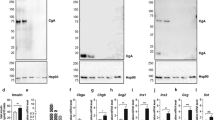
Abstract
Zymogen granules (ZG) are specialized organelles in the exocrine pancreas which allow digestive enzyme storage and regulated secretion. The molecular mechanisms of their biogenesis and the sorting of zymogens are still incompletely understood. Here, we investigated the role of proteoglycans in granule formation and secretion of zymogens in pancreatic AR42J cells, an acinar model system. Cupromeronic Blue cytochemistry and biochemical studies revealed an association of proteoglycans primarily with the granule membrane. Removal of proteoglycans by carbonate treatment led to a loss of membrane curvature indicating a supportive role in the maintenance of membrane shape and stability. Chemical inhibition of proteoglycan synthesis impaired the formation of normal electron-dense granules in AR42J cells and resulted in the formation of unusually small granule structures. These structures still contained the zymogen carboxypeptidase, a cargo molecule of secretory granules, but migrated to lighter fractions after density gradient centrifugation. Furthermore, the basal secretion of amylase was increased in AR42J cells after inhibitor treatment. In addition, irregular-shaped granules appeared in pancreatic lobules. We conclude that the assembly of a proteoglycan scaffold at the ZG membrane is supporting efficient packaging of zymogens and the proper formation of stimulus-competent storage granules in acinar cells of the pancreas.









Similar content being viewed by others
Abbreviations
- CEL:
-
Carboxyl ester lipase
- TGN:
-
Trans-Golgi network
- ZG:
-
Zymogen granule
- ZGC:
-
Zymogen granule content
- ZGM:
-
Zymogen granule membrane
References
Åbrink M, Grujic M, Pejler G (2004) Serglycin is essential for maturation of mast cell secretory granule. J Biol Chem 279:40897–40905
Allain F, Vanpouille C, Carpentier M, Slomianny M-C, Durieux S, Spik G (2002) Interaction with glycosaminoglycans is required for cyclophilin B to trigger integrin-mediated adhesion of peripheral blood T lymphocytes to extracellular matrix. PNAS 99:2714–2719
Berg NB, Young RW (1971) Sulfate metabolism in pancreatic acinar cells. J Cell Biol 50(2):469–483
Bonnemaison ML, Eipper BA, Mains RE (2013) Role of adaptor proteins in secretory granule biogenesis and maturation. Front Endocrinol 4:101
Borgonovo B, Ouwendijk J, Solimena M (2006) Biogenesis of secretory granules. Curr Opin Cell Biol 18(4):365–370
Borta H, Aroso M, Rinn C, Gomez-Lazaro M, Vitorino R et al (2010) Analysis of low abundance membrane-associated proteins from rat pancreatic zymogen granules. J Proteome Res 9(10):4927–4939
Braga T, Grujic M, Lukinius A, Hellman L, Abrink M, Pejler G (2007) Serglycin proteoglycan is required for secretory granule integrity in mucosal mast cells. Biochem J 403(1):49–57
Chen X, Ulintz PJ, Simon ES, Williams JA, Andrews PC (2008) Global topology analysis of pancreatic zymogen granule membrane proteins. Mol Cell Proteomics 7(12):2323–2336
Colomer V, Kicska GA, Rindler MJ (1996) Secretory granule content proteins and the luminal domains of granule membrane proteins aggregate in vitro at mildly acidic pH. J Biol Chem 271(1):48–55
Cronshagen U, Voland P, Kern HF (1994) cDNA cloning and characterization of a novel 16 kDa protein located in zymogen granules of rat pancreas and goblet cells of the gut. Eur J Cell Biol 65:366–377
Dartsch H, Kleene R, Kern HE (1998) In vitro condensation-sorting of enzyme proteins isolated from rat pancreatic acinar cells. Eur J Cell Biol 75(3):211–222
De Lisle RC (2002) Role of sulfated O-linked glycoproteins in zymogen granule formation. J Cell Sci 115(14):2941–2952
De Lisle RC, Norkina O, Roach E, Ziemer D (2005) Expression of pro-Muclin in pancreatic AR42J cells induces functional regulated secretory granules. Am J Physiol Cell Physiol 289(5):C1169–C1178
Dikeakos JD, Reudelhuber TL (2007) Sending proteins to dense core secretory granules: still a lot to sort out. J Cell Biol 177(2):191–196
Dvorak AM, Morgan ES (1998) Ribonuclease-gold labels heparin in human mast cell granules. New use for an ultrastructural enzyme affinity technique. J Histochem Cytochem 46:695–706
Faust F, Gomez-Lazaro M, Borta H, Agricola B, Schrader M (2008) Rab8 is involved in zymogen granule formation in pancreatic acinar AR42J cells. Traffic 9(6):964–979
Forsberg E, Pejler G, Ringvall M, Lunderius C, Tomasini-Johansson B et al (1999) Abnormal mast cells in mice deficient in a heparin-synthesizing enzyme. Nature 400:773–776
Freedman SD, Scheele GA (1993) Regulated secretory proteins in the exocrine pancreas aggregate under conditions that mimic the trans-Golgi network. Biochem Biophys Res Commun 197:992–999
Freedman SD, Kern HF, Scheele GA (1998) Cleavage of GPI-anchored proteins from the plasma membrane activates apical endocytosis in pancreatic acinar cells. Eur J Cell Biol 75(2):163–173
Gómez-Lázaro M, Rinn C, Aroso M, Amado F, Schrader M (2010) Proteomic analysis of zymogen granules. Expert Rev Proteomics 7(5):735–747
Gundersen HJ (1977) Notes on the estimation of the numerical density of particles: the edge effect. J Microsc 111:219–223
Humphries DE, Wong GW, Friend DS, Gurish MF, Qiu WT et al (1999) Heparin is essential for the storage of specific granule proteases in mast cells. Nature 400:769–772
Huttner WB, Zimmerberg J (2001) Implications of lipid microdomains for membrane curvature, budding and fission. Curr Opin Cell Biol 13:478–484
Ikonen E (2001) Roles of lipid rafts in membrane transport. Curr Opin Cell Biol 13:470–477
Kalus I, Hodel A, Koch A, Kleene R, Edwardson JM, Schrader M (2002) Interaction of syncollin with GP-2, the major membrane protein of pancreatic zymogen granules, and association with lipid microdomains. Biochem J 362:433–442
Kleene R, Dartsch H, Kern H (1999) The secretory lectin ZG16p mediates sorting of enzyme proteins to the zymogen granule membrane in pancreatic acinar cells. Eur J Cell Biol 78(2):79–90
Kleinman HK, McGoodwin EB, Rennard SI, Martin GR (1979) Preparation of collagen substrates for cell attachment: effect of collagen concentration and phosphate buffer. Anal Biochem 94(2):308–312
Kumazawa-Inoue K, Mimura T, Hosokawa-Tamiya S, Nakano Y, Dohmae N et al (2012) ZG16p, an animal homolog of β-prism fold plant lectins, interacts with heparan sulfate proteoglycans in pancreatic zymogen granules. Glycobiology 22(2):258–266
Leblond FA, Viau G, Lainé J, Lebel D (1993) Reconstitution in vitro of the pH-dependent aggregation of pancreatic zymogens en route to the secretory granule: implication of GP-2. Biochem J 291:289–296
Limi S, Ojakian G, Raffaniello R (2012) Rab3D regulates amylase levels, not agonist-induced amylase release, in AR42J cells. Cell Mol Biol Lett 17(2):258–273
Logsdon CD (1986) Glucocorticoids increase cholecystokinin receptors and amylase secretion in pancreatic acinar AR42J cells. J Biol Chem 261:2096–2101
Logsdon CD, Moessner J, Williams JA, Goldfine ID (1985) Glucocorticoids increase amylase mRNA levels, secretory organelles, and secretion in pancreatic acinar AR42J cells. J Cell Biol 100(4):1200–1208
Metkar SS, Wang B, Aguilar-Santelises M, Raja SM, Uhlin-Hansen L et al (2002) Cytotoxic cell granule-mediated apoptosis: perforin delivers granzyme b-serglycin complexes into target cells without plasma membrane pore formation. Immunity 16:417–428
Niemann CU, Cowland JB, Ralfkiaer E, Åbrink M, Pejler G, Borregaard N (2009) Serglycin proteoglycan is not implicated in localizing exocrine pancreas enzymes to zymogen granules. Eur J Cell Biol 88:473–479
Pejler G, Sadler JE (1999) Mechanism by which heparin proteoglycan modulates mast cell chymase activity. Biochemistry 38:12187–12195
Rausch U, Adler G, Weidenbach H, Weidenbach F, Rudolff D et al (1987) Stimulation of pancreatic secretory process in the rat by low-molecular weight proteinase inhibitor—I. Dose-response study on enzyme content and secretion, cholecystokinin release and pancreatic fine structure. Cell Tissue Res 247:187–193
Rebaï O, Le Petit-Thevenin J, Bruneau N, Lombardo D, Vérine A (2005) In vitro angiogenic effects of pancreatic bile salt-dependent lipase. Arterioscler Thromb Vasc Biol 25:359–364
Reggio HA, Dagorn JC (1978) Ionic interactions between bovine chymotrypsinogen A and chondroitin sulfate A.B.C. A possible model for molecular aggregation in zymogen granules. J Cell Biol 78:951–957
Reggio HA, Palade GE (1978) Sulfated compounds in the zymogen granules of the guinea pig pancreas. J Cell Biol 77(2):288–314
Rinn C, Aroso M, Prüssing J, Islinger M, Schrader M (2012) Modulating zymogen granule formation in pancreatic AR42J cells. Exp Cell Res 318(15):1855–1866
Rönnberg E, Melo FR, Pejler G (2012) Mast cell proteoglycans. J Histochem Cytochem 60(12):950–962
Scheele GA, Palade GE (1975) Studies on the guinea pig pancreas. Parallel discharge of exocrine enzyme activities. J Biol Chem 250:2660–2670
Scheele GA, Fukuoka S, Freedman SD (1994) Role of the GP2/THP family of GPI-anchored proteins in membrane trafficking during regulated exocrine secretion. Pancreas 9(2):139–149
Schmidt K, Dartsch H, Linder D, Kern HF, Kleene R (2000) A submembranous matrix of proteoglycans on zymogen granule membranes is involved in granule formation in rat pancreatic acinar cells. J Cell Sci 113:2233–2242
Schmidt K, Schrader M, Kern HF, Kleene R (2001) Regulated apical secretion of zymogens in rat pancreas. Involvement of the glycosylphosphatidylinositol-anchored glycoprotein GP-2, the lectin ZG16p, and cholesterol-glycosphingolipid-enriched microdomains. J Biol Chem 276(17):14315–14323
Schrader M (2004) Membrane targeting in secretion. In: Quinn PJ (ed) Subcellular biochemistry: membrane dynamics and domains, vol 37. Kluwer Academic/Plenum Publishers, New York, pp 391–421
Schwartz NB (1977) Regulation of chondroitin sulfate synthesis. Effect of beta-xylosides on synthesis of chondroitin sulfate proteoglycan, chondroitin sulfate chains, and core protein. J Biol Chem 252:6316–6321
Scott JE (1980) Collagen–proteoglycan interactions. Localization of proteoglycans in tendon by electron microscopy. Biochem J 187:887–891
Scott JE (1985) Proteoglycan histochemistry—a valuable tool for connective tissue biochemists. Coll Relat Res 5:541–575
Swarovsky B, Steinhilber W, Scheele GA, Kern HF (1988) Coupled induction of exocrine proteins and intracellular compartments involved in the secretory pathway in AR4-2J cells by glucocorticoids. Eur J Cell Biol 47:101–111
Tartakoff AM, Jamieson JD, Scheele GA, Palade GE (1975) Studies on the pancreas of the guinea pig. Parallel processing and discharge of exocrine proteins. J Biol Chem 250(7):2671–2677
Tooze SA, Martens GJM, Huttner WB (2001) Secretory granule biogenesis: rafting to the SNARE. Trends Cell Biol 11(3):116–122
Unger ML, Hokland M, Basse PH, Nannmark U, Johansson BR (1997) High dose IL-2-activated murine natural killer (A-NK) cells accumulate glycogen and granules, lose cytotoxicity, and alter target cell interaction in vitro. Scand J Immunol 45:623–636
Wernersson S, Pejler G (2014) Mast cell secretory granules: armed for battle. Nat Rev Immunol 14(7):478–494
Acknowledgments
We would like to thank Waltraud Ackermann (Marburg) and Tina Schrader (Exeter) for excellent technical assistance, Katja Schmidt for helpful comments, Ines Castro for help with the Figures, Hans and Beate Schrader for data mining and all those colleagues who provided antibodies (see “Materials and methods” section). This work was supported by the German Research Foundation (DFG; SCHR 518/5-1, 2) and the Portuguese Foundation for Science and Technology (FCT) [SFRH/BD/48722/2008 to M. A.].
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.

418_2015_1339_MOESM1_ESM.jpg
Supplementary Figure S1. Inhibition of proteoglycan synthesis in AR42J cells. Dexamethasone-stimulated AR42J cells were treated with different concentrations of β-d-xyloside (b-D-xyl) (0–2.5 mM) for 48 h, and the relative amounts of precipitable proteoglycans in cell homogenates were analyzed using the Blyscan assay. The data are from 4 independent experiments and are expressed as mean ± S.D. (JPEG 179 kb)

418_2015_1339_MOESM2_ESM.jpg
Supplementary Figure S2. Co-localisation of carboxypeptidase A and the Golgi marker p115 in unstimulated AR42J cells. Unstimulated AR42J cells (- dexa) were processed for immunofluorescence and stained with antibodies directed against the Golgi marker protein p115 (A) and the granule enzyme carboxypeptidase A (CBP A) (B). Note the co-localisation of both proteins at the Golgi complex (arrows) and the presence of fine punctate structures positive for carboxypeptidase A, which may represent constitutive cargo containers (B). Bar, 10 µm. (JPEG 500 kb)
Rights and permissions
About this article
Cite this article
Aroso, M., Agricola, B., Hacker, C. et al. Proteoglycans support proper granule formation in pancreatic acinar cells. Histochem Cell Biol 144, 331–346 (2015). https://doi.org/10.1007/s00418-015-1339-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-015-1339-x