Abstract
Background
The Richalet hypoxia sensitivity test (RT), which quantifies the cardiorespiratory response to acute hypoxia during exercise at an intensity corresponding to a heart rate of ~130 bpm in normoxia, can predict susceptibility of altitude sickness. Its ability to predict exercise performance in hypoxia is unknown.
Objectives
Investigate: (1) whether cerebral blood flow (CBF) and cerebral tissue oxygenation (O2Hb; oxygenated hemoglobin, HHb; deoxygenated hemoglobin) responses during RT predict time-trial cycling (TT) performance in severe hypoxia; (2) if subjects with blunted cardiorespiratory responses during RT show greater impairment of TT performance in severe hypoxia.
Study design
Thirteen men [27 ± 7 years (mean ± SD), Wmax: 385 ± 30 W] were evaluated with RT and the results related to two 15 km TT, in normoxia and severe hypoxia (FIO2 = 0.11).
Results
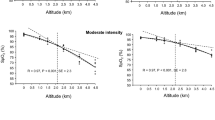
During RT, mean middle cerebral artery blood velocity (MCAv: index of CBF) was unaltered with hypoxia at rest (p > 0.05), while it was increased during normoxic (+22 ± 12 %, p < 0.05) and hypoxic exercise (+33 ± 17 %, p < 0.05). Resting hypoxia lowered cerebral O2Hb by 2.2 ± 1.2 μmol (p < 0.05 vs. resting normoxia); hypoxic exercise further lowered it to −7.6 ± 3.1 μmol below baseline (p < 0.05). Cerebral HHb, increased by 3.5 ± 1.8 μmol in resting hypoxia (p < 0.05), and further to 8.5 ± 2.9 μmol in hypoxic exercise (p < 0.05). Changes in CBF and cerebral tissue oxygenation during RT did not correlate with TT performance loss (R = 0.4, p > 0.05 and R = 0.5, p > 0.05, respectively), while tissue oxygenation and SaO2 changes during TT did (R = −0.76, p < 0.05). Significant correlations were observed between SaO2, MCAv and HHb during RT (R = −0.77, −0.76 and 0.84 respectively, p < 0.05 in all cases).
Conclusions
CBF and cerebral tissue oxygenation changes during RT do not predict performance impairment in hypoxia. Since the changes in SaO2 and brain HHb during the TT correlated with performance impairment, the hypothesis that brain oxygenation plays a limiting role for global exercise in conditions of severe hypoxia remains to be tested further.







Similar content being viewed by others
References
Ainslie PN, Ogoh S (2010) Regulation of cerebral blood flow in mammals during chronic hypoxia: a matter of balance. Exp Physiol 95:251–262
Amann M, Kayser B (2009) Nervous System Function during Exercise in Hypoxia. High Alt Med Biol 10:149–164
Amann M, Romer LM, Subudhi AW et al (2007) Severity of arterial hypoxaemia affects the relative contributions of peripheral muscle fatigue to exercise performance in healthy humans. J Physiol 581:389–403
Bernardi L, Schneider A, Pomidori L et al (2006) Hypoxic ventilatory response in successful extreme altitude climbers. Eur Respir J 27:165–171
Bourdillon N, Perrey S (2012) What does cerebral oxygenation tell us about central motor output? Neuroimaging. Cogn Clin Neurosci. doi:10.5772/23609
Bourdillon N, Mollard P, Letournel M et al (2009) Interaction between hypoxia and training on NIRS signal during exercise: contribution of a mathematical model. Respir Physiol Neurobiol 169:50–61
Brugniaux JV, Hodges ANH, Hanly PJ, Poulin MJ (2007) Cerebrovascular responses to altitude. Respir Physiol Neurobiol 158:212–223
Calbet JAL, Lundby C (2009) Air to muscle O 2 delivery during exercise at altitude. High Alt Med Biol 10:123–134
Calbet JAL, Robach P, Lundby C (2009) The exercising heart at altitude. Cell Mol Life Sci 66:3601–3613
Chapman RF, Emery M, Stager JM (1999) Degree of arterial desaturation in normoxia influences VO2max decline in mild hypoxia. Med Sci Sports Exerc 31:658–663
Ekblom B, Huot R, Stein EM, Thorstensson AT (1975) Effect of changes in arterial oxygen content on circulation and physical performance. J Appl Physiol 39:71–75
Fan J-L, Kayser B (2013) The effect of adding CO2 to hypoxic inspired gas on cerebral blood flow velocity and breathing during incremental exercise. PLoS One 8:e81130
Fan J-L, Bourdillon N, Kayser B (2013) Effect of end-tidal CO2 clamping on cerebrovascular function, oxygenation, and performance during 15-km time trial cycling in severe normobaric hypoxia: the role of cerebral O2 delivery. Physiol Rep 1:e00066
Ferretti G, Moia C, Thomet JM, Kayser B (1997) The decrease of maximal oxygen consumption during hypoxia in man: a mirror image of the oxygen equilibrium curve. J Physiol 498(Pt 1):231–237
Gore CJ, Hahn AG, Scroop GC et al (1996) Increased arterial desaturation in trained cyclists during maximal exercise at 580 m altitude. J Appl Physiol 80:2204–2210
Hackett PH, Rennie D, Levine HD (1976) The incidence, importance, and prophylaxis of acute mountain sickness. Lancet 2:1149–1155
Hornbein TF, Townes BD, Schoene RB et al (1989) The cost to the central nervous system of climbing to extremely high altitude. N Engl J Med 321:1714–1719
Ide K, Horn A, Secher NH (1999) Cerebral metabolic response to submaximal exercise. J Appl Physiol 87:1604–1608
Jørgensen LG, Perko G, Secher NH (1992) Regional cerebral artery mean flow velocity and blood flow during dynamic exercise in humans. J Appl Physiol 73:1825–1830
Kayser B (2003) Exercise starts and ends in the brain. Eur J Appl Physiol 90:411–419
Kayser B, Narici M, Binzoni T et al (1994) Fatigue and exhaustion in chronic hypobaric hypoxia: influence of exercising muscle mass. J Appl Physiol 76:634–640
Kleinschmidt A, Obrig H, Requardt M et al (1996) Simultaneous recording of cerebral blood oxygenation changes during human brain activation by magnetic resonance imaging and near-infrared spectroscopy. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 16:817–826
Koglin L, Kayser B (2013) Control and sensation of breathing during cycling exercise in hypoxia under naloxone: a randomised controlled crossover trial. Extrem Physiol Med 2:1
Lawler J, Powers SK, Thompson D (1988) Linear relationship between VO2max and VO2max decrement during exposure to acute hypoxia. J Appl Physiol 64:1486–1492
Lindegaard KF, Lundar T, Wiberg J et al (1987) Variations in middle cerebral artery blood flow investigated with noninvasive transcranial blood velocity measurements. Stroke J Cereb Circ 18:1025–1030
Madsen PL, Sperling BK, Warming T et al (1993) Middle cerebral artery blood velocity and cerebral blood flow and O2 uptake during dynamic exercise. J Appl Physiol 74:245–250
Martin D, O’Kroy J (1993) Effects of acute hypoxia on the VO2 max of trained and untrained subjects. J Sports Sci 11:37–42
Nishimura N, Iwasaki K, Ogawa Y, Aoki K (2010) Decreased steady-state cerebral blood flow velocity and altered dynamic cerebral autoregulation during 5-h sustained 15% O2 hypoxia. J Appl Physiol 108:1154–1161
Peltonen JE, Kowalchuk JM, Paterson DH et al (2007) Cerebral and muscle tissue oxygenation in acute hypoxic ventilatory response test. Respir Physiol Neurobiol 155:71–81
Peltonen JE, Paterson DH, Shoemaker JK et al (2009) Cerebral and muscle deoxygenation, hypoxic ventilatory chemosensitivity and cerebrovascular responsiveness during incremental exercise. Respir Physiol Neurobiol 169:24–35
Rasmussen P, Nielsen J, Overgaard M et al (2010) Reduced muscle activation during exercise related to brain oxygenation and metabolism in humans. J Physiol 588:1985–1995
Richalet JP, Rathat C, Keromes A et al (1983) Plasma volume, body weight, and acute mountain sickness. Lancet 1:525
Richalet J-P, Herry J-P, Blein J-P (2006) Médecine de l’alpinisme et des sports de montagne. Masson, Paris, pp 258–260
Richalet J-P, Larmignat P, Poitrine E et al (2012) Physiological risk factors for severe high-altitude illness: a prospective cohort study. Am J Respir Crit Care Med 185:192–198
Roach RC, Kayser B (2001) Exercise and hypoxia: performance, limits, and training. High Alt. Explor. Hum. Adapt., Marcel Dekker. T. Hornbein and R. B. Schoene, New York, pp 663–705
Roy SB, Guleria JS, Khanna PK et al (1968) Immediate circulatory response to high altitude hypoxia in man. Nature 217:1177–1178
Rupp T, Perrey S (2008) Prefrontal cortex oxygenation and neuromuscular responses to exhaustive exercise. Eur J Appl Physiol 102:153–163
Sato K, Sadamoto T, Hirasawa A et al (2012) Differential blood flow responses to CO2 in human internal and external carotid and vertebral arteries. J Physiol 590:3277–3290
Schoene RB, Lahiri S, Hackett PH et al (1984) Relationship of hypoxic ventilatory response to exercise performance on Mount Everest. J Appl Physiol 56:1478–1483
Seifert T, Fisher JP, Young CN et al (2010) Glycopyrrolate abolishes the exercise-induced increase in cerebral perfusion in humans. Exp Physiol 95:1016–1025
Siggaard-Andersen O (1974) The acid-base status of the blood. Williams and Wilkins, Baltimore
Siggaard-Andersen O (2003) OSA.exe. Charlottenlund, Denmark
Smith KJ, Wong LE, Eves ND et al (2012) Regional cerebral blood flow distribution during exercise: influence of oxygen. Respir Physiol Neurobiol 184:97–105
Smith ZM, Krizay E, Guo J et al (2013) Sustained high-altitude hypoxia increases cerebral oxygen metabolism. J Appl Physiol 114:11–18
Strangman G, Culver JP, Thompson JH, Boas DA (2002) A quantitative comparison of simultaneous BOLD fMRI and NIRS recordings during functional brain activation. Neuroimage 17:719–731
Subudhi AW, Dimmen AC, Roach RC (2007) Effects of acute hypoxia on cerebral and muscle oxygenation during incremental exercise. J Appl Physiol 103:177–183
Subudhi AW, Lorenz MC, Fulco CS, Roach RC (2008) Cerebrovascular responses to incremental exercise during hypobaric hypoxia: effect of oxygenation on maximal performance. Am J Physiol Heart Circ Physiol 294:H164–H171
Subudhi AW, Miramon BR, Granger ME, Roach RC (2009) Frontal and motor cortex oxygenation during maximal exercise in normoxia and hypoxia. J Appl Physiol 106:1153–1158
Subudhi AW, Olin JT, Dimmen AC et al (2011) Does cerebral oxygen delivery limit incremental exercise performance? J Appl Physiol 111:1727–1734
Tew GA, Ruddock AD, Saxton JM (2010) Skin blood flow differentially affects near-infrared spectroscopy-derived measures of muscle oxygen saturation and blood volume at rest and during dynamic leg exercise. Eur J Appl Physiol 110:1083–1089
Van der Zee P, Cope M, Arridge SR et al (1992) Experimentally measured optical pathlengths for the adult head, calf and forearm and the head of the newborn infant as a function of inter optode spacing. Adv Exp Med Biol 316:143–153
Van Dorp E, Los M, Dirven P et al (2007) Inspired carbon dioxide during hypoxia: effects on task performance and cerebral oxygen saturation. Aviat Space Environ Med 78:666–672
Verges S, Rupp T, Jubeau M et al (2012) Cerebral perturbations during exercise in hypoxia. Am J Physiol Regul Integr Comp Physiol 302:R903–R916
Woorons X, Mollard P, Pichon A et al (2007) Moderate exercise in hypoxia induces a greater arterial desaturation in trained than untrained men. Scand J Med Sci Sports 17:431–436
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Carsten Lundby.
Rights and permissions
About this article
Cite this article
Bourdillon, N., Fan, JL. & Kayser, B. Cerebral oxygenation during the Richalet hypoxia sensitivity test and cycling time-trial performance in severe hypoxia. Eur J Appl Physiol 114, 1037–1048 (2014). https://doi.org/10.1007/s00421-014-2835-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-014-2835-8