Abstract
Background
In Coats’ disease, the most recent development in the treatment has been the intravitreal injection of anti-VEGF agents. The purpose of this article was to evaluate the effect of intravitreal bevacizumab as the initial treatment for Coats’ disease in children and adults.
Methods
The study included 14 pediatric patients and five adult patients with Coats’ disease. They were treated with intravitreal bevacizumab (1.25 mg/0.05 ml) as the initial treatment, combined with or without other treatments. The analyses included the evaluation of basic clinical conditions.
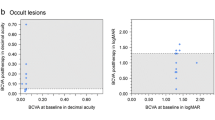
Results
In the pediatric group, after a mean of 9.1 months of follow-up, the differences in visual acuity were significant for the comparisons between the baseline examination and the follow-up examinations carried out at weeks 6, 12, and 24 after the baseline (P = 0.006, P = 0.004, P = 0.005 respectively). Vitreoretinal fibrosis was observed in three patients (n = 3, 21.4 %), among whom two showed fibrosis before treatment. All of the pediatric patients showed a resolution of fluid and exudation, and regression of the telangiectasia. In the adult group, after a mean of 10.6 months of follow-up, the differences in visual acuity were not statistically significant (P > 0.05) between the baseline and follow-up examinations. Vitreoretinal fibrosis (n = 2, 40 %) was observed in two patients who both showed fibrosis before treatment. All of the adult patients showed a resolution of fluid and exudation, and regression of the telangiectasia. The differences in the change of BCVA between children and adults were not significant (P > 0.05) during the follow-up examinations.
Conclusion
The intravitreal injection of bevacizumab as the initial treatment is associated with a measurable gain in visual acuity in patients with Coats’ disease. Resolution of the subretinal fluid and exudation, and regression of the telangiectasia were observed in both pediatric and adult patients. Vitreoretinal fibrosis may be one of the natural courses of Coats’ disease, and it remains uncertain whether bevacizumab accelerates the fibrosis phenomenon.





Similar content being viewed by others
References
Coats G (1908) Forms of retinal disease with massive exudation. Royal London Ophthalmic Hosp Rep 17:440–525
Shields JA, Shields CL, Honavar SG, Demirci H, Cater J (2001) Classification and management of Coats disease: the 2000 Proctor Lecture. Am J Ophthalmol 131:572–583
Shields JA, Shields CL, Honavar SG, Demirci H (2001) Clinical variations and complications of Coats disease in 150 cases: the 2000 Sanford Gifford Memorial Lecture. Am J Ophthalmol 131:561–571
Rishi P, Rishi E, Uparkar M, Sharma T, Gopal L, Bhende P, Bhende M, Sen PR, Sen P (2010) Coats’ disease: an Indian perspective. Indian J Ophthalmol 58:119–124
Smithen LM, Brown GC, Brucker AJ, Yannuzzi LA, Klais CM, Spaide RF (2005) Coats’ disease diagnosed in adulthood. Ophthalmology 112:1072–1078
Entezari M, Ramezani A, Safavizadeh L, Bassirnia N (2010) Resolution of macular edema in Coats’ disease with intravitreal bevacizumab. Indian J Ophthalmol 58:80–82
Lin CJ, Hwang JF, Chen YT, Chen SN (2010) The effect of intravitreal bevacizumab in the treatment of Coats disease in children. Retina 30:617–622
Ray R, Baranano DE, Hubbard GB (2013) Treatment of Coats’ disease with intravitreal bevacizumab. Br J Ophthalmol 97(3):272–277
Wells JR, Hubbard GB 3rd (2011) The effect of intravitreal bevacizumab in the treatment of Coats disease in children. Retina 31:427–428
Zhao T, Wang K, Ma Y, Jiang YR (2011) Resolution of total retinal detachment in Coats’ disease with intravitreal injection of bevacizumab. Graefes Arch Clin Exp Ophthalmol 249:1745–1746
Zhang H, Liu ZL (2012) Increased nitric oxide and vascular endothelial growth factor levels in the aqueous humor of patients with Coats’ disease. J Ocul Pharmacol Ther 28:397–401
Kase S, Rao NA, Yoshikawa H, Fukuhara J, Noda K, Kanda A, Ishida S (2013) Expression of vascular endothelial growth factor in eyes with Coats’ disease. Invest Ophthalmol Vis Sci 54:57–62
Budning AS, Heon E, Gallie BL (1998) Visual prognosis of Coats’ disease. J AAPOS 2:356–359
Aiello LP, Northrup JM, Keyt BA, Takagi H, Iwamoto MA (1995) Hypoxic regulation of vascular endothelial growth factor in retinal cells. Arch Ophthalmol 113:1538–1544
Zhou J, Wang S, Xia X (2012) Role of intravitreal inflammatory cytokines and angiogenic factors in proliferative diabetic retinopathy. Curr Eye Res 37:416–420
Jonas JB, Tao Y, Neumaier M, Findeisen P (2010) Monocyte chemoattractant protein 1, intercellular adhesion molecule 1, and vascular cell adhesion molecule 1 in exudative age-related macular degeneration. Arch Ophthalmol 128:1281–1286
Tao Y, Jonas JB (2010) Intravitreal bevacizumab for exudative age-related macular degeneration: effect on different subfoveal membranes. Retina 30:1426–1431
Moradian S, Ahmadieh H, Malihi M, Soheilian M, Dehghan MH, Azarmina M (2008) Intravitreal bevacizumab in active progressive proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 246:1699–1705
He YG, Wang H, Zhao B, Lee J, Bahl D, McCluskey J (2010) Elevated vascular endothelial growth factor level in Coats’ disease and possible therapeutic role of bevacizumab. Graefes Arch Clin Exp Ophthalmol 248:1519–1521
Kim J, Park KH, Woo SJ (2010) Combined photodynamic therapy and intravitreal bevacizumab injection for the treatment of adult Coats’ disease: a case report. Korean J Ophthalmol 24:374–376
Stergiou PK, Symeonidis C, Dimitrakos SA (2009) Coats’ disease: treatment with intravitreal bevacizumab and laser photocoagulation. Acta Ophthalmol 87:687–688
Ramasubramanian A, Shields CL (2012) Bevacizumab for Coats’ disease with exudative retinal detachment and risk of vitreoretinal traction. Br J Ophthalmol 96:356–359
Arevalo JF, Maia M, Flynn HW Jr, Saravia M, Avery RL, Wu L, Eid Farah M, Pieramici DJ, Berrocal MH, Sanchez JG (2008) Tractional retinal detachment following intravitreal bevacizumab (Avastin) in patients with severe proliferative diabetic retinopathy. Br J Ophthalmol 92:213–216
Honda S, Hirabayashi H, Tsukahara Y, Negi A (2008) Acute contraction of the proliferative membrane after an intravitreal injection of bevacizumab for advanced retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol 246:1061–1063
Hwang JC, Del Priore LV, Freund KB, Chang S, Iranmanesh R (2011) Development of subretinal fibrosis after anti-VEGF treatment in neovascular age-related macular degeneration. Ophthalmic Surg Lasers Imaging 42:6–11
Kuiper EJ, Van Nieuwenhoven FA, de Smet MD, van Meurs JC, Tanck MW, Oliver N, Klaassen I, Van Noorden CJ, Goldschmeding R, Schlingemann RO (2008) The angio-fibrotic switch of VEGF and CTGF in proliferative diabetic retinopathy. PLoS One 3:e2675
O’Neill EC, Qin Q, Van Bergen NJ, Connell PP, Vasudevan S, Coote MA, Trounce IA, Wong TT, Crowston JG (2010) Antifibrotic activity of bevacizumab on human Tenon’s fibroblasts in vitro. Invest Ophthalmol Vis Sci 51:6524–6532
Karatay M, Erdem Y, Koktekir E, Erkoc YS, Caydere M, Bayar MA (2012) The effect of bevacizumab on spinal epidural fibrosis in a postlaminectomy rat model. Turk Neurosurg 22:753–757
Kubota T, Kurihara K, Ishibashi T, Inomata H (1995) Proliferative vitreoretinopathy in Coats’ disease. Clinicohistopathological case report. Ophthalmologica 209:44–46
Muftuoglu G, Gulkilik G (2011) Pars plana vitrectomy in advanced Coats’ disease. Case Rep Ophthalmol 2:15–22
Acknowledgements
Publication of this article was supported by Study and Development Fund of People’s Hospital of Peking University(no. RDC2012-21); National Natural Science Foundation of China(no. 81271027).
Commercial or financial interests
None.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, XX., Jiang, YR. The effect of intravitreal bevacizumab injection as the initial treatment for Coats’ disease. Graefes Arch Clin Exp Ophthalmol 252, 35–42 (2014). https://doi.org/10.1007/s00417-013-2409-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-013-2409-1