Abstract
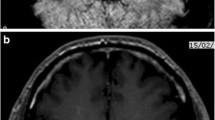
We report a 76-year-old MS patient, treated with DMF for 3 years. Lymphocytes never showed values below 1240/µl. CSF analysis revealed 1,988,880 copies/ml of JCV-DNA, JCV-DNA was detectable in serum and anti-JCV-antibody in CSF and serum were highly positive. Stratify®-JCV-test was positive. CD8-positive T-lymphocytes were reduced. Therapy with mefloquine, mirtazapine and cidofovir resulted in complete elimination of the virus in serum and 90% reduction of viral load in CSF. This case shows that despite careful monitoring for lymphopenia JCV spreading to the CSF may occur during treatment with DMF.

Similar content being viewed by others
References
Berger JR (2017) Classifying PML risk with disease modifying therapies. Mult Scler Relat Disord 12:59–63. https://doi.org/10.1016/j.msard.2017.01.006
Rosenkranz T, Novas M, Terborg C (2015) PML in a patient with lymphocytopenia treated with dimethyl fumarate. N Engl J Med 372:1476–1478. https://doi.org/10.1056/NEJMc1415408
Baharnoori M, Lyons J, Dastagir A et al (2016) Nonfatal PML in a patient with multiple sclerosis treated with dimethyl fumarate. Neurol Neuroimmunol Neuroinflamm 3:e274. https://doi.org/10.1212/NXI.0000000000000274
Lehmann-Horn K, Penkert H, Grein P et al (2016) PML during dimethyl fumarate treatment of multiple sclerosis: how does lymphopenia matter? Neurology 87:440–441. https://doi.org/10.1212/WNL.0000000000002900
European Medicines Agency (2017) Tecfidera-procedural steps taken and scientific information after the authorisation
Major EO, Nath A (2016) A link between long-term natalizumab dosing in MS and PML: putting the puzzle together. Neurol Neuroimmunol Neuroinflamm 3:e235. https://doi.org/10.1212/NXI.0000000000000235
Major EO (2010) Progressive multifocal leukoencephalopathy in patients on immunomodulatory therapies. Annu Rev Med 61:35–47. https://doi.org/10.1146/annurev.med.080708.082655
Longbrake EE, Ramsbottom MJ, Cantoni C et al (2016) Dimethyl fumarate selectively reduces memory T cells in multiple sclerosis patients. Mult Scler J 22:1061–1070. https://doi.org/10.1177/1352458515608961
Funding
Jeremias Motte: received travel Grants from Biogen idec; Janina Kneiphof: none; Katrin Straßburger-Krogias: received travels Grants for scientific meetings from Bayer Vital; Anja Klasing: none; Ortwin Adams: received personal compensation as a speaker from Biogen Idec. Aiden Haghikia: received speaker’s honoraria from Bayer Healthcare, Biogen Idec, Merck Serono, Novartis and Teva. Ralf Gold: serves on scientific advisory boards for Teva Pharmaceutical Industries Ltd., Biogen Idec, Bayer Schering Pharma, and Novartis; has received speaker honoraria from Biogen Idec, Teva Pharmaceutical Industries Ltd., Bayer Schering Pharma, and Novartis; serves as editor for Therapeutic Advances in Neurological Diseases and on the editorial boards of Experimental Neurology and the Journal of Neuroimmunology; and receives research support from Teva Pharmaceutical Industries Ltd., Biogen Idec, Bayer Schering Pharma, Genzyme, Merck Serono, and Novartis, none related to this manuscript.
Author information
Authors and Affiliations
Contributions
Jeremias Motte: data collection, drafting, and revising the manuscript; Janina Kneiphof: data collection, and revising the manuscript; Katrin Straßburger-Krogias: data collection, and revising the manuscript; Anja Klasing: data collection, and revising the manuscript; Ortwin Adams: data collection, and revising the manuscript; Aiden Haghikia: critical comments during data collection, drafting and manuscript revision; Ralf Gold: critical comments during data collection, drafting and manuscript revision.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standards
All procedures performed in studies involving humans participants were in accordance with the ethical standard of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendment or comparable ethical standards.
Informed consent
The patient gave consent prior to participation to the study.
Rights and permissions
About this article
Cite this article
Motte, J., Kneiphof, J., Straßburger-Krogias, K. et al. Detection of JC virus archetype in cerebrospinal fluid in a MS patient with dimethylfumarate treatment without lymphopenia or signs of PML. J Neurol 265, 1880–1882 (2018). https://doi.org/10.1007/s00415-018-8931-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-8931-7