Abstract
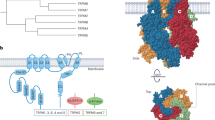
Transient receptor potential (TRP) channel proteins are divided into seven subgroups that are currently designated as TRPC (canonical), TRPV (vanilloid), TRPM (melastatin), TRPN (NOMP-C, from no mechanoreceptor potential-C), TRPA (ankyrin-like with transmembrane domains 1) and TRPP (polycystin). TRPC, TRPV and TRPM are related to canonical TRP proteins whereas TRPN, TRPA and TRPP (polycystin) are more divergent. Most TRP channels are linked to sensory stimuli, including phototransduction, thermosensation and mechanosensation. The TRPP subfamily was named after its founding member, polycystin kidney disease-2 (PKD2), a gene product mutated in many cases of autosomal dominant polycystic kidney disease (ADPKD). ADPKD is a major inherited nephropathy, affecting over 1:1,000 of the worldwide population, characterized by the progressive development of fluid-filled cysts from the tubules and collecting ducts of affected kidneys. Loss-of-function mutations in either polycystin-2, a non-selective cation channel, or polycystin-1 (PKD1), a large plasma membrane integral protein, give rise to ADPKD. PKD1 and PKD2 are thought to function together as part of a multiprotein receptor/ion-channel complex or independently and may be involved in transducing Ca2+-dependent mechanosensitive signals in response to cilia bending in renal epithelial cells and endodermally derived cells. Further information on the growing number and physiological properties of these TRP-polycystins is the basis of this review.




Similar content being viewed by others
References
Alenghat FJ, Nauli SM, Kolb R, Zhou J, Ingber DE (2004) Global cytoskeletal control of mechanotransduction in kidney epithelial cells. Exp Cell Res 301:23–30
Alessandri-Haber N, Yeh JJ, Boyd AE, Parada CA, Chen X, Reichling DB, Levine JD (2003) Hypotonicity induces TRPV4-mediated nociception in rat. Neuron 39:497–511
Barr MM, DeModena J, Braun D, Nguyen CQ, Hall DH, Sternberg PW (2001) The Caenorhabditis elegans autosomal dominant polycystic kidney disease gene homologs lov-1 and pkd-2 act in the same pathway. Curr Biol 11:1341–1346
Barr MM, Sternberg PW (1999) A polycystic kidney-disease gene homologue required for male mating behaviour in C. elegans. Nature 401:386–389
Bhunia AK, Piontek K, Boletta A, Liu L, Qian F, Xu PN, Germino FJ, Germino GG (2002) PKD1 induces p21(waf1) and regulation of the cell cycle via direct activation of the JAK-STAT signaling pathway in a process requiring PKD2. Cell 109:157–168
Cai Y, Anyatonwu G, Okuhara D, Lee KB, Yu Z, Onoe T, Mei CL, Qian Q, Geng L, Witzgall R, Ehrlich BE, Somlo S (2004) Calcium dependence of polycystin-2 channel activity is modulated by phosphorylation at Ser812. J Biol Chem 279:19987–19995
Cai Y, Maeda Y, Cedzich A, Torres VE, Wu G, Hayashi T, Mochizuki T, Park JH, Wiztgall R, Somlo S (1999) Identification and characterization of polycystin-2, the PKD2 gene product. J Biol Chem 274:28557–28565
Calvet JP (2003) New insights into ciliary function: kidney cysts and photoreceptors. Proc Natl Acad Sci USA 100:5583–5585
Chen XZ, Segal Y, Basora N, Guo L, Peng JB, Babakhanlou H, Vassilev PM, Brown EM, Hediger MA, Zhou J (2001) Transport function of the naturally occurring pathogenic polycystin-2 mutant, R742X. Biochem Biophys Res Commun 282:1251–1256
Chen XZ, Vassilev PM, Basora N, Peng JB, Nomura H, Segal Y, Brown EM, Reeders ST, Hediger MA, Zhou J (1999) Polycystin-L is a calcium-regulated cation channel permeable to calcium ions. Nature 401:383–386
Corey DP (2003) New TRP channels in hearing and mechanosensation. Neuron 39:585–588
Corey DP, Garcia-Anoveros J, Holt JR, Kwan KY, Lin SY, Vollrath MA, Amalfitano A, Cheung EL, Derfler BH, Duggan A, Geleoc GS, Gray PA, Hoffman MP, Rehm HL, Tamasauskas D, Zhang DS (2004) TRPA1 is a candidate for the mechanosensitive transduction channel of vertebrate hair cells. Nature 432:723–730
Delmas P (2004) Assembly and gating of TRPC channels in signalling microdomains. Novartis Found Symp 258:75–102
Delmas P (2004) Polycystins: from mechanosensation to gene regulation. Cell 118:145–148
Delmas P, Padilla F, Osorio N, Coste B, Raoux M, Crest M (2004) Polycystins, calcium signaling, and human diseases. Biochem Biophys Res Commun 322:1374–1383
Delmas P, Nauli SM, Li X, Coste B, Osorio N, Crest M, Brown DA, Zhou J (2004) Gating of the polycystin ion channel signaling complex in neurons and kidney cells. FASEB J 18:740–742
Delmas P, Nomura H, Li X, Lakkis M, Luo Y, Segal Y, Fernandez-Fernandez JM, Harris P, Frischauf AM, Brown DA, Zhou J (2002) Constitutive activation of G-proteins by polycystin-1 is antagonized by polycystin-2. J Biol Chem 277:11276–11283
Foggensteiner L, Bevan AP, Thomas R, Coleman N, Boulter C, Bradley J, Ibraghimov-Beskrovnaya O, Klinger K, Sandford R (2000) Cellular and subcellular distribution of polycystin-2, the protein product of the PKD2 gene. J Am Soc Nephrol 11:814–827
Gallagher AR, Cedzich A, Gretz N, Somlo S, Witzgall R (2000) The polycystic kidney disease protein PKD2 interacts with Hax-1, a protein associated with the actin cytoskeleton. Proc Natl Acad Sci USA 97:4017–4022
Gao Z, Ruden DM, Lu X (2003) PKD2 cation channel is required for directional sperm movement and male fertility. Curr Biol 13:2175–2178
Gattone VH, Wang X, Harris PC, Torres VE (2003) Inhibition of renal cystic disease development and progression by a vasopressin V2 receptor antagonist. Nat Med 9:1323–1326
Gonzalez-Perrett S, Kim K, Ibarra C, Damiano AE, Zotta E, Batelli M, Harris PC, Reisin IL, Arnaout MA, Cantiello HF (2001) Polycystin-2, the protein mutated in autosomal dominant polycystic kidney disease (ADPKD), is a Ca2+-permeable nonselective cation channel. Proc Natl Acad Sci USA 98:1182–1187
Hamill OP, Martinac B (2001) Molecular basis of mechanotransduction in living cells. Physiol Rev 81:685–740
Hanaoka K, Qian F, Boletta A, Bhunia AK, Piontek K, Tsiokas L, Sukhatme VP, Guggino WB, Germino GG (2000) Co-assembly of polycystin-1 and −2 produces unique cation-permeable currents. Nature 408:990–994
Hang Le N et al (2004) Aberrant polycystin-1 expression results in modification of activator protein-1 activity, whereas Wnt signalling remains unaffected. J Biol Chem 26:27472–27481
Hirohashi S, Kanai Y (2003) Cell adhesion system and human cancer morphogenesis. Cancer Sci 94:575–581
Hirohashi N, Vacquier VD (2002) High molecular mass egg fucose sulfate polymer is required for opening both Ca2+ channels involved in triggering the sea urchin sperm acrosome reaction. J Biol Chem 277:1182–1189
Huan Y, van Adelsberg J (1999) Polycystin-1, the PKD1 gene product, is in a complex containing E-cadherin and the catenins. J Clin Invest 104:1459–1468
Jordt SE, Bautista DM, Chuang HH, McKemy DD, Zygmunt PM, Hogestatt ED, Meng ID, Julius D (2004) Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 427:260–265
Kim E et al (1999) Interaction between RGS7 and polycystin. Proc Natl Acad Sci USA 96:6371–6376
Kim E, Arnould T, Sellin LK, Benzing T, Fan MJ, Gruning W, Sokol SY, Drummond I, Walz G (1999) The polycystic kidney disease 1 gene product modulates Wnt signalling. J Biol Chem 274:4947–4953
Köttgen M et al (2005) Trafficking of TRPP2 by PACS proteins represents a novel mechanism of ion channel regulation. EMBO J 24:705–716
Koulen P, Cai Y, Geng L, Maeda Y, Nishimura S, Witzgall R, Ehrlich BE, Somlo S (2002) Polycystin-2 is an intracellular calcium release channel. Nat Cell Biol 4:191–197
Li Q, Dai Y, Guo L, Liu Y, Hao C, Wu G, Basora N, Michalak M, Chen XZ (2003) Polycystin-2 associates with tropomyosin-1, an actin microfilament component. J Mol Biol 325:949–362
Li Q, Liu Y, Zhao W, Chen XZ (2002) The calcium-binding EF-hand in polycystin-L is not a domain for channel activation and ensuing inactivation. FEBS Lett 516:270–278
Li A, Tian X, Sung SW, Somlo S (2003) Identification of two novel polycystic kidney disease-1-like genes in human and mouse genomes. Genomics 81:596–608
Liedtke W, Choe Y, Marti-Renom MA, Bell AM, Denis CS, Sali A, Hudspeth AJ, Friedman JM, Heller S (2000) Vanilloid receptor-related osmotically activated channel (VR-OAC), a candidate vertebrate osmoreceptor. Cell 103:525–535
Luo Y, Vassilev PM, Li X, Kawanabe Y, Zhou J (2003) Native polycystin 2 functions as a plasma membrane Ca2+-permeable cation channel in renal epithelia. Mol Cell Biol 23:2600–2607
McGrath J, Somlo S, Makova S, Tian X, Brueckner M (2003) Two populations of node monocilia initiate left-right asymmetry in the mouse. Cell 114:61–73
Maroto R, Raso A, Wood TG, Kurosky A, Martinac B, Hamill OP (2005) TRPC1 forms the stretch-activated cation channel in vertebrate cells. Nat Cell Biol 7:179–185
Mengerink KJ, Moy GW, Vacquier VD (2000) suREJ proteins: new signalling molecules in sea urchin spermatozoa. Zygote 8:S28–S30
Montell C et al (2002) A unified nomenclature for the superfamily of TRP cation channels. Mol Cell 9:229–231
Moy GW, Mendoza LM, Schulz JR, Swanson WJ, Glabe CG, Vacquier VD (1996) The sea urchin sperm receptor for egg jelly is a modular protein with extensive homology to the human polycystic kidney disease protein, PKD1. J Cell Biol 133:809–817
Nauli SM, Alenghat FJ, Luo Y, Williams E, Vassilev P, Li X, Elia AE, Lu W, Brown EM, Quinn SJ, Ingber DE, Zhou J (2003) Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet 33:129–137
Neill AT, Moy GW, Vacquier VD (2004) Polycystin-2 associates with the polycystin-1 homolog, suREJ3, and localizes to the acrosomal region of sea urchin spermatozoa. Mol Reprod Dev 67:472–477
Newby LJ, Streets AJ, Zhao Y, Harris PC, Ward CJ, Ong AC (2002) Identification, characterization, and localization of a novel kidney polycystin-1- polycystin-2 complex. J Biol Chem 277:20763–20773
Ong AC, Wheatley DN (2003) Polycystic kidney disease-the ciliary connection. Lancet 361:774–776
Parnell SC, Magenheimer BS, Maser RL, Rankin CA, Smine A, Okamoto T, Calvet JP (1998) The polycystic kidney disease-1 protein, polycystin-1, binds and activates heterotrimeric G-proteins in vitro. Biochem Biophys Res Commun 251:625–631
Parnell SC, Magenheimer BS, Maser RL, Zien CA, Frischauf AM, Calvet JP (2002) Polycystin-1 activation of c-Jun N-terminal kinase and AP-1 is mediated by heterotrimeric G proteins. J Biol Chem 277:19566–19572
Pazour GJ, San Agustin JT, Follit JA, Rosenbaum JL, Witman GB (2002) Polycystin-2 localizes to the kidney cilia and the ciliary level is elevated in ORPK mice with polycystic kidney disease. Curr Biol 12:R378–R380
Pennekamp P, Karcher C, Fischer A, Schweickert A, Skryabin B, Horst J, Blum M, Dworniczak B (2002) The ion channel polycystin-2 is required for left–right axis determination in mice. Curr Biol 12:938–943
Praetorius HA, Frokiaer J, Leipziger J (2005) Transepithelial pressure pulses induce nucleotide release in polarized MDCK cells. Am J Physiol 288:F133–F141
Praetorius HA, Spring KR (2001) Bending the MDCK cell primary cilium increases intracellular calcium. J Membr Biol 184:71–79
Praetorius HA, Spring KR (2003) Removal of the MDCK cell primary cilium abolishes flow sensing. J Membr Biol 191:69–76
Praetorius HA, Spring KR (2003) The renal cell primary cilium functions as a flow sensor. Curr Opin Nephrol Hypertens 12:517–520
Puri S, Magenheimer BS, Maser RL, Ryan EM, Zien CA, Walker DD, Wallace DP, Hempson SJ, Calvet JP (2004) Polycystin-1 activates the calcineurin/NFAT (nuclear factor of activated T-cells) signaling pathway. J Biol Chem 279:55455–55464
Qian F, Boletta A, Bhunia AK, Xu H, Liu L, Ahrabi AK, Watnick TJ, Zhou F, Germino GG (2002) Cleavage of polycystin-1 requires the receptor for egg jelly domain and is disrupted by human autosomal-dominant polycystic kidney disease 1-associated mutations. Proc Natl Acad Sci USA 99:16981–16986
Roitbak T, Ward CJ, Harris PC, Bacallao R, Ness SA, Wandinger-Ness A (2004) A polycystin-1 multiprotein complex is disrupted in polycystic kidney disease cells. Mol Biol Cell 15:1334–1346
Scheffers MS, van der Bent P, Prins F, Spruit L, Breuning MH, Litvinov SV, de Heer E, Peters DJ (2000) Polycystin-1, the product of the polycystic kidney disease 1 gene, co-localizes with desmosomes in MDCK cells. Hum Mol Genet 9:2743–2750
Scheffers MS, van der Bent P, van de Wal A, van Eendenburg J, Breuning MH, de Heer E, Peters DJ (2004) Altered distribution and co-localization of polycystin-2 with polycystin-1 in MDCK cells after wounding stress. Exp Cell Res 292:219–230
Stacey M, Lin HH, Gordon S, McKnight AJ (2000). LNB-TM7, a group of seven-transmembrane proteins related to family-B G-protein-coupled receptors. Trends Biochem Sci 25:284–289
Story GM, Peier AM, Reeve AJ, Eid SR, Mosbacher J, Hricik TR, Earley TJ, Hergarden AC, Andersson DA, Hwang SW, McIntyre P, Jegla T, Bevan S, Patapoutian A (2003) ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 112:819–829
Sutters M, Germino GG (2003) Autosomal dominant polycystic kidney disease: molecular genetics and pathophysiology. J Lab Clin Med 141:91–101
Sutters M, Yamaguchi T, Maser RL, Magenheimer BS, St John PL, Abrahamson DR, Grantham JJ (2001) Polycystin-1 transforms the cAMP growth-responsive phenotype of M-1 cells. Kidney Int 60:484–494
Syntichaki P, Tavernarakis N (2004) Genetic models of mechanotransduction: the nematode Caenorhabditis elegans. Physiol Rev 84:1097–1153
Torres VE, Harris PC (2003) Autosomal dominant polycystic kidney disease. Nefrologia 23:14–22
Torres VE, Wang X, Qian Q, Somlo L, Harris PC, Gattone VH (2004) Effective treatment of an orthologous model of autosomal dominant polycystic kidney disease. Nat Med 10:363–364
Tsiokas L, Arnould T, Zhu C, Kim E, Walz G, Sukhatme VP (1999) Specific association of the gene product of PKD2 with the TRPC1 channel. Proc Natl Acad Sci USA 96:3934–3939
Vassilev PM, Guo L, Chen XZ, Segal Y, Peng JB, Basora N, Babakhanlou H, Cruger G, Kanazirska M, Ye C, Brown EM, Hediger MA, Zhou J (2001) Polycystin-2 is a novel cation channel implicated in defective intracellular Ca2+ homeostasis in polycystic kidney disease. Biochem Biophys Res Commun 282:341–350
Watnick TJ, Jin Y, Matunis E, Kernan MJ, Montell C (2003) A flagellar polycystin-2 homolog required for male fertility in Drosophila. Curr Biol 13:2179–2184
Wu G, D’Agati V, Cai Y, Markowitz G, Park JH, Reynolds DM, Maeda Y, Le TC, Hou H Jr, Kucherlapati R, Edelmann W, Somlo S (1998) Somatic inactivation of Pkd2 results in polycystic kidney disease. Cell 93:177–188
Wu G, Somlo S (2000) Molecular genetics and mechanism of autosomal dominant polycystic kidney disease. Mol Genet Metab 69:1–15
Xu GM, Gonzalez-Perrett S, Essafi M, Timpanaro GA, Montalbetti N, Arnaout MA, Cantiello HF (2003) Polycystin-1 activates and stabilizes the polycystin-2 channel. J Biol Chem 278:1457–1462
Xu GM, Sikaneta T, Sullivan BM, Zhang Q, Andreucci M, Stehle T, Drummond I, Arnaout MA (2001) Polycystin-1 interacts with intermediate filaments. J Biol Chem 276:46544–46552
Yamaguchi T et al (2003) Cyclic AMP activates B-raf and ERK in cyst epithelial cells from autosomal-dominant polycystic kidneys. Kidney Int 63:1983–1994
Yamaguchi T, Wallace DP, Magenheimer BS, Hempson SJ, Grantham JJ, Calvet JP (2004) Calcium restriction allows cAMP activation of the B-Raf/ERK pathway, switching cells to a cAMP-dependent growth-stimulated phenotype. J Biol Chem 279:40419–40430
Yoder BK, Hou X, Guay-Woodford LM (2002) The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J Am Soc Nephrol 13:2508–2516
Yuasa T, Takakura A, Denker BM, Venugopal B, Zhou J (2004) Polycystin-1L2 is a novel G-protein-binding protein. Genomics 84:126–138
Acknowledgement
This work was supported by the Centre National de la Recherche Scientifique (CNRS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Delmas, P. Polycystins: polymodal receptor/ion-channel cellular sensors. Pflugers Arch - Eur J Physiol 451, 264–276 (2005). https://doi.org/10.1007/s00424-005-1431-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-005-1431-5