Abstract
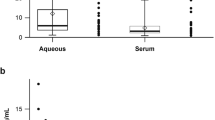
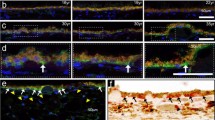
Age-related macular degeneration (AMD) is a leading cause of blindness in the United States, and increasing evidence suggests that it is an inflammatory disease. The prokaryotic obligate intracellular pathogen Chlamydia pneumoniae is emerging as a novel risk factor in cardiovascular disease, and recent sero-epidemiological data suggest that C. pneumoniae infection is also associated with AMD. In this study, we examined choroidal neovascular membrane (CNV) tissue from patients with neovascular AMD for the presence of C. pneumoniae and determined whether the pathogen can dysregulate the function of key cell types in ways that can cause neovascular AMD. Nine CNV removed from patients with neovascular AMD were examined for the presence of C. pneumoniae by immunohistochemistry (IHC) and polymerase chain reaction (PCR); in addition, we performed PCR on nine non-AMD eyes, and IHC on five non-AMD CNV, seven non-AMD eyes, and one internal limiting membrane specimen. Finally, human monocyte-derived macrophages and retinal pigment epithelial (RPE) cells were exposed to C. pneumoniae and assayed in vitro for the production of pro-angiogenic immunomodulators (VEGF, IL-8, and MCP-1). C. pneumoniae was detected in four of nine AMD CNV by IHC and two of nine AMD CNV by PCR, induced VEGF production by human macrophages, and increased production of IL-8 and MCP-1 by RPE cells. In contrast, none of the 22 non-AMD specimens showed evidence for C. pneumoniae. These data indicate that a pathogen capable of inducing chronic inflammation and pro-angiogenic cytokines can be detected in some AMD CNV, and suggest that infection may contribute to the pathogenesis of AMD.





Similar content being viewed by others
References
Anderson DH, Mullins RF, Hageman GS, Johnson LV (2002) A role for local inflammation in the formation of drusen in the aging eye. Am J Ophthalmol 134:411–431
Baffi J, Byrnes G, Chan CC, Csaky KG (2000) Choroidal neovascularization in the rat induced by adenovirus mediated expression of vascular endothelial growth factor. Invest Ophthalmol Vis Sci 41:3582–3589
Beatty WL, Byrne GI, Morrison RP (1993) Morphologic and antigenic characterization of interferon gamma-mediated persistent Chlamydia trachomatis infection in vitro. Proc Natl Acad Sci U S A 90:3998–4002
Belperio JA, Keane MP, Arenberg DA, Addison CL, Ehlert JE, Burdick MD, Strieter RM (2000) CXC chemokines in angiogenesis. J Leukoc Biol 68:1–8
Bonin S, Petrera F, Niccolini B, Stanta G (2003) PCR analysis in archival postmortem tissues. Mol Pathol 56:184–186
Carratelli CR, Rizzo A, Catania MR, Galle F, Losi E, Hasty DL, Rossano F (2002) Chlamydia pneumoniae infections prevent the programmed cell death on THP-1 cell line. FEMS Microbiol Lett 215:69–74
Danesh J,Collins R, Peto R (1997) Chronic infections and coronary heart disease: is there a link? Lancet 350:430–436
Danesh J, Whincup P, Walker M, Lennon L, Thomson A, Appleby P, Wong Y, Bernardes-Silva M, Ward M (2000) Chlamydia pneumoniae IgG titres and coronary heart disease: prospective study and meta-analysis. BMJ 321:208–213
Eckhart L, Bach J, Ban J, Tschachler E (2000) Melanin binds reversibly to thermostable DNA polymerase and inhibits its activity. Biochem Biophys Res Commun 271:726–730
Eyetech Study Group (2003) Anti-vascular endothelial growth factor therapy for subfoveal choroidal neovascularization secondary to age-related macular degeneration: phase II study results. Ophthalmology 110:979–986
Ferrara N, Gerber HP, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676
Fine SL, Berger JW, Maguire MG, Ho AC (2000) Age-related macular degeneration. N Engl J Med 342:483–492
Frank RN, Amin RH, Eliott D, Puklin JE, Abrams GW (1996) Basic fibroblast growth factor and vascular endothelial growth factor are present in epiretinal and choroidal neovascular membranes. Am J Ophthalmol 122:393–403
Gottlieb JL (2002) Age-related macular degeneration. JAMA 288:2233–2236
Hattenbach LO, Falk B, Nurnberger F, Koch FH, Ohrloff C (2002) Detection of inducible nitric oxide synthase and vascular endothelial growth factor in choroidal neovascular membranes. Ophthalmologica 216:209–214
Higgins GT, Wang JH, Dockery P, Cleary PE, Redmond HP (2003) Induction of angiogenic cytokine expression in cultured RPE by ingestion of oxidized photoreceptor outer segments. Invest Ophthalmol Vis Sci 44:1775–1782
Ishida O, Oku H, Ikeda T, Nishimura M, Kawagoe K, Nakamura K (2003) Is Chlamydia pneumoniae infection a risk factor for age related macular degeneration? Br J Ophthalmol 87:523–524
Kalayoglu MV, Byrne GI (2001) Chlamydia. In: Dworkin et al (eds) Prokaryotes. Springer, Berlin Heidelberg New York, http://link.springer-ny.com.ezp2.harvard.edu/link/service/books/10125/
Kalayoglu MV, Byrne GI (1998) Induction of macrophage foam cell formation by Chlamydia pneumoniae. J Infect Dis 177:725–729
Kalayoglu MV, Galvan C, Mahdi OS, Byrne GI, Mansour S (2003) Serological association between Chlamydia pneumoniae infection and age-related macular degeneration. Arch Ophthalmol 121:478–482
Kalayoglu MV, Hoerneman B, LaVerda D, Morrison SG, Morrison RP, Byrne GI (1999) Cellular oxidation of low-density lipoprotein by Chlamydia pneumoniae. J Infect Dis 180:780–790
Kalayoglu MV, Indrawati, Morrison RP, Morrison SG, Yuan Y, Byrne GI (2000) Chlamydial virulence determinants in atherogenesis: the role of chlamydial lipopolysaccharide and heat shock protein 60 in macrophage-lipoprotein interactions. J Infect Dis 181(Suppl 3):S483–S489
Kalayoglu MV, Libby P, Byrne GI (2002) Chlamydia pneumoniae as an emerging risk factor in cardiovascular disease. JAMA 288:2724–2731
Kalayoglu MV, Miranpuri GS, Golenbock DT, Byrne GI (1999) Characterization of low-density lipoprotein uptake by murine macrophages exposed to Chlamydia pneumoniae. Microbes Infect 1:409–418
Kaukoranta-Tolvanen SS, Teppo AM, Laitinen K, Saikku P, Linnavuori K, Leinonen M (1996) Growth of Chlamydia pneumoniae in cultured human peripheral blood mononuclear cells and induction of a cytokine response. Microb Pathog 21:215–221
Kociok N, Heppekausen H, Schraermeyer U, Esser P, Thumann G, Grisanti S, Heimann K (1998) The mRNA expression of cytokines and their receptors in cultured iris pigment epithelial cells: a comparison with retinal pigment epithelial cells. Exp Eye Res 67:237–250
Krzystolik MG, Afshari MA, Adamis AP, Gaudreault J, Gragoudas ES, Michaud NA, Li W, Connolly E, O’Neill CA, Miller JW (2002) Prevention of experimental choroidal neovascularization with intravitreal anti-vascular endothelial growth factor antibody fragment. Arch Ophthalmol 120:338–346
Kuo C, Campbell LA (2000) Detection of Chlamydia pneumoniae in arterial tissues. J Infect Dis 181(Suppl 3):S432–S436
Kuo CC (1999) Pathologic manifestation of chlamydial infection. Am Heart J 138:S496–S499
Lambert HM, Lopez PF (1993) Surgical excision of subfoveal choroidal neovascular membranes. Curr Opin Ophthalmol 4:19–24
Lopez PF, Sippy BD, Lambert HM, Thach AB, Hinton DR (1996) Transdifferentiated retinal pigment epithelial cells are immunoreactive for vascular endothelial growth factor in surgically excised age-related macular degeneration-related choroidal neovascular membranes. Invest Ophthalmol Vis Sci 37:855–868
Lutty GA, McLeod DS, Merges C, Diggs A, Plouet J (1996) Localization of vascular endothelial growth factor in human retina and choroid. Arch Ophthalmol 114:971–977
Miller JW, Adamis AP, Shima DT, D’Amore PA, Moulton RS, O’Reilly MS, Folkman J, Dvorak HF, Brown LF, Berse B et al (1994) Vascular endothelial growth factor/vascular permeability factor is temporally and spatially correlated with ocular angiogenesis in a primate model. Am J Pathol 145:574–584
Relman DA, Schmidt TM, MacDermott RP, Falkow S (1992) Identification of the uncultured bacillus of Whipple’s disease. N Engl J Med 327:293–301
Ross R (1999) Atherosclerosis—an inflammatory disease. N Engl J Med 340:115–126
Rund S, Lindner B, Brade H, Holst O (1999) Structural analysis of the lipopolysaccharide from Chlamydia trachomatis serotype L2. J Biol Chem 274:16819–16824
Salcedo R, Ponce ML, Young HA, Wasserman K, Ward JM, Kleinman HK, Oppenheim JJ, Murphy WJ (2000) Human endothelial cells express CCR2 and respond to MCP-1: direct role of MCP-1 in angiogenesis and tumor progression. Blood 96:34–40
Spilsbury K, Garrett KL, Shen WY, Constable IJ, Rakoczy PE (2000) Overexpression of vascular endothelial growth factor (VEGF) in the retinal pigment epithelium leads to the development of choroidal neovascularization. Am J Pathol 157:135–144
Tong CY, Sillis M (1993) Detection of Chlamydia pneumoniae and Chlamydia psittaci in sputum samples by PCR. J Clin Pathol 46:313–317
Wada M, Ogata N, Otsuji T, Uyama M (1999) Expression of vascular endothelial growth factor and its receptor (KDR/flk-1) mRNA in experimental choroidal neovascularization. Curr Eye Res 18:203–213
Wiegand P, Domhover J, Brinkmann B (1996) DNA degradation in formalin fixed tissues. Pathologe 17:451–454
Wu L, Patten N, Yamashiro CT, Chui B (2002) Extraction and amplification of DNA from formalin-fixed, paraffin-embedded tissues. Appl Immunohistochem Mol Morphol 10:269–274
Yu MJ, Shen WY, Lai MC, Constable IJ, Papadimitriou JM, Rakoczy PE (2000) The role of vascular endothelial growth factor (VEGF) in abnormal vascular changes in the adult rat eye. Growth Factors 17:301–312
Acknowledgements
We thank the New England Eye Bank for its generous gift of whole eye tissue, Gerry Byrne and Ted Dryja for reviewing the manuscript, and Ed Connolly, Holly Goolsby, and Cheryl Greene for their technical expertise and help. This work was supported by an institutional grant from Research to Prevent Blindness (Massachusetts Eye and Ear Infirmary).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalayoglu, M.V., Bula, D., Arroyo, J. et al. Identification of Chlamydia pneumoniae within human choroidal neovascular membranes secondary to age-related macular degeneration. Graefe's Arch Clin Exp Ophthalmo 243, 1080–1090 (2005). https://doi.org/10.1007/s00417-005-1169-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-005-1169-y