Abstract
Purpose
To investigate the expression profiles of Toll-like receptor 4 (TLR4), the effect of TLR4 on inflammation, and apoptosis of retinal ganglion cells (RGCs) cultured in high glucose and the underlying mechanism.
Methods
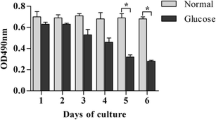
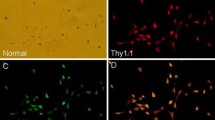
A high-glucose model was established in RGCs isolated from Sprague-Dawley (SD) rats (2–3 days old) and identified with Brn3a. Primary cultured RGCs were divided into control (0 mM), HG1 (10 mM glucose), HG2 (20 mM glucose), HG3 (30 mM glucose), HG (20 mM glucose) + TAK-242 (1.0 μM), and HG (20 mM glucose) + vehicle (1% DMSO) groups. The expression levels of TLR4, its downstream signalling molecules, and pro-inflammatory cytokines were measured by real-time PCR, Western blot or ELISA at 24 h and 48 h. The apoptosis rate of RGCs was measured by flow cytometry.
Results
The mRNA and protein expression levels of TLR4 were increased in high-glucose groups (10 mM, 20 mM, 30 mM). Consistent with these findings, four TLR4 downstream signalling molecules (MyD88, NF-κB, TRAF6, NLRP3) and pro-inflammatory cytokines (IL-1β, IL-18) were upregulated in the three high-glucose groups. Apoptosis of RGCs was clearly increased in the high-glucose group. The administration of TAK-242, an antagonist of TLR4, inhibited inflammation and apoptosis of RGCs in the high-glucose group.
Conclusion
Our results demonstrated that TLR4 plays a critical role in the inflammation and apoptosis of RGCs induced by high glucose. TLR4 might become a novel potential pharmacological target for preventing the progression of DR.






Similar content being viewed by others
References
Cheung N, Mitchell P, Wong TY (2010) Diabetic retinopathy. Lancet 376:124–136. doi:10.1016/S0140-6736(09)62124-3
Group DER, Aiello LP, Sun W, Das A, Gangaputra S, Kiss S, Klein R, Cleary PA, Lachin JM, Nathan DM (2015) Intensive diabetes therapy and ocular surgery in type 1 diabetes. N Engl J Med 372:1722–1733. doi:10.1056/NEJMoa1409463
Leasher JL, Bourne RR, Flaxman SR, Jonas JB, Keeffe J, Naidoo K, Pesudovs K, Price H, White RA, Wong TY, Resnikoff S, Taylor HR, Vision Loss Expert Group of the Global Burden of Disease S (2016) Global estimates on the number of people blind or visually impaired by diabetic retinopathy: a meta-analysis from 1990 to 2010. Diabetes Care 39:1643–1649. doi:10.2337/dc15-2171
Ghassibi MP, Chien JL, Abumasmah RK, Liebmann JM, Ritch R, Park SC (2016) Optic nerve head Drusen prevalence and associated factors in clinically normal subjects measured using optical coherence tomography. Ophthalmology DOI. doi:10.1016/j.ophtha.2016.10.035
Grassi MA, Sun W, Gangaputra S, Cleary PA, Hubbard L, Lachin JM, Gao X, Kiss S, Barkmeier AJ, Almony A, Davis M, Klein R, Danis RP, Group DER (2013) Validity of self-report in type 1 diabetic subjects for laser treatment of retinopathy. Ophthalmology 120:2580–2586. doi:10.1016/j.ophtha.2013.06.002
Polack S, Yorston D, Lopez-Ramos A, Lepe-Orta S, Baia RM, Alves L, Grau-Alvidrez C, Gomez-Bastar P, Kuper H (2012) Rapid assessment of avoidable blindness and diabetic retinopathy in Chiapas, Mexico. Ophthalmology 119:1033–1040. doi:10.1016/j.ophtha.2011.11.002
Simo R, Hernandez C, European Consortium for the Early Treatment of Diabetic R (2014) Neurodegeneration in the diabetic eye: new insights and therapeutic perspectives. Trends Endocrinol Metab 25:23–33. doi:10.1016/j.tem.2013.09.005
Ali TK, Matragoon S, Pillai BA, Liou GI, El-Remessy AB (2008) Peroxynitrite mediates retinal neurodegeneration by inhibiting nerve growth factor survival signaling in experimental and human diabetes. Diabetes 57:889–898. doi:10.2337/db07-1669
Rao RC, Dlouhy BJ (2012) Diabetic retinopathy. N Engl J Med 367:184. doi:10.1056/NEJMc1205011#SA2
Sohn EH, van Dijk HW, Jiao C, Kok PH, Jeong W, Demirkaya N, Garmager A, Wit F, Kucukevcilioglu M, van Velthoven ME, DeVries JH, Mullins RF, Kuehn MH, Schlingemann RO, Sonka M, Verbraak FD, Abramoff MD (2016) Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc Natl Acad Sci U S A 113:E2655–E2664. doi:10.1073/pnas.1522014113
Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW (1998) Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest 102:783–791. doi:10.1172/JCI2425
Carrasco E, Hernandez C, Miralles A, Huguet P, Farres J, Simo R (2007) Lower somatostatin expression is an early event in diabetic retinopathy and is associated with retinal neurodegeneration. Diabetes Care 30:2902–2908. doi:10.2337/dc07-0332
Cheung N, Donaghue KC, Liew G, Rogers SL, Wang JJ, Lim SW, Jenkins AJ, Hsu W, Li Lee M, Wong TY (2009) Quantitative assessment of early diabetic retinopathy using fractal analysis. Diabetes Care 32:106–110. doi:10.2337/dc08-1233
Xu H, Chen M, Forrester JV (2009) Para-inflammation in the aging retina. Prog Retin Eye Res 28:348–368. doi:10.1016/j.preteyeres.2009.06.001
Adamis AP, Berman AJ (2008) Immunological mechanisms in the pathogenesis of diabetic retinopathy. Semin Immunopathol 30:65–84. doi:10.1007/s00281-008-0111-x
Powell ED, Field RA (1964) Diabetic retinopathy and rheumatoid arthritis. Lancet 2:17–18
Zheng L, Howell SJ, Hatala DA, Huang K, Kern TS (2007) Salicylate-based anti-inflammatory drugs inhibit the early lesion of diabetic retinopathy. Diabetes 56:337–345. doi:10.2337/db06-0789
Adamis AP (2002) Is diabetic retinopathy an inflammatory disease? Br J Ophthalmol 86:363–365
Tang J, Kern TS (2011) Inflammation in diabetic retinopathy. Prog Retin Eye Res 30:343–358. doi:10.1016/j.preteyeres.2011.05.002
Miyake K (2007) Innate immune sensing of pathogens and danger signals by cell surface toll-like receptors. Semin Immunol 19:3–10. doi:10.1016/j.smim.2006.12.002
Akira S, Takeda K, Kaisho T (2001) Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol 2:675–680. doi:10.1038/90609
Reynolds JM, Martinez GJ, Chung Y, Dong C (2012) Toll-like receptor 4 signaling in T cells promotes autoimmune inflammation. Proc Natl Acad Sci U S A 109:13064–13069. doi:10.1073/pnas.1120585109
Racke MK, Hu W, Lovett-Racke AE (2005) PTX cruiser: driving autoimmunity via TLR4. Trends Immunol 26:289–291. doi:10.1016/j.it.2005.03.012
Babazada H, Yamashita F, Hashida M (2014) Suppression of experimental arthritis with self-assembling glycol-split heparin nanoparticles via inhibition of TLR4-NF-kappaB signaling. J Control Release 194:295–300. doi:10.1016/j.jconrel.2014.09.015
Sheedy FJ, Marinou I, O'Neill LA, Wilson AG (2008) The mal/TIRAP S180L and TLR4 G299D polymorphisms are not associated with susceptibility to, or severity of, rheumatoid arthritis. Ann Rheum Dis 67:1328–1331. doi:10.1136/ard.2007.083337
Shichita T, Hasegawa E, Kimura A, Morita R, Sakaguchi R, Takada I, Sekiya T, Ooboshi H, Kitazono T, Yanagawa T, Ishii T, Takahashi H, Mori S, Nishibori M, Kuroda K, Akira S, Miyake K, Yoshimura A (2012) Peroxiredoxin family proteins are key initiators of post-ischemic inflammation in the brain. Nat Med 18:911–917. doi:10.1038/nm.2749
Hakimizadeh E, Kazemi Arababadi M, Shamsizadeh A, Roohbakhsh A, Allahtavakoli M (2016) The possible role of toll-like receptor 4 in the pathology of stroke. Neuroimmunomodulation 23:131–136. doi:10.1159/000446481
Devaraj S, Dasu MR, Rockwood J, Winter W, Griffen SC, Jialal I (2008) Increased toll-like receptor (TLR) 2 and TLR4 expression in monocytes from patients with type 1 diabetes: further evidence of a proinflammatory state. J Clin Endocrinol Metab 93:578–583. doi:10.1210/jc.2007-2185
Berger EA, Carion TW, Jiang Y, Liu L, Chahine A, Walker RJ, Steinle JJ (2016) Beta-adrenergic receptor agonist, compound 49b, inhibits TLR4 signaling pathway in diabetic retina. Immunol Cell Biol 94:656–661. doi:10.1038/icb.2016.21
Tezel G, Yang X (2004) Caspase-independent component of retinal ganglion cell death, in vitro. Invest Ophthalmol Vis Sci 45:4049–4059. doi:10.1167/iovs.04-0490
Mead B, Tomarev S (2016) Evaluating retinal ganglion cell loss and dysfunction. Exp Eye Res 151:96–106. doi:10.1016/j.exer.2016.08.006
Yang H, Zheng S, Qiu Y, Yang Y, Wang C, Yang P, Li Q, Lei B (2014) Activation of liver X receptor alleviates ocular inflammation in experimental autoimmune uveitis. Invest Ophthalmol Vis Sci 55:2795–2804. doi:10.1167/iovs.13-13323
Xiao J, Fu C, Zhang X, Zhu D, Chen W, Lu Y, Ye Z (2015) Soluble monosodium urate, but not its crystal, induces toll like receptor 4-dependent immune activation in renal mesangial cells. Mol Immunol 66:310–318. doi:10.1016/j.molimm.2015.03.250
Murakami T, Nishijima K, Sakamoto A, Ota M, Horii T, Yoshimura N (2011) Foveal cystoid spaces are associated with enlarged foveal avascular zone and microaneurysms in diabetic macular edema. Ophthalmology 118:359–367. doi:10.1016/j.ophtha.2010.03.035
Antonetti DA, Barber AJ, Bronson SK, Freeman WM, Gardner TW, Jefferson LS, Kester M, Kimball SR, Krady JK, KF LN, Norbury CC, Quinn PG, Sandirasegarane L, Simpson IA, Group JDRC (2006) Diabetic retinopathy: seeing beyond glucose-induced microvascular disease. Diabetes 55:2401–2411. doi:10.2337/db05-1635
Kern TS, Barber AJ (2008) Retinal ganglion cells in diabetes. J Physiol 586:4401–4408. doi:10.1113/jphysiol.2008.156695
Xu Y, Yang B, Hu Y, Lu L, Lu X, Wang J, Xu F, Yu S, Huang J, Liang X (2016) Wogonin prevents TLR4-NF-kappaB-medicated neuro-inflammation and improves retinal ganglion cells survival in retina after optic nerve crush. Oncotarget DOI 10.18632/oncotarget.12700
Szatmari-Toth M, Kristof E, Vereb Z, Akhtar S, Facsko A, Fesus L, Kauppinen A, Kaarniranta K, Petrovski G (2016) Clearance of autophagy-associated dying retinal pigment epithelial cells - a possible source for inflammation in age-related macular degeneration. Cell Death Dis 7:e2367. doi:10.1038/cddis.2016.133
Zhao Y, Zhao Y, Zhang M, Zhao J, Ma X, Huang T, Pang H, Li J, Song J (2016) Inhibition of TLR4 Signalling-induced inflammation attenuates secondary injury after diffuse axonal injury in rats. Mediat Inflamm 2016:4706915. doi:10.1155/2016/4706915
Qi Y, Zhao M, Bai Y, Huang L, Yu W, Bian Z, Zhao M, Li X (2014) Retinal ischemia/reperfusion injury is mediated by toll-like receptor 4 activation of NLRP3 inflammasomes. Invest Ophthalmol Vis Sci 55:5466–5475. doi:10.1167/iovs.14-14380
Wang YL, Wang K, Yu SJ, Li Q, Li N, Lin PY, Li MM, Guo JY (2015) Association of the TLR4 signaling pathway in the retina of streptozotocin-induced diabetic rats. Graefes Arch Clin Exp Ophthalmol 253:389–398. doi:10.1007/s00417-014-2832-y
Badshah H, Ali T, Kim MO (2016) Osmotin attenuates LPS-induced neuroinflammation and memory impairments via the TLR4/NFkappaB signaling pathway. Sci Rep 6:24493. doi:10.1038/srep24493
Zhao M, Li CH, Liu YL (2016) Toll-like receptor (TLR)-2/4 expression in retinal ganglion cells in a high-glucose environment and its implications. Genet Mol Res 15. doi:10.4238/gmr.15026998
Van Bergen NJ, Wood JP, Chidlow G, Trounce IA, Casson RJ, Ju WK, Weinreb RN, Crowston JG (2009) Recharacterization of the RGC-5 retinal ganglion cell line. Invest Ophthalmol Vis Sci 50:4267–4272. doi:10.1167/iovs.09-3484
Krishnamoorthy RR, Clark AF, Daudt D, Vishwanatha JK, Yorio T (2013) A forensic path to RGC-5 cell line identification: lessons learned. Invest Ophthalmol Vis Sci 54:5712–5719. doi:10.1167/iovs.13-12085
Kritis AA, Stamoula EG, Paniskaki KA, Vavilis TD (2015) Researching glutamate - induced cytotoxicity in different cell lines: a comparative/collective analysis/study. Front Cell Neurosci 9:91. doi:10.3389/fncel.2015.00091
Li CP, Wang SH, Wang WQ, Song SG, Liu XM (2017) Long noncoding RNA-Sox2OT knockdown alleviates diabetes mellitus-induced retinal ganglion cell (RGC) injury. Cell Mol Neurobiol 37:361–369. doi:10.1007/s10571-016-0380-1
Tsutsumi T, Iwao K, Hayashi H, Kirihara T, Kawaji T, Inoue T, Hino S, Nakao M, Tanihara H (2016) Potential Neuroprotective effects of an LSD1 inhibitor in retinal ganglion cells via p38 MAPK activity. Invest Ophthalmol Vis Sci 57:6461–6473. doi:10.1167/iovs.16-19494
Chen S, Sorrentino R, Shimada K, Bulut Y, Doherty TM, Crother TR, Arditi M (2008) Chlamydia pneumoniae-induced foam cell formation requires MyD88-dependent and -independent signaling and is reciprocally modulated by liver X receptor activation. J Immunol 181:7186–7193
Deng L, Wang C, Spencer E, Yang L, Braun A, You J, Slaughter C, Pickart C, Chen ZJ (2000) Activation of the IkappaB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell 103:351–361
Kim Y, Wang W, Okla M, Kang I, Moreau R, Chung S (2016) Suppression of NLRP3 inflammasome by gamma-tocotrienol ameliorates type 2 diabetes. J Lipid Res 57:66–76. doi:10.1194/jlr.M062828
Author information
Authors and Affiliations
Contributions
Hongxia Yang and Lili Hu conceived and designed the project. Lili Hu performed most of the experiments and wrote the manuscript. Lili Hu and Shuanghong Jiang analysed the data. Ming Ai and Hongxia Yang reviewed and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Funding information
The funding organizations had no role in the design or conduct of this research.
Animal experiments
All procedures performed in the studies were in accordance with the ethical standards of the institution and national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Hu, L., Yang, H., Ai, M. et al. Inhibition of TLR4 alleviates the inflammation and apoptosis of retinal ganglion cells in high glucose. Graefes Arch Clin Exp Ophthalmol 255, 2199–2210 (2017). https://doi.org/10.1007/s00417-017-3772-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-017-3772-0