Abstract
Aim
To evaluate the incidence and clinical indications for which eyes were treated for retinopathy of prematurity (ROP) outside the guidelines set by International Classification of ROP (ICROP).
Methods
Medical records of the patients treated at a single tertiary care ophthalmology hospital for ROP from January 2016 to December 2019 were retrospectively analysed to evaluate the indications for which they were treated.
Results
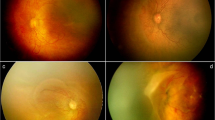
Out of 241 eyes, 33 eyes (13.7%) were treated outside the guidelines. The reasons for the treatment outside the guidelines were structural changes (n = 24, 72.7%), persistent stage 3 ROP that did not show any sign of regression for 6 weeks (n = 7, 21.2%) and active ROP with fellow eye being treated (n = 2, 6.1%). The recorded specific structural changes were tangential traction with temporal vessel straightening concerning for macular distortion and ectopia (n = 5, 15.2%), and stage 3 neovascularisation or ridge with anteroposterior traction with risk of progression to stage 4 disease (n = 19, 57.6%). Pre-plus disease was present in 11 eyes (33.3%).After the treatment, ROP stages regressed and retinal vessels grew either until the ora or at least into zone III in all the treated eyes. None of the eyes showed worsening of structural changes after treatment. The mean follow-up of the patients was 12.4 ± 11.7 months.
Conclusion
Experts occasionally recommend treatment in eyes with disease milder than type 1 ROP. This study may help paediatric retinal practitioners in decision-making in borderline cases.




Similar content being viewed by others
References
Kim SJ, Port AD, Swan R et al (2018) Retinopathy of prematurity: a review of risk factors and their clinical significance. Surv Ophthalmol 63(5):618–637
Stenkuller PG, Du L, Gilbert C et al (1999) Childhood blindness. J AAPOS 3(1):26–32
Shah PK, Prabhu V, Karandikar SS et al (2016) Retinopathy of prematurity: past, present and future. World J Clin Pediatr 5(1):35–46
International Committee for the Classification of Retinopathy of Prematurity (2005) The International Classification of Retinopathy of Prematurity revisited. Arch Ophthalmol 123(7):991–999
Early treatment for retinopathy of prematurity cooperative group (2003) Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol 121(12):1684–1694
Gupta MP, Chan RVP, Anzures R et al (2016) Practice patterns in retinopathy of prematurity treatment for disease milder than recommended by guidelines. Am J Ophthalmol 163:1–10
Darlow BA, Lui K, Kusuda S et al (2017) International variations and trends in the treatment for retinopathy of prematurity. Br J Ophthalmol 101(10):1399–1404
Liu T, Tomlinson LA, Ying GS et al (2019) Treatment of non-type 1 retinopathy of prematurity in the postnatal growth and retinopathy of prematurity (G-ROP) study. J AAPOS 23(6):332.e1–332.e6
Moshfeghi DM (2018) Top five legal pitfalls in retinopathy of prematurity. Curr Opin Ophthalmol 29(3):206–209
Bettman JW (1985) The retinopathy of prematurity: medicolegal aspects. Surv Ophthalmol 29(5):371–373
Demorest BH (1996) Retinopathy of prematurity requires diligent follow-up care. Surv Ophthalmol 41(2):175–178
Day S, Menke AM, Abbott RL (2009) Retinopathy of prematurity malpractice claims: the ophthalmic mutual insurance company experience. Arch Ophthalmol 127(6):794–798
Mills MD (2009) Retinopathy of prematurity malpractice claims. Arch Ophthalmol 127(6):803–804
Reynolds JD (2007) Malpractice and the quality of care in retinopathy of prematurity (an American Ophthalmological Society thesis). Trans Am Ophthalmol Soc 105:461–480
Wiggins RE Jr, Gold RS, Menke AM (2015) Twenty-five years of professional liability in pediatric ophthalmology and strabismus: the OMIC experience. J AAPOS 19(6):535–540
Engelhard SB, Collins M, Shah C et al (2016) Malpractice litigation in pediatric ophthalmology. JAMA Ophthalmol 134(11):1230–1235
Vinekar A, Jayadev C, Dogra MR, Shetty B (2016) Improving follow-up of infants during retinopathy of prematurity screening in rural areas. Indian Paediatr 53:S151–S154
Vinekar A, Avadhani K, Dogra M et al (2012) A novel, low-cost method of enrolling infants at risk for retinopathy of prematurity in centers with no screening program: the REDROP study. Ophthalmic Epidemiol 19(5):317–321
Adams GG, Bunce C, Xing W et al (2017) Treatment trends for retinopathy of prematurity in the UK: active surveillance study of infants at risk. BMJ Open 7(3):e013366
Sekeroglu MA, Hekimoglu E, Sekeroglu HT, Arslan U (2013) Retinopathy of prematurity: a nationwide survey to evaluate current practices and preferences of ophthalmologists. Eur J Ophthalmol 23(4):546–552
Dhawan A, Dogra M, Vinekar A et al (2008) Structural sequelae and refractive outcome after successful laser treatment for threshold retinopathy of prematurity. J Pediatr Ophthalmol Strabismus 45(6):356–361
Axer-Siegel R, Maharshak I, Snir M et al (2008) Diode laser treatment of retinopathy of prematurity: anatomical and refractive outcomes. Retina. 28(6):839–846
Wu WC, Lien R, Liao PJ et al (2015) Serum levels of vascular endothelial growth factor and related factors after intravitreous bevacizumab injection for retinopathy of prematurity. JAMA Ophthalmol 133(4):391–397
Kong L, Bhatt AR, Demny AB et al (2015) Pharmacokinetics of bevacizumab and its effects on serum VEGF and IGF-1 in infants with retinopathy of prematurity. Invest Ophthalmol Vis Sci 56(2):956–961
Sato T, Wada K, Arahori H et al (2012) Serum concentrations of bevacizumab (avastin) and vascular endothelial growth factor in infants with retinopathy of prematurity. Am J Ophthalmol 153(2):327–333
Hong YR, Kim YH, Kim SY et al (2015) Plasma concentrations of vascular endothelial growth factor in retinopathy of prematurity after intravitreal bevacizumab injection. Retina. 35(9):1772–1777
Shukla R, Murthy GVS, Gilbert C et al (2020) Operational guidelines for ROP in India: a summary. Indian J Ophthalmol 68:S108–S114
Multicenter trial of cryotherapy for retinopathy of prematurity (1988) Preliminary results. Cryotherapy for Retinopathy of Prematurity Cooperative Group. Arch Ophthalmol 106(4):471–479
Multicenter trial of cryotherapy for retinopathy of prematurity (1993) 3 1/2- year outcome-structure and function. Cryotherapy for Retinopathy of Prematurity Cooperative Group. Arch Ophthalmol 111(3):339–344
Cryotherapy for Retinopathy of Prematurity Cooperative Group (2001) Multicenter trial of Cryotherapy for Retinopathy of Prematurity: ophthalmological outcomes at 10 years. Arch Ophthalmol 119(8):1110–1118
Backeljauw B, Holland SK, Altaye M, Loepke AW (2015) Cognition and brain structure following early childhood surgery with anesthesia. Pediatrics. 136:e1–e2
Wang X, Xu Z, Miao C-H (2014) Current clinical evidence on the effect of general anesthesia on neurodevelopment in children: an updated systematic review with meta-regression. PLoS One 9:e85760
Quinn GE, Dobson V, Biglan A et al (1995) Correlation of retinopathy of prematurity in fellow eyes in the cryotherapy for retinopathy of prematurity study. Arch Ophthalmol 113(4):469–473
Fielder AR, Shaw DE, Robinson J, Ng YK (1992) Natural history of retinopathy of prematurity: a prospective study. Eye. 6(3):233–242
Good WV, Early Treatment for Retinopathy of Prematurity Cooperative Group (2004) Final results of the Early Treatment for Retinopathy of Prematurity (ETROP) randomized trial. Trans Am Ophthalmol Soc 102:233–248 discussion 248-50
Ying GS, Pan W, Quinn GE et al (2017) Inter eye agreement of retinopathy of prematurity from image evaluation in the Telemedicine Approaches to Evaluating of Acute-Phase ROP (e-ROP) study. Ophthalmol Retina 1:347–354
Geloneck MM, Chuang AZ, Clark WL et al (2014) Refractive outcomes following bevacizumab monotherapy compared with conventional laser treatment: a randomized clinical trial. JAMA Ophthalmol 132(11):1327–1333
Roohipoor R, Karkhaneh R, Riazi-Esfahani M et al (2018) Comparison of intravitreal bevacizumab and laser photocoagulation in the treatment of retinopathy of prematurity. Ophthalmol Retina 2(9):942–948
Anilkumar SE, Anandi V, Shah PK et al (2019) Refractive, sensory, and biometric outcome among retinopathy of prematurity children with a history of laser therapy: a retrospective review from a tertiary care center in South India. Indian J Ophthalmol 67(6):871–876
Bal S, Ying GS, Tomlinson L, Binenbaum G (2019) Postnatal Growth and Retinopathy of Prematurity (G-ROP) Study Group. Association of weight gain acceleration with risk of retinopathy of prematurity. JAMA Ophthalmol 137(11):1301–1305
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Aravind Medical Research Foundation Institutional Ethics Committee, 1, Anna Nagar, Madurai, Tamil Nadu, India (Registration No. ECR/182/INST/TN/ 2013, dated 20 April 2013), institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from parents of all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rajan, R.P., Kohli, P., Babu, N. et al. Treatment of retinopathy of prematurity (ROP) outside International Classification of ROP (ICROP) guidelines. Graefes Arch Clin Exp Ophthalmol 258, 1205–1210 (2020). https://doi.org/10.1007/s00417-020-04706-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04706-8