ca2+-activated tension development in single adult cardiac myocytes. Circ Res 84:1310–1317
Miller MS, Palmer BM, Ruch S, Martin LA, Farman GP, Wang Y, Robbins J, Irving TC, Maughan DW (2005) The essential light chain N-terminal extension alters force and fiber kinetics in mouse cardiac muscle. J Biol Chem 280:24434–34427
Morano I, Haase H (1997) Different actin affinities of human cardiac essential myosin light chain isoforms. FEBS Lett 408:71–74
Morano I, Arndt H, Gärtner C, Rüegg JC (1988) Skinned fibers of human atrium and ventricle: myosin isoenzymes and contractility. Circ Res 62:632–639
Morano I, Ritter O, Bonz A, Timek T, Vahl CF, Michel G (1995) Myosin light chain–actin interaction regulates cardiac contractility. Circ Res 76:720–725
Morano I (1999) Tuning the human heart molecular motors by myosin light chains. J Mol Med 77:544–555
Morano M, Zacharzowski U, Maier M, Lange PE, Alexi-Meskishvili V, Haase H, Morano I (1996) Regulation of human heart contractility by essential myosin light chain isoforms. J Clin Invest 98:467–473
Narolska N, van Loon R, Boontje N, Zaremba R, Penas S, Russell J, Spiegelenberg SR, Huybregts MA, Visser FC, de Jong JW, van der Velden J, Stienen GJ (2005) Myocardial contraction is 5-fold more economical in ventricular than in atrial human tissue. Cardiovasc Res 65:221–229
Piazzesi G, Linari M, Reconditi M, Vanzi F, Lombardi (1997) Cross-bridge detachment and attachment following a step stretch imposed on active single frog muscle fibres. J Physiol 498:3–15
Puchert E, Andruchov O, Wagner A, Grassberger H, Lahnsteiner F, Sobieszek A, Galler S (2003) Slowing effect of Mg2+ on contractile kinetics of skinned preparations of rat hearts depending on myosin heavy chain isoform content. Pflugers Arch 447:135–141
Rayment I, Rypniewski WR, Schmidt-Bäse K, Smith R, Tomchick DR, Benning MM, Winkelmann DA, Wesenberg G, Holden HM (1993) Three-dimensional structure of a myosin subfragment-1: a molecular motor. Science 261:50–58
Reiser PJ, Kline WO (1998) Electrophoretic separation and quantitation of cardiac myosin heavy chain isoforms in eight mammalian species. Am J Physiol 274:H1048–H1053
Rossmanith GH, Hoh JF, Kirman A, Kwan LJ (1986) Influence of V1 and V3 isomyosins on the mechanical behaviour of rat papillary muscle as studied by pseudo-random binary noise modulated length perturbations. J Muscle Res Cell Motil 7:307–319
Rundell VL, Manaves V, Martin AF, de Tombe PP (2005) Impact of b-myosin heavy chain isoform expression on cross-bridge cycling kinetics. Am J Physiol 288:H896–H903
Saeki Y, Kawai M, Zhao Y (1991) Comparison of crossbridge dynamics between intact and skinned myocardium from ferret right ventricles. Circ Res 68:772–781
Sanbe A, Gulick J, Hayes E, Warshaw D, Osinska H, Chan C-B, Klevitsky R, Robbins J (2000) Myosin light chain replacement in the heart. Am J Physiol 279:H1355–H1364
Schaub MC, Hefti MA, Zuellig RA, Morano I (1998) Modulation of contractility in human cardiac hypertrophy by myosin essential light chain isoforms. Cardiovasc Res 37:381–404
Timson DJ (2003) Fine tuning of myosin motor: the role of essential light chain in striated muscle myosin. Biochimie 85:639–645
VanBuren P, Harris DE, Alpert NR, Warshaw DM (1995) Cardiac V1 and V3 myosins differ in their hydrolytic and mechanical activities in vitro. Circ Res 77:439–444
Vemuri R, Lankford EB, Poetter K, Hassanzadeh S, Takeda K, Yu Z-X, Ferrans VJ, Epstein ND (1999) The stretch-activation response may be critical to the proper functioning of the mammalian heart. Proc Natl Acad Sci U S A 96:1048–1053
Yamashita H, Sugiura S, Fujita H, Yasuda S, Nagai R, Saeki Y, Sunagawa K, Sugi H (2003) Myosin light chain isoforms modify force-generating ability of cardiac myosin by changing the kinetics of actin–myosin interaction. Cardiovasc Res 60:580–588
Acknowledgements
We are grateful to Dr. D. Günzel and Mag. H. Grassberger for critically reading the manuscript. This work was supported by a grant from the Austrian Fonds zur Förderung der Wissenschaftlichen Forschung (FWF-P16709-B09) and by the South Tyrolean Sparkasse.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andruchov, O., Andruchova, O. & Galler, S. Fine-tuning of cross-bridge kinetics in cardiac muscle of rat and mouse by myosin light chain isoforms. Pflugers Arch - Eur J Physiol 452, 667–673 (2006). https://doi.org/10.1007/s00424-006-0080-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-006-0080-7