Abstract
Purpose
Pericytes, which are recruited to vessels via platelet-derived growth factor (PDGF), are linked to vessel formation and stabilization during pathological corneal neovascularization. In advanced stage, maturated vessels are restricted to anti-VEGF therapy. This study was undertaken to investigate the potential anti-angiogenesis effect of PDGF receptor inhibitor and the therapeutic effect of simultaneously targeting VEGF/PDGF signaling in advanced stage mouse corneal neovascularization.
Method
Corneal neovascularization was induced in wild-type BALB/C mice by removal of the corneal and limbal epithelia. To study the effects of anti-angiogenic inhibitor on new vessel growth prevention, mice were treated with VEGF receptor tyrosin kinase inhibitor II(anti-VEGF/VEGFR group) for 10 days immediately following injury at day 0 (D0). To study the effects of anti-angiogenic inhibitors on vessel regression, mice were treated with VEGF receptor tyrosin kinase inhibitor II or PDGF receptor inhibitor AG 1296 (anti-PDGF/PDGFR group) or both (combined group) for 10 days starting at day 10 (D10) post-injury. Corneas were harvested and immunostained with CD31 for endothelial cells and Ng2 for pericytes. Threshold analysis was used to calculate the area of corneal new vessels. Corneal flatmounts were studied under fluorescence microscope and confocal microscope.
Results
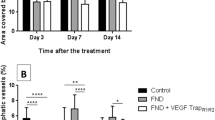
Mice treated with the VEGF receptor tyrosin kinase inhibitor II from D0 showed an inhibition of corneal neovascularization of 15.4% compared with untreated animals (P < 0.01). But the same amount of VEGF receptor tyrosin kinase inhibitor II had no significant anti-angiogenic effect when used at D10 (P > 0.05). Mice treated with either AG1296 or AG1296 plus VEGF receptor tyrosin kinase inhibitor II at D10 showed an inhibition of corneal neovascularization of 12.59% (P < 0.05) and 20.85% (P < 0.01) respectively. Decreased pericyte coverage and vessel density was observed in AG1296 group. Compared to the anti-VEGF/VEGFR group, only the combined therapy group showed a significant anti-angiogenesis effect (P < 0.01).
Conclusion
Inhibition of the PDGF signal pathway results in loss of pericytes and a reduction in vessel density in advanced stage mouse corneal neovascularization. Furthermore, pericyte attenuation caused by blockade PDGF signaling can enhance the anti-angiogenesis efficacy of VEGF receptor inhibitor. Combined treatment against both endothelial cells and pericytes is required for advanced stage corneal new vessels.






Similar content being viewed by others
References
Epstein RJ, Stulting RD, Hendricks RL, Harris DM (1987) Corneal neovascularization. Pathogenesis and inhibition. Cornea 6(4):250–257
Lee P, Wang CC, Adamis AP (1998) Ocular neovascularization: an epidemiologic review. Surv Ophthalmol 43(3):245–269
Risau W (1997) Mechanisms of angiogenesis. Nature 386:671–674
Nehls V, Denzer K, Drenckhahn D (1992) Pericyte involvement in capillary sprouting during angiogenesis in situ. Cell Tissue Res 270:469–474
Cabebe E, Wakelee H (2007) Role of anti-angiogenesis agents in treating NSCLC: focus on bevacizumab and VEGFR tyrosine kinase inhibitors. Curr Treat Options Oncol 8(1):15–27
Adamis AP, Shima DT, Tolentino MJ, Gragoudas ES, Ferrara N, Folkman J, D’Amore PA, Miller JW (1996) Inhibition of vascular endothelial growth factor prevents retinal ischemia-associated iris neovascularization in a nonhuman primate. Arch Ophthalmol 114:66–71
Amano S, Rohan R, Kuroki M, Tolentino M, Adamis AP (1998) Requirement for vascular endothelial growth factor in wound- and inflammation-related corneal neovascularization. Invest Ophthalmol Vis Sci 39:18–22
Robinson GS, Pierce EA, Rook SL, Foley E, Webb R, Smith LE (1996) Oligodeoxynucleotides inhibit retinal neovascularization in a murine model of proliferative retinopathy. Proc Natl Acad Sci USA 93:4851–4856
Krzystolik MG, Afshari MA, Adamis AP, Gaudreault J, Gragoudas ES, Michaud NA, Li W, Connolly E, O’Neill CA, Miller JW (2002) Prevention of experimental choroidal neovascularization with intravitreal anti-vascular endothelial growth factor antibody fragment. Arch Ophthalmol 120:338–346
Singh N, Higgins E, Amin S, Jani P, Richter E, Patel A, Kaur R, Wang J, Ambati J, Dong Z, Ambati BK (2007) Unique homologous siRNA blocks hypoxia-induced VEGF upregulation in human corneal cells and inhibits and regresses murine corneal neovascularization. Cornea 26(1):65–72
Singh N, Higgins E, Amin S, Jani P, Richter E, Patel A, Kaur R, Wang J, Ambati J, Dong Z, Ambati BK (2005) Flt-1 intraceptors inhibit hypoxia-induced VEGF expression in vitro and corneal neovascularization in vivo. Invest Ophthalmol Vis Sci 46(5):1647–1652
Benjamin LE, Hemo I, Keshet E (1998) A plasticity window for blood vessel remodelling is defined by pericyte coverage of the preformed endothelial network and is regulated by PDGF-B and VEGF. Development 125:1591–1598
Benjamin LE, Golijanin D, Itin A, Pode D, Keshet E (1999) Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J Clin Invest 103:159–165
Joussen AM, Poulaki V, Mitsiades N, Stechschulte SU, Kirchhof B, Dartt DA, Fong GH, Rudge J, Wiegand SJ, Yancopoulos GD, Adamis AP (2003) VEGF-dependent conjunctivalization of the corneal surface. Invest Ophthalmol Vis Sci 44:117–123
Proia AD, Chandler DB, Haynes WL, Smith CF, Suvarnamani C, Erkel FH, Klintworth GK (1988) Quantitation of corneal neovascularization using computerized image analysis. Lab Invest 58:473–479
Ebrahem Q, Chaurasia SS, Vasanji A, Qi JH, Klenotic PA, Cutler A, Asosingh K, Erzurum S, Anand-Apte B (2010) Cross-talk between vascular endothelial growth factor and matrix metalloproteinases in the induction of neovascularization in vivo. Am J Pathol 176(1):496–503
Conners MS, Urbano F, Vafeas C, Stoltz RA, Dunn MW, Schwartzman ML (1997) Alkali burn-induced synthesis of inflammatory eicosanoids in rabbit corneal epithelium. Invest Ophthalmol Vis Sci 38(10):1963–1971
Campos-Mollo E, Pérez-Santonja JJ, Lledó-Riquelme M, Ortega Pastor E, Alió JL (2011) New corneal neovascularization model in rabbits for angiogenesis research. Ophthalmic Res 45(3):135–141
Ma DH, Chen JK, Zhang F, Lin KY, Yao JY, Yu JS (2006) Regulation of corneal angiogenesis in limbal stem cell deficiency. Prog Retin Eye Res 25(6):563–590
Alon T, Hemo I, Itin A, Pe’er J, Stone J, Keshet E (1995) Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nat Med 1:1024–1028
Pierce EA, Avery RL, Foley ED, Aiello LP, Smith LE (1995) VEGF expression in a mouse model of retinal neovascularization. Proc Natl Acad Sci USA 92:905–909
Shibuya M, Claesson-Welsh L (2006) Signal transduction by VEGF receptors in regulation of angiogenesis and lymphangiogenesis. Exp Cell Res 312:549–560
Gee MS, Procopio WN, Makonnen S, Feldman MD, Yeilding NM, Lee WM (2003) Tumor vessel development and maturation impose limits on the effectiveness of anti-vascular therapy. Am J Pathol 162:1883–193
Helfrich I, Scheffrahn I, Bartling S, Weis J, von Felbert V, Middleton M, Kato M, Ergün S, Schadendorf D (2010) Resistance to antiangiogenic therapy is directed by vascular phenotype, vessel stabilization, and maturation in malignant melanoma. J Exp Med 207(3):491–503
Cursiefen C, Hofmann-Rummelt C, Küchle M, Schlötzer-Schrehardt U (2003) Pericyte recruitment in human corneal angiogenesis: an ultrastructural study with clinicopathological correlation. Br J Ophthalmol 87(1):101–106
Hoppenreijs VPT, Pels E, Vrensen GF, Felten PC, Treffers WF (1993) Platelet-derived growth factor: receptor expression in corneas and effects on corneal cells. Invest Ophthalmol Vis Sci 34:637–649
Kim WJ, Mohan RR, Mohan RR, Wilson SE (1999) Effect of PDGF, IL-1alpha, and BMP2/4 on corneal fibroblast chemotaxis: expression of the platelet-derived growth factor system in the cornea. Invest Ophthalmol Vis Sci 40(7):1364–1372
Abramsson A, Lindblom P, Betsholtz C (2003) Endothelial and nonendothelial sources of PDGF-B regulate pericyte recruitment and influence vascular pattern formation in tumors. J Clin Invest 112(8):1142–1151
Lindblom P, Gerhardt H, Liebner S, Abramsson A, Enge M, Hellstrom M, Backstrom G, Fredriksson S, Landegren U, Nystrom HC, Bergstrom G, Dejana E, Ostman A, Lindahl P, Betsholtz C (2003) Endothelial PDGF-B retention is required for proper investment of pericytes in the microvessel wall. Genes Dev 17(15):1835–1840
Jain RK, Booth MF (2003) What brings pericytes to tumor vessels? J Clin Invest 112(8):1134–1136
Reinmuth N, Liu W, Jung YD, Ahmad SA, Shaheen RM, Fan F, Bucana CD, McMahon G, Gallick GE, Ellis LM (2001) Induction of VEGF in perivascular cells defines a potential paracrine mechanism for endothelial cell survival. FASEB J 15(7):1239–1241
Bergers G, Song S (2005) The role of pericytes in blood-vessel formation and maintenance. Neuro Oncol 7(4):452–464
Hasumi Y, Kłosowska-Wardega A, Furuhashi M, Ostman A, Heldin CH, Hellberg C (2007) Identification of a subset of pericytes that respond to combination therapy targeting PDGF and VEGF signaling. Int J Cancer 121(12):2606–2614
Erber R, Thurnher A, Katsen AD, Groth G, Kerger H, Hammes HP, Menger MD, Ullrich A, Vajkoczy P (2004) Combined inhibition of VEGF and PDGF signaling enforces tumor vessel regression by interfering with pericyte-mediated endothelial cell survival mechanisms. FASEB J 18(2):338–340
Bergers G, Song S, Meyer-Morse N, Bergsland E, Hanahan D (2003) Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J Clin Investig 111(9):1287–1295
Uzui H, Lee JD, Shimizu H, Tsutani H, Ueda T (2000) The role of protein–tyrosine phosphorylation and gelatinase production in the migration and proliferation of smooth muscle cells. Atherosclerosis 149(1):51–59
Westermark B, Claesson-Welsh L, Heldin CH (1989) Structural and functional aspects of the receptors for platelet-derived growth factor. Prog Growth Factor Res 1(4):253–266
Bradley DA, Dunn R, Nanus D, Stadler W, Dreicer R, Rosenberg J, Smith DC, Hussain M (2007) Randomized, double-blind, placebo-controlled phase II trial of maintenance sunitinib versus placebo after chemotherapy for patients with advanced urothelial carcinoma: scientific rationale and study design. Clin Genitourin Cancer 5(7):460–463
Kim DW, Lu B, Hallahan DE (2004) Receptor tyrosine kinase inhibitors as anti-angiogenic agents. Curr Opin Investig Drugs 5(6):597–604
Pérez-Santonja JJ, Campos-Mollo E, Lledó-Riquelme M, Javaloy J, Alió JL (2010) Inhibition of corneal neovascularization by topical bevacizumab (Anti-VEGF) and sunitinib (anti-VEGF and anti-PDGF) in an animal model. Am J Ophthalmol 150:519–528
No financial relationship with the organization that sponsored the research exists. The authors have full control of all primary data, and agree to allow Graefe’s Archive for Clinical and Experimental Ophthalmology to review their data upon request.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 29 kb)
Rights and permissions
About this article
Cite this article
Chaoran, Z., Zhirong, L. & Gezhi, X. Combination of vascular endothelial growth factor receptor/platelet-derived growth factor receptor inhibition markedly improves the antiangiogenic efficacy for advanced stage mouse corneal neovascularization. Graefes Arch Clin Exp Ophthalmol 249, 1493–1501 (2011). https://doi.org/10.1007/s00417-011-1709-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-011-1709-6