Abstract
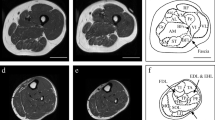
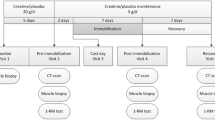
Metabolic factors have been proposed to explain strength deficits observed in skeletal muscle with immobilization that are not completely accounted for by changes in muscle cross-sectional area (CSA) and neural adaptations. The aim of this study was to quantify changes in the resting inorganic phosphate (Pi) concentration from the medial gastrocnemius muscle during immobilization, reloading and rehabilitation. Additionally, we assessed the contributions of CSA, muscle activation and Pi concentration to plantar flexor torque during rehabilitation following immobilization. Eight persons with a surgically stabilized ankle fracture participated. Subjects were immobilized for 6–8 weeks and subsequently participated in 10 weeks of rehabilitation. Localized 31P-Magnetic resonance spectroscopy, magnetic resonance imaging, isometric torque and activation testing were performed on the immobilized and uninvolved limbs. At 6 weeks of immobilization, significant differences were noted between the immobilized and uninvolved limbs for the Pi concentration and the Pi/PCr ratio (P < 0.05). From 6 weeks of immobilization to 3–5 days of reloading, the increase in Pi concentration (15%, P = 0.26) and Pi/PCr (20%, P = 0.29) was not significant. During rehabilitation, the relative contributions of CSA, muscle activation and Pi concentration to plantarflexor torque were 32, 44 and 40%, respectively. Together, CSA, muscle activation and Pi concentration accounted for 76% of the variance in torque (P < 0.01). In summary, our findings suggest that immobilization, independent of reloading, leads to a significant increase in the resting Pi concentration of human skeletal muscle. Additionally, alterations in resting Pi concentration may contribute to strength deficits with immobilization not accounted for by changes in muscle CSA or neural adaptations.






Similar content being viewed by others
Notes
GE Medical Systems global headquarters: Waukesha, WI
Biodex Medical Systems, Inc., 20 Ramsay Road, Shirley, NY 11967
Grass Instruments, West Warwick, RI
SPSS Inc, 233 S. Wacker Drive, 11th floor, Chicago, IL 60606
References
Berg HE, Dudley GA, Haggmark T, Ohlsen H, Tesch PA (1991) Effects of lower limb unloading on skeletal muscle mass and function in humans. J Appl Physiol 70(4):1882–1885
Berg HE, Larsson L, Tesch PA (1997) Lower limb skeletal muscle function after 6 wk of bed rest. J Appl Physiol 82(1):182–188
Cooke R, Franks K, Luciani GB, Pate E (1988) The inhibition of rabbit skeletal muscle contraction by hydrogen ions and phosphate. J Physiol 395:77–97
Dahlstedt AJ, Katz A, Westerblad H (2001) Role of myoplasmic phosphate in contractile function of skeletal muscle: studies on creatine kinase-deficient mice. J Physiol 533(Pt 2):379–388
Dantzig JA, Goldman YE, Millar NC, Lacktis J, Homsher E (1992) Reversal of the cross-bridge force-generating transition by photogeneration of phosphate in rabbit psoas muscle fibres. J Physiol 451:247–278
Deschenes MR, Giles JA, McCoy RW, Volek JS, Gomez AL, Kraemer WJ (2002) Neural factors account for strength decrements observed after short-term muscle unloading. Am J Physiol Regul Integr Comp Physiol 282(2):R578–583
Desplanches D (1997) Structural and functional adaptations of skeletal muscle to weightlessness. Int J Sports Med 18(Suppl 4):S259–264
Duchateau J (1995) Bed rest induces neural and contractile adaptations in triceps surae. Med Sci Sports Exerc 27(12):1581–1589
Duchateau J, Hainaut K (1987) Electrical and mechanical changes in immobilized human muscle. J Appl Physiol 62(6):2168–2173
Duchateau J, Hainaut K (1990) Effects of immobilization on contractile properties, recruitment and firing rates of human motor units. J Physiol 422:55–65
Edgerton VR, Roy RR, Allen DL, Monti RJ (2002) Adaptations in skeletal muscle disuse or decreased-use atrophy. Am J Phys Med Rehabil 81(11 Suppl):S127–147
Elliott MA, Walter GA, Gulish H, Sadi AS, Lawson DD, Jaffe W, Insko EK, Leigh JS, Vandenborne K (1997) Volumetric measurement of human calf muscle from magnetic resonance imaging. Magma 5(2):93–98
Gallego R, Kuno M, Nunez R, Snider WD (1979) Disuse enhances synaptic efficacy in spinal mononeurones. J Physiol 291:191–205
Halkjaer-Kristensen J, Ingemann-Hansen T (1985) Wasting of the human quadriceps muscle after knee ligament injuries. Scand J Rehabil Med 13(Suppl):5–55
Harris RC, Hultman E, Nordesjo LO (1974) Glycogen, glycolytic intermediates and high-energy phosphates determined in biopsy samples of musculus quadriceps femoris of man at rest. Methods and variance of values. Scand J Clin Lab Invest 33(2):109–120
Kentish JC (1986) The effects of inorganic phosphate and creatine phosphate on force production in skinned muscles from rat ventricle. J Physiol 370:585–604
MacDougall JD, Elder GC, Sale DG, Moroz JR, Sutton JR (1980) Effects of strength training and immobilization on human muscle fibres. Eur J Appl Physiol Occup Physiol 43(1):25–34
Manabe T, Kaneko S, Kuno M (1989) Disuse-induced enhancement of Ia synaptic transmission in spinal motoneurons of the rat. J Neurosci 9(7):2455–2461
McComas AJ (1994) Human neuromuscular adaptations that accompany changes in activity. Med Sci Sports Exerc 26(12):1498–1509
Millar NC, Homsher E (1990) The effect of phosphate and calcium on force generation in glycerinated rabbit skeletal muscle fibers. A steady-state and transient kinetic study. J Biol Chem 265(33):20234–20240
Pathare N, Walter GA, Stevens JE, Yang Z, Okerke E, Gibbs JD, Esterhai JL, Scarborough MT, Gibbs CP, Sweeney HL, Vandenborne K (2005) Changes in inorganic phosphate and force production in human skeletal muscle after cast immobilization. J Appl Physiol 98(1):307–314
Ploutz-Snyder LL, Tesch PA, Hather BM, Dudley GA (1996) Vulnerability to dysfunction and muscle injury after unloading. Arch Phys Med Rehabil 77(8):773–777
Shaffer MA, Okereke E, Esterhai JL Jr, Elliott MA, Walker GA, Yim SH, Vandenborne K (2000) Effects of immobilization on plantar-flexion torque, fatigue resistance, and functional ability following an ankle fracture. Phys Ther 80(8):769–780
Shield A, Zhou S (2004) Assessing voluntary muscle activation with the twitch interpolation technique. Sports Med 34(4):253–267
Stevens JE, PN, Tillman SM, Scarborough MT, Gibbs CP, Shah P, Jayaraman A, Walter GA, Vandenborne K (2006) Relative contributions of muscle activation and muscle size to Plantarflexor torque during rehabilitation after immobilization. J Ortho Res (in press)
Stevens JE, Walter GA, Okereke E, Scarborough MT, Esterhai JL, George SZ, Kelley MJ, Tillman SM, Gibbs JD, Elliott MA, Frimel TN, Gibbs CP, Vandenborne K (2004) Muscle Adaptations with Immobilization and Rehabilitation after Ankle Fracture. Med Sci Sports Exerc 36(10):1695–1701
Taylor DJ, Bore PJ, Styles P, Gadian DG, Radda GK (1983) Bioenergetics of intact human muscle. A 31P nuclear magnetic resonance study. Mol Biol Med 1(1):77–94
Todd G, Gorman RB, Gandevia SC (2004) Measurement and reproducibility of strength and voluntary activation of lower-limb muscles. Muscle Nerve 29(6):834–842
Vandenborne K, Elliott MA, Walter GA, Abdus S, Okereke E, Shaffer M, Tahernia D, Esterhai JL (1998) Longitudinal study of skeletal muscle adaptations during immobilization and rehabilitation. Muscle Nerve 21(8):1006–1012
Vandenborne K, Walter G, Ploutz-Snyder L, Staron R, Fry A, De Meirleir K, Dudley GA, Leigh JS (1995) Energy-rich phosphates in slow and fast human skeletal muscle. Am J Physiol 268(4 Pt 1):C869–C876
Vanhamme L, van den Boogaart A, Van Huffel S (1997) Improved method for accurate, efficient quantification of MRS data with use of prior knowledge. J Mag Res, 1997 129:35–43
Walter GA (1997) Noninvasive evaluation of metabolic capacities in human skeletal muscle. Diss Abstr Int 58(03B):1163
Warren GL, Hayes DA, Lowe DA, Williams JH, Armstrong RB (1994) Eccentric contraction-induced injury in normal and hindlimb-suspended mouse soleus and EDL muscles. J Appl Physiol 77(3):1421–1430
Zanette G, Tinazzi M, Bonato C, di Summa A, Manganotti P, Polo A, Fiaschi A (1997) Reversible changes of motor cortical outputs following immobilization of the upper limb. Electroencephalogr Clin Neurophysiol 105(4):269–279
Acknowledgments
Supported in part by the National Institutes of Health, National Center for Medical Rehabilitation Research grants RO1HD37645 and RO1HD048051 to Krista Vandenborne, Ph.D., PT.
Author information
Authors and Affiliations
Corresponding author
Additional information
Preliminary reports of these experiments were presented at the Combined Sections Meeting held in New Orleans, 2005 and American College of Sports Medicine held in Nashville, June 2005. A subset of subjects’ data for plantar flexor torque, muscle cross-sectional area and muscle activation were included in a previously published article (Stevens JE 2006).
Rights and permissions
About this article
Cite this article
Pathare, N.C., Stevens, J.E., Walter, G.A. et al. Deficit in human muscle strength with cast immobilization: contribution of inorganic phosphate. Eur J Appl Physiol 98, 71–78 (2006). https://doi.org/10.1007/s00421-006-0244-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-006-0244-3