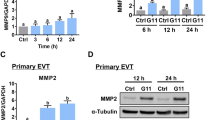
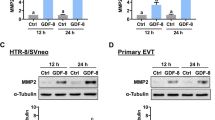
Abstract
Hyaluronan (HA) is the major glycosaminoglycan present in the extracellular matrix. It is produced by some tumours and promotes proliferation, differentiation and migration among others cellular processes. Gestational trophoblastic disease (GTD) is composed by non-tumour entities, such as hydatidiform mole (HM), which is the most common type of GTD and also malignant entities such as choriocarcinoma (CC) and placental site trophoblastic tumour (PSTT), being CC the most aggressive tumour. Although there is a growing understanding of GTD biology, the role of HA in the pathogenesis of this group of diseases remains largely unknown. The aim of this work was to study the role of HA in the pathogenesis of GTD by defining the expression pattern of HA and its receptors CD44 and RHAMM, as well as to determine if HA can modulate proliferation, differentiation and migration of CC cells. Receptors and signalling pathways involved were also analyzed. We demonstrated that HA and RHAMM are differently expressed among GTD entities and even among trophoblast subtypes. We also showed that HA is able to enhance the expression of extravillous trophoblast markers and also to induce migration of JEG-3 cells, the latter mediated by RHAMM as well as PI3K and MAPK pathways. These findings indicate a novel regulatory mechanism for CC cell biology and also contribute to the understanding of GTD pathophysiology.








Similar content being viewed by others
References
Alaniz L, García MG, Gallo-Rodriguez C, Agusti R, Sterín-Speziale N, Hajos SE, Alvarez E (2006) Hyaluronan oligosaccharides induce cell death through PI3-K/Akt pathway independently of NF- B transcription factor. Glycobiology 16:359–367
Assmann V, Marshall JF, Fieber C, Hofmann M, Hart IR (1998) The human hyaluronan receptor RHAMM is expressed as an intracellular protein in breast cancer cells. J Cell Sci 111:1685–1694
Averbeck M, Gebhardt CA, Voigt S, Beilharz S, Anderegg U, Termeer CC, Sleeman JP, Simon JC (2006) Differential regulation of hyaluronan metabolism in the epidermal and dermal compartments of human skin by UVB irradiation. J Invest Dermatol 127:687–697
Choudhary M, Zhang X, Stojković P, Hyslop L, Anyfantis G, Herbert M, Murdoch AP, Stojković M, Lako M (2007) Putative role of hyaluronan and its related genes, HAS2andRHAMM, in human early preimplantation embryogenesis and embryonic stem cell characterization. Stem Cells 25:3045–3057
Cole LA (2010) Biological functions of hCG and hCG-related molecules. Reprod Biol Endocrinol 8:102
Cordo Russo RI, Alaniz LD, Saccodossi N, Lompardía S, Blanco G, Álvarez É, García MG, Hajos SE (2010) Hyaluronan induces migration of multidrug-resistant lymphoma cell lines in vitro through Tiam1 activation by a PI3K-dependent mechanism. Leuk Res 34:1525–1532
Cordo Russo RI, Ernst G, Lompardía S, Blanco G, Álvarez É, Garcia MG, Hajos S (2012) Increased hyaluronan levels and decreased dendritic cell activation are associated with tumor invasion in murine lymphoma cell lines. Immunobiology 217:842–850
Dong Y, Arif A, Olsson M, Cali V, Hardman B, Dosanjh M, Lauer M, Midura RJ, Hascall VC, Brown KL, Johnson P (2016) Endotoxin free hyaluronan and hyaluronan fragments do not stimulate TNF-α, interleukin-12 or upregulate costimulatory molecules in dendritic cells or macrophages. Sci Rep 6:36928
Ebi H, Costa C, Faber AC, Nishtala M, Kotani H, Juric D, Della Pelle P, Song Y, Yano S, Mino-Kenudson M, Benes CH, Engelman JA (2013) PI3K regulates MEK/ERK signaling in breast cancer via the Rac-GEF, P-Rex1. Proc Natl Acad Sci USA 110:21124–21129
Ernst G, Jancic C, Auteri S, Rodriguez Moncalvo J, Galíndez F, Grynblat P, Hajos SE (2014) Increased levels of hyaluronic acid in bronchoalveolar lavage from patients with interstitial lung diseases. Relationsh Lung Funct Inflamm Cells Recruit Modern Res Inflamm 03:27–36
Gebäck T, Peter Schulz MM, Koumoutsakos P, Detmar M (2009) TScratch: a novel and simple software tool for automated analysis of monolayer wound healing assays. Biotechniques 46:265–274
Goshen R, Ariel I, Shuster S, Hochberg A, Vlodavsky I, De Groot N, Ben-Rafael Z, Stern R (1996) Hyaluronan, CD44 and its variant exons in human trophoblast invasion and placental angiogenesis. Mol Hum Reprod 2:685–691
Goueffic Y, Guilluy C, Guerin P, Patra P, Pacaud P, Loirand G (2006) Hyaluronan induces vascular smooth muscle cell migration through RHAMM-mediated PI3K-dependent Rac activation. Cardiovasc Res 72:339–348
Greiner J, Ringhoffer M, Taniguchi M, Schmitt A, Kirchner D, Krähn G, Heilmann V, Gschwend J, Bergmann L, Döhner H, Schmitt M (2002) Receptor for hyaluronan acid–mediated motility (RHAMM) is a new immunogenic leukemia-associated antigen in acute and chronic myeloid leukemia. Exp Hematol 30:1029–1035
Gust KM, Hofer MD, Perner SR, Kim R, Chinnaiyan AM, Varambally S, Moller P, Rinnab L, Rubin MA, Greiner J, Schmitt M, Kuefer R, Ringhoffer M (2009) RHAMM (CD168) is overexpressed at the protein level and may constitute an immunogenic antigen in advanced prostate cancer disease. Neoplasia 11:956–963
Hamilton SR, Fard SF, Paiwand FF, Tolg C, Veiseh M, Wang C, Mccarthy JB, Bissell MJ, Koropatnick J, Turley EA (2007) The hyaluronan receptors CD44 and rhamm (CD168) form complexes with ERK1,2 that sustain high basal motility in breast cancer cells. J Biol Chem 282:16667–16680
Hatano H, Shigeishi H, Kudo Y, Higashikawa K, Tobiume K, Takata T, Kamata N (2010) RHAMM/ERK interaction induces proliferative activities of cementifying fibroma cells through a mechanism based on the CD44–EGFR. Lab Invest 91:379–391
Huang Z, Zhao C, Chen Y, Cowell JA, Wei G, Kultti A, Huang L, Thompson CB, Rosengren S, Frost GI, Shepard HM (2014) Recombinant human hyaluronidase PH20 does not stimulate an acute inflammatory response and inhibits lipopolysaccharide-induced neutrophil recruitment in the air pouch model of inflammation. J Immunol 192:5285–5295
Jiang D, Liang J, Noble P (2011) Hyaluronan as an immune regulator in human diseases. Physiol Rev 91:221–264
Jiang J, Mohan P, Maxwell CA (2013) The cytoskeletal protein RHAMM and ERK1/2 activity mantain the pluripotency of murine embryonic stem cells. PLoS One 8:e73548
Kato HD, Terao Y, Ogawa M, Matsuda T, Arima T, Kato K, Yong Z, Wake N (2002) Growth-associated gene expression profiles by microarray analysis of trophoblast of molar pregnancies and normal villi. Int J Gynecol Pathol 21:255–260
Kouvidi K, Berdiaki A, Nikitovic D, Katonis P, Afratis N, Hascall VC, Karamanos NK, Tzanakakis GN (2011) Role of receptor for hyaluronic acid-mediated motility (RHAMM) in low molecular weight hyaluronan (LMWHA)-mediated fibrosarcoma cell adhesion. J Biol Chem 286:38509–38520
Lompardía SL, Papademetrio DL, Mascaró M, Álvarez EMDC, Hajos SE (2013) Human leukemic cell lines synthesize hyaluronan to avoid senescence and resist chemotherapy. Glycobiology 23:1463–1476
Lompardía SL, Díaz M, Papademetrio DL, Mascaró M, Pibuel M, Álvarez E, Hajos SE (2016) Hyaluronan oligomers sensitize chronic myeloid leukemia cell lines to the effect of Imatinib. Glycobiology 26:343–352
Lugli A, Iezzi G, Hostettler I, Muraro MG, Mele V, Tornillo L, Carafa V, Spagnoli G, Terracciano L, Zlobec I (2010) Prognostic impact of the expression of putative cancer stem cell markers CD133, CD166, CD44s, EpCAM, and ALDH1 in colorectal cancer. Br J Cancer 103:382–390
Mangili G, Lorusso D, Brown J, Pfisterer J, Massuger L, Vaughan M, Ngan H, Golfier F, Sekharan P, Cortés Charry R, Poveda A, Kim J, Xiang Y, Berkowtiz R, Seckl M (2014) Trophoblastic disease review for diagnosis and management. Int J Gynecol Cancer 24:109–116
Mao T-L, Kurman RJ, Huang C-C, Lin M-C, Shih I-M (2007) Immunohistochemistry of choriocarcinoma: an aid in differential diagnosis and in elucidating pathogenesis. Am J Surg Pathol 31:1726–1732
Maxwell CA, Mccarthy J, Turley E (2008) Cell-surface and mitotic-spindle RHAMM: moonlighting or dual oncogenic functions? J Cell Sci 121:925–932
Monslow J, Govindaraju P, Puré E (2015) Hyaluronan—a functional and structural sweet spot in the tissue microenvironment frontiers. Immunology 6:231
Naor D (2016) Editorial: interaction between hyaluronic acid and its receptors (CD44 RHAMM) regulates the activity of inflammation cancer. Front Immunol 7:39
Niba ETE, Nagaya H, Kanno T, Tsuchiya A, Gotoh A, Tabata C, Kuribayashi K, Nakano T, Nishizaki T (2013) Crosstalk between PI3 Kinase/PDK1/Akt/Rac1 and Ras/Raf/MEK/ERK pathways downstream PDGF receptor. Cell Physiol Biochem 31:905–913
Novakovic B, Rakyan V, Ng HK, Manuelpillai U, Dewi C, Wong NC, Morley R, Down T, Beck S, Craig JM, Saffery R (2008) Specific tumour-associated methylation in normal human term placenta and first-trimester cytotrophoblasts. Mol Hum Reprod 14:547–554
Park D, Kim Y, Kim H, Kim K, Lee Y-S, Choe J, Hahn J-H, Lee H, Jeon J, Choi C, Kim Y-M, Jeoung D (2012) Hyaluronic acid promotes angiogenesis by inducing RHAMM-TGFβ receptor interaction via CD44-PKCδ. Mol Cells 33:563–574
Red-Horse K (2004) Trophoblast differentiation during embryo implantation and formation of the maternal-fetal interface. J Clin Invest 114:744–754
Reynolds E, Dubeshter B, Katzman P, Larson K, Katzel S, Glantz C (2007) Expression of CD44, E-cadherin, and antimetastatic protein nm23-H1 in complete hydatidiform moles. Gynecol Oncol 104:451–454
Savani RC, Cao G, Pooler PM, Zaman A, Zhou Z, Delisser HM (2001) Differential involvement of the hyaluronan (HA) receptors CD44 and receptor for HA-mediated motility in endothelial cell function and angiogenesis. J Biol Chem 276:36770–36778
Sebire N (2010) Histopathological diagnosis of hydatiform mole: contemporary features and clinical implications fetal and pediatric. Pathology 29:1–16
Sebire NJ, Lindsay I (2010) Current issues in the histopathology of gestational trophoblastic tumors fetal and pediatric. Pathology 29:30–44
Shih I-M (2007) Gestational trophoblastic neoplasia—pathogenesis and potential therapeutic targets The. Lancet Oncol 8:642–650
Singer G, Kurman RJ, Mcmaster M, Shih I (2002) HLA-G immunoreactivity is specific for intermediate trophoblast in gestational trophoblastic disease and can serve as a useful marker in differential diagnosis. Am J Surg Pathol 7:914–920
Sironen RK, Tammi M, Tammi R, Auvinen PK, Anttila M, Kosma VM (2011) Hyaluronan in human malignancies. Exp Cell Res 317:383–391
Stern R, Jedrzejas M (2006) Hyaluronidases: their genomics, structures, and mechanisms of action. Chem Rev 106:818–839
Takahashi H, Takizawa T, Matsubara S, Ohkuchi A, Kuwata T, Usui R, Matsumoto H, Sato Y, Fujiwara H, Okamoto A, Suzuki M, Takizawa T (2014) Extravillous trophoblast cell invasion is promoted by the CD44-hyaluronic acid interaction. Placenta 35(3):163–170
Toole BP (2009) Hyaluronan-CD44 Interactions in Cancer: Paradoxes and Possibilities. Clin Cancer Res 15:7462–7468
Turley EA, Naor D (2012) RHAMM and CD44 peptides-analytic tools and potential drugs. Front Biosci 1:1775–1794
Turley E, Noble P, Bourguignon L (2001) Signaling properties of hyaluronan receptors. J Biol Chem 277:4589–4592
Turley EA, Wood DK, Mccarthy JB (2016) Carcinoma cell hyaluronan as a “portable” cancerized prometastatic microenvironment. Cancer Res 76:2507–2512
Twarock S, Tammi MI, Savani RC, Fischer JW (2010) Hyaluronan stabilizes focal adhesions, filopodia, and the proliferative phenotype in esophageal squamous carcinoma cells. J Biol Chem 285:23276–23284
Vargas A, Moreau J, Landry S, Lebellego F, Toufaily C, Rassart E, Lafond J, Barbeau B (2009) Syncytin-2 plays an important role in the fusion of human trophoblast cells. J Mol Biol 392:301–318
Wang C, Thor A, Moore DN, Zhao Y, Kerschmann R, Stern R, Watson P, Turley E (1998) The overexpression of RHAMM, a hyaluronan-binding protein that regulates ras signaling, correlates with overexpression of mitogen-activated protein kinase and is a significant parameter in breast cancer progression. Clin Cancer Res 4:567–576
Weigel PH, Deangelis PL (2007) Hyaluronan synthases: a decade-plus of novel glycosyltransferases. J Biol Chem 282:36777–36781
Zhang S, Chang M, Zylka D, Turley S, Harrison R, Turley E (1998) The hyaluronan receptor RHAMM regulates extracellular-regulated kinase. J Biol Chem 273:11342–11348
Zhu R, Huang YH, Tao Y, Wang SC, Sun C, Piao HL, Wang XQ, Du MR, Li DJ (2013) Hyaluronan up-regulates growth and invasion of trophoblasts in an autocrine manner via PI3K/AKT and MAPK/ERK1/2 pathways in early human pregnancy. Placenta 34:784–791
Zoller M (2015) CD44, hyaluronan, the hematopoietic stem cell, and leukemia-initiating cells frontiers. Immunology 6:235
Acknowledgements
Authors wish to thank Florencia Cappuccio for help in tissue samples selection and Flavia Sacerdoti for valuable comments and suggestions on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This work was supported by grants from University of Buenos Aires (B021 and 01/W373 to SEH) and CONICET (PIP0199 to SEH).
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
418_2017_1559_MOESM1_ESM.tif
Figure S1. p57kip2 immunohistochemistry. A) First trimester placenta. Nuclear positive expression in cytotrophoblast (CT) layer is shown (arrow). In contrast, syncytiotrophoblast (ST) was negative (asterisk). B) Complete hydatiform mole. Staining was negative for CT and ST (asterisk). Nuclear expression of extravillous trophoblast (EVT) was positive (internal control, arrow). Scale bar represents 50μm (TIF 8185 KB)
Rights and permissions
About this article
Cite this article
Mascaró, M., Pibuel, M.A., Lompardía, S.L. et al. Low molecular weight hyaluronan induces migration of human choriocarcinoma JEG-3 cells mediated by RHAMM as well as by PI3K and MAPK pathways. Histochem Cell Biol 148, 173–187 (2017). https://doi.org/10.1007/s00418-017-1559-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-017-1559-3