Abstract
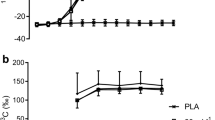
Intramyocellular lipids (IMCL) and muscle glycogen provide local energy during exercise (EX). The objective of this study was to clarify the role of high versus low IMCL levels at equal initial muscle glycogen on fuel selection during EX. After 3 h of depleting exercise, 11 endurance-trained males consumed in a crossover design a high-carbohydrate (7 g kg−1 day−1) low-fat (0.5 g kg−1 day−1) diet (HC) for 2.5 days or the same diet with 3 g kg−1 day−1 more fat provided during the last 1.5 days of diet (four meals; HCF). Respiratory exchange, thigh muscle substrate breakdown by magnetic resonance spectroscopy, and plasma FFA oxidation ([1-13C]palmitate) were measured during EX (3 h, 50% W max). Pre-EX IMCL concentrations were 55% higher after HCF. IMCL utilization during EX in HCF was threefold greater compared with HC (P < 0.001) and was correlated with aerobic power and highly correlated (P < 0.001) with initial content. Glycogen values and decrements during EX were similar. Whole-body fat oxidation (Fatox) was similar overall and plasma FFA oxidation smaller (P < 0.05) during the first EX hour after HCF. Myocellular fuels contributed 8% more to whole-body energy demands after HCF (P < 0.05) due to IMCL breakdown (27% Fatox). After EX, when both IMCL and glycogen concentrations were again similar across trials, a simulated 20-km time-trial showed no difference in performance between diets. In conclusion, IMCL concentrations can be increased during a glycogen loading diet by consuming additional fat for the last 1.5 days. During subsequent exercise, IMCL decrease in proportion to their initial content, partly in exchange for peripheral fatty acids.







Similar content being viewed by others
References
Achten J, Venables MC, Jeukendrup AE (2003) Fat oxidation rates are higher during running compared with cycling over a wide range of intensities. Metabolism 52:747–752
Achten J, Halson SL, Moseley L, Rayson MP, Casey A, Jeukendrup AE (2004) Higher dietary carbohydrate content during intensified running training results in better maintenance of performance and mood state. J Appl Physiol 96:1331–1340
Bachmann OP, Dahl DB, Brechtel K, Machann J, Haap M, Maier T, Loviscach M, Stumvoll M, Claussen CD, Schick F, Haring HU, Jacob S (2001) Effects of intravenous and dietary lipid challenge on intramyocellular lipid content and the relation with insulin sensitivity in humans. Diabetes 50:2579–2584
Boden G, Lebed B, Schatz M, Homko C, Lemieux S (2001) Effects of acute changes of plasma free fatty acids on intramyocellular fat content and insulin resistance in healthy subjects. Diabetes 50:1612–1617
Boesch C, Slotboom J, Hoppeler H, Kreis R (1997) In vivo determination of intra-myocellular lipids in human muscle by means of localized 1H-MR-spectroscopy. Magn Reson Med 37:484–493
Boesch C, Décombaz J, Slotboom J, Kreis R (1999) Observation of intramyocellular lipids by means of 1H magnetic resonance spectroscopy. Proc Nutr Soc 58:841–850
Bottomley PA, Lee Y, Weiss RG (1997) Total creatine in muscle: imaging and quantification with proton MR spectroscopy. Radiology 204:403–410
Burke LM, Angus DJ, Cox GR, Cummings NK, Febbraio MA, Gawthorn K, Hawley JA, Minehan M, Martin DT, Hargreaves M (2000) Effect of fat adaptation and carbohydrate restoration on metabolism and performance during prolonged cycling. J Appl Physiol 89:2413–2421
Carey AL, Staudacher HM, Cummings NK, Stepto NK, Nikolopoulos V, Burke LM, Hawley JA (2001) Effects of fat adaptation and carbohydrate restoration on prolonged endurance exercise. J Appl Physiol 91:115–122
Christ ER, Zehnder M, Boesch C, Trepp R, Mullis PE, Diem P, Decombaz J (2006) The effect of increased lipid intake on hormonal responses during aerobic exercise in endurance-trained men. Eur J Endocrinol 154:397–403
Clarkson PM, Kroll W, McBride TC (1980) Maximal isometric strength and fiber type composition in power and endurance athletes. Eur J Appl Physiol Occup Physiol 44:35–42
Coggan AR, Raguso CA, Gastaldelli A, Sidossis LS, Yeckel CW (2000) Fat metabolism during high-intensity exercise in endurance-trained and untrained men. Metabolism 49:122–128
Coron A, Vanhamme L, Antoine JP, Van Hecke P, Van Huffel S (2001) The filtering approach to solvent peak suppression in MRS: a critical review. J Magn Reson 152:26–40
Coyle EF, Jeukendrup AE, Wagenmakers AJ, Saris WH (1997) Fatty acid oxidation is directly regulated by carbohydrate metabolism during exercise. Am J Physiol 273:E268–E275
Coyle EF, Jeukendrup AE, Oseto MC, Hodgkinson BJ, Zderic TW (2001) Low-fat diet alters intramuscular substrates and reduces lipolysis and fat oxidation during exercise. Am J Physiol Endocrinol Metab 280:E391–E398
De Bock K, Richter EA, Russell AP, Eijnde BO, Derave W, Ramaekers M, Koninckx E, Leger B, Verhaeghe J, Hespel P (2005) Exercise in the fasted state facilitates fibre type-specific intramyocellular lipid breakdown and stimulates glycogen resynthesis in humans. J Physiol 564:649–660
Décombaz J, Fleith M, Hoppeler H, Kreis R, Boesch C (2000) Effect of diet on the replenishment of intramyocellular lipids after exercise. Eur J Nutr 39:244–247
Décombaz J, Schmitt B, Ith M, Decarli B, Diem P, Kreis R, Hoppeler H, Boesch C (2001) Postexercise fat intake repletes intramyocellular lipids but no faster in trained than in sedentary subjects. Am J Physiol Regul Integr Comp Physiol 281:R760–R769
Erlenbusch M, Haub M, Munoz K, MacConnie S, Stillwell B (2005) Effect of high-fat or high-carbohydrate diets on endurance exercise: a meta-analysis. Int J Sport Nutr Exerc Metab 15:1–14
Flatt JP, Ravussin E, Acheson KJ, Jequier E (1985) Effects of dietary fat on postprandial substrate oxidation and on carbohydrate and fat balances. J Clin Invest 76:1019–1024
Fox EL, Mathews DK (1981) Bases physiologiques de l’activité physique. WB Saunders, Philadelphia p 210
Goedecke JH, Christie C, Wilson G, Dennis SC, Noakes TD, Hopkins WG, Lambert EV (1999) Metabolic adaptations to a high-fat diet in endurance cyclists. Metab Clin Exp 48:1509–1517
Havemann L, West SJ, Goedecke JH, Macdonald IA, St Clair GA, Noakes TD, Lambert EV (2006) Fat adaptation followed by carbohydrate loading compromises high-intensity sprint performance. J Appl Physiol 100:194–202
Helge JW, Watt PW, Richter EA, Rennie MJ, Kiens B (2002) Partial restoration of dietary fat induced metabolic adaptations to training by 7 days of carbohydrate diet. J Appl Physiol 93:1797–1805
Ivy JL (2001) Dietary strategies to promote glycogen synthesis after exercise. Can J Appl Physiol 26 Suppl:S236–S245
Jansson E, Hjemdahl P, Kaijser L (1982) Diet induced changes in sympatho-adrenal activity during submaximal exercise in relation to substrate utilization in man. Acta Physiol Scand 114:171–178
Jéquier E, Acheson K, Schutz Y (1987) Assessment of energy expenditure and fuel utilization in man. Annu Rev Nutr 7:187–208
Jeukendrup A, Saris WH, Brouns F, Kester AD (1996) A new validated endurance performance test. Med Sci Sports Exerc 28:266–270
Jeukendrup AE, Saris WH, Wagenmakers AJ (1998) Fat metabolism during exercise: a review. Part I: fatty acid mobilization and muscle metabolism. Int J Sports Med 19:231–244
Johnson NA, Stannard SR, Mehalski K, Trenell MI, Sachinwalla T, Thompson CH, Thompson MW (2003) Intramyocellular triacylglycerol in prolonged cycling with high- and low-carbohydrate availability. J Appl Physiol 94:1365–1372
Johnson NA, Stannard SR, Rowlands DS, Chapman PG, Thompson CH, O’Connor H, Sachinwalla T, Thompson MW (2006) Effect of short-term starvation vs. high-fat diet on intramyocellular triglyceride accumulation and insulin resistance in physically fit humans. Exp Physiol Apr 20 [Epub ahead of print]
Larson-Meyer DE, Newcomer BR, Hunter GR (2002) Influence of endurance running and recovery diet on intramyocellular lipid content in women: a 1H NMR study. Am J Physiol Endocrinol Metab 282:E95–E106
Naressi A, Couturier C, Devos JM, Janssen M, Mangeat C, de Beer R, Graveron-Demilly D (2001) Java-based graphical user interface for the MRUI quantitation package. MAGMA 12:141–152
Noble BJ (1982) Clinical applications of perceived exertion. Med Sci Sports Exerc 14:406–411
Okano G, Sato Y, Murata Y (1998) Effect of elevated blood FFA levels on endurance performance after a single fat meal ingestion. Med Sci Sports Exerc 30:763–768
Roepstorff C, Steffensen CH, Madsen M, Stallknecht B, Kanstrup IL, Richter EA, Kiens B (2002) Gender differences in substrate utilization during submaximal exercise in endurance-trained subjects. Am J Physiol Endocrinol Metab 282:E435–E447
Roepstorff C, Donsmark M, Thiele M, Vistisen B, Stewart G, Vissing K, Schjerling P, Hardie DG, Galbo H, Kiens B (2006) Gender differences in hormone-sensitive lipase expression, activity and phosphorylation in skeletal muscle at rest and during exercise. Am J Physiol Endocrinol Metab July 5 [Epub ahead of print]
Romijn JA, Coyle EF, Sidossis LS, Gastaldelli A, Horowitz JF, Endert E, Wolfe RR (1993) Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am J Physiol 265:E380–E391
Rotman S, Slotboom J, Kreis R, Boesch C, Jequier E (2000) Muscle glycogen recovery after exercise measured by 13C-magnetic resonance spectroscopy in humans: effect of nutritional solutions. MAGMA 11:114–121
Sacchetti M, Saltin B, Osada T, van Hall G (2002) Intramuscular fatty acid metabolism in contracting and non-contracting human skeletal muscle. J Physiol 540:387–395
Schneider DA, Lacroix KA, Atkinson GR, Troped PJ, Pollack J (1990) Ventilatory threshold and maximal oxygen uptake during cycling and running in triathletes. Med Sci Sports Exerc 22:257–264
Sidossis LS, Wolfe RR (1996) Glucose and insulin-induced inhibition of fatty acid oxidation: the glucose-fatty acid cycle reversed. Am J Physiol 270:E733–E738
Sidossis LS, Coggan AR, Gastaldelli A, Wolfe RR (1995) A new correction factor for use in tracer estimations of plasma fatty acid oxidation. Am J Physiol 269:E649–E656
Sinning WE, Dolny DG, Little KD, Cunningham LN, Racaniello A, Siconolfi SF, Sholes JL (1985) Validity of “generalized” equations for body composition analysis in male athletes. Med Sci Sports Exerc 17:124–130
Souci SW et al (2002) Food composition and nutrition tables, 6th edn. Medpharm Scientific Publishers, Stuttgart
Standl E, Lotz N, Dexel T, Janka HU, Kolb HJ (1980) Muscle triglycerides in diabetic subjects. Effect of insulin deficiency and exercise. Diabetologia 18:463–469
Starling RD, Trappe TA, Parcell AC, Kerr CG, Fink WJ, Costill DL (1997) Effects of diet on muscle triglyceride and endurance performance. J Appl Physiol 82:1185–1189
Steele R (1959) Influences of glucose loading and of injected insulin on hepatic glucose output. Ann NY Acad Sci 82:420–430
Steffensen CH, Roepstorff C, Madsen M, Kiens B (2002) Myocellular triacylglycerol breakdown in females but not in males during exercise. Am J Physiol Endocrinol Metab 282:E634–E642
Stellingwerff T, Spriet LL, Watt MJ, Kimber NE, Hargreaves M, Hawley JA, Burke LM (2006) Decreased PDH activation and glycogenolysis during exercise following fat adaptation with carbohydrate restoration. Am J Physiol Endocrinol Metab 290:E380–E388
Stepto NK, Carey AL, Staudacher HM, Cummings NK, Burke LM, Hawley JA (2002) Effect of short-term fat adaptation on high-intensity training. Med Sci Sports Exerc 34:449–455
van Hall G, Gonzalez-Alonso J, Sacchetti M, Saltin B (1999) Skeletal muscle substrate metabolism during exercise: methodological considerations. Proc Nutr Soc 58:899–912
Vanhamme L, van den Boogaart A, Van Huffel S (1997) Improved method for accurate and efficient quantification of MRS data with use of prior knowledge. J Magn Reson 129:35–43
van Loon LJC (2004) Use of intramuscular triacylglycerol as a substrate source during exercise in humans. J Appl Physiol 97:1170–1187
van Loon LJ, Koopman R, Stegen JH, Wagenmakers AJ, Keizer HA, Saris WH (2003a) Intramyocellular lipids form an important substrate source during moderate intensity exercise in endurance-trained males in a fasted state. J Physiol 553:611–625
van Loon LJ, Schrauwen-Hinderling VB, Koopman R, Wagenmakers AJ, Hesselink MK, Schaart G, Kooi ME, Saris WH (2003b) Influence of prolonged endurance cycling and recovery diet on intramuscular triglyceride content in trained males. Am J Physiol Endocrinol Metab 285:E804–E811
Watt MJ, Heigenhauser GJ, Dyck DJ, Spriet LL (2002a) Intramuscular triacylglycerol, glycogen and acetyl group metabolism during 4 h of moderate exercise in man. J Physiol 541:969–978
Watt MJ, Heigenhauser GJ, Spriet LL (2002b) Intramuscular triacylglycerol utilization in human skeletal muscle during exercise: is there a controversy? J Appl Physiol 93:1185–1195
Watt MJ, Holmes AG, Steinberg GR, Mesa JL, Kemp BE, Febbraio MA (2004) Reduced plasma FFA availability increases net triacylglycerol degradation, but not GPAT or HSL activity, in human skeletal muscle. Am J Physiol Endocrinol Metab 287:E120–E127
White LJ, Robergs RA, Sibbitt WL Jr, Ferguson MA, McCoy S, Brooks WM (2003) Effects of intermittent cycle exercise on intramyocellular lipid use and recovery. Lipids 38:9–13
Wolfe RR (1992) Radioactive and stable isotope traces in biomedicine. Principles and practice of kinetic analysis. Wiley-Liss, New York
Zderic TW, Davidson CJ, Schenk S, Byerley LO, Coyle EF (2004) High-fat diet elevates resting intramuscular triglyceride concentration and whole body lipolysis during exercise. Am J Physiol Endocrinol Metab 286:E217–E225
Zehnder M, Saillen P, Kreis R, Boesch C, Vermathen P (2005a) Evaluation of intramyocellular lipids concentration in the different thigh and calf muscles before and after endurance exercise. Proc Intl Soc Magn Reson Med 13:798 (abstr)
Zehnder M, Ith M, Kreis R, Saris W, Boutellier U, Boesch C (2005b) Gender-specific usage of intramyocellular lipids and glycogen during exercise. Med Sci Sports Exerc 37:1517–1524
Acknowledgments
We thank the subjects of this study for their time and effort, Charles Schindler for preparing the sterile, pyrogene-free isotope solutions, Stéphane Berger, Anny Blondel, Bernard Decarli, Irina Monnard Jean-Marc Schneider, and Irène Zbinden for their skilled technical assistance. For evaluation of the glycogen spectra, the MRUI software package was kindly provided by the participants of the EU Network programs: Human Capital and Mobility, CHRX-CT94-0432 and Training and Mobility of Researchers, ERB-FMRX-CT970160. We appreciate Karin Zwygart for data acquisition. This work was supported by a grant from the Swiss National Science Foundation (3100-065315).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zehnder, M., Christ, E.R., Ith, M. et al. Intramyocellular lipid stores increase markedly in athletes after 1.5 days lipid supplementation and are utilized during exercise in proportion to their content. Eur J Appl Physiol 98, 341–354 (2006). https://doi.org/10.1007/s00421-006-0279-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-006-0279-5