Abstract
Progressive multifocal leukoencephalopathy (PML) is a rare and potentially fatal condition caused by a brain infection with JC polyomavirus (JCV). PML develops almost exclusively in immunocompromised patients and has recently been associated with use of fumaric acid esters (FAEs), or fumarates. We reviewed the literature and the Dutch and European pharmacovigilance databases in order to identify all available FAE-associated PML cases and distinguish possible common features among these patients. A total of 19 PML cases associated with FAE use were identified. Five cases were associated with FAE use for multiple sclerosis and 14 for psoriasis. Ten patients were male and nine were female. The median age at PML diagnosis was 59 years. The median duration of FAE therapy to PML symptom onset or appearance of first PML lesion on brain imaging was 31 months (range 6–110). In all cases a certain degree of lymphocytopenia was reported. The median duration of lymphocytopenia to PML symptom onset was 23 months (range 6–72). The median lymphocyte count at PML diagnosis was 414 cells/µL. CD4 and CD8 counts were reported in ten cases, with median cell count of 137 and 39 cells/µL, respectively. Three patients died (16% mortality). The association between occurrence of PML in patients with low CD4 and CD8 counts is reminiscent of PML cases in the HIV population and suggests that loss of T cells is the most important risk factor.

Similar content being viewed by others
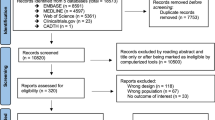
Change history
15 July 2017
An erratum to this article has been published.
References
Monaco MC, Major EO (2015) Immune system involvement in the pathogenesis of JC virus induced PML: what is learned from studies of patients with underlying diseases and therapies as risk factors. Front Immunol 6:159. doi:10.3389/fimmu.2015.00159
Tan CS, Koralnik IJ (2010) Progressive multifocal leukoencephalopathy and other disorders caused by JC virus: clinical features and pathogenesis. Lancet Neurol 9:425–437
Hirsch HH, Kardas P, Kranz D, Leboeuf C (2013) The human JC polyomavirus (JCPyV): virological background and clinical implications. APMIS 121(8):685–727
Maas RP, Muller-Hansma AH, Esselink RA, Murk JL, Warnke C, Killestein J et al (2016) Drug-associated progressive multifocal leukoencephalopathy: a clinical, radiological, and cerebrospinal fluid analysis of 326 cases. J Neurol 263(10):2004–2021. doi:10.1007/s00415-016-8217-x
Berger JR, Aksamit AJ, Clifford DB, Davis L, Koralnik IJ, Sejvar JJ et al (2013) PML diagnostic criteria: consensus statement from the AAN neuroinfectious disease section. Neurology 80(15):1430–1438
Ermis U, Weis J, Schulz JB (2013) PML in a patient treated with fumaric acid. N Engl J Med 368(17):1657–1658. doi:10.1056/NEJMc1211805
Van Oosten BW, Killestein J, Barkhof F, Polman CH, Wattjes MP (2013) PML in a patient treated with dimethyl fumarate from a compounding pharmacy. N Engl J Med 368(17):1658–1659
Sweetser MT, Dawson KT, Bozic C (2013) Manufacturer’s response to case reports of PML. N Engl J Med 368(17):1659–1661. doi:10.1056/NEJMc1300283
Buttmann M, Stoll G (2013) Case reports of PML in patients treated for psoriasis. N Engl J Med 369(11):1081. doi:10.1056/NEJMc1307680#SA2
Stoppe M, Thoma E, Liebert UG, Major EO, Hoffmann KT, Classen J et al (2014) Cerebellar manifestation of PML under fumarate and after efalizumab treatment of psoriasis. J Neurol 261(5):1021–1024
US Food & Drug Administration (2013) FDA approves new multiple sclerosis treatment: Tecfidera. https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm345528.htm
European Medicines Agency (2014) Tecfidera: EPAR—Public assessment report. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/002601/WC500162070.pdf
Rosenkranz T, Novas M, Terborg C (2015) PML in a patient with lymphocytopenia treated with dimethyl fumarate. N Engl J Med 372(15):1476–1478. doi:10.1056/NEJMc1415408
Hughes S (2015) Fourth PML case with Tecfidera in MS calls for vigilance. Medscape. http://www.medscape.com/viewarticle/856148#vp_2. Accessed 21 February 2017
Dubey D, Kieseier BC, Hartung HP, Hemmer B, Warnke C, Menge T et al (2015) Dimethyl fumarate in relapsing-remitting multiple sclerosis: rationale, mechanisms of action, pharmacokinetics, efficacy and safety. Expert Rev Neurother 15(4):339–346. doi:10.1586/14737175.2015.1025755
Spencer CM, Crabtree-Hartman EC, Lehmann-Horn K, Cree BA, Zamvil SS (2015) Reduction of CD8(+) T lymphocytes in multiple sclerosis patients treated with dimethyl fumarate. Neurol Neuroimmunol Neuroinflamm 2(3):e76. doi:10.1212/nxi.0000000000000076
Longbrake EE, Ramsbottom MJ, Cantoni C, Ghezzi L, Cross AH, Piccio L (2016) Dimethyl fumarate selectively reduces memory T cells in multiple sclerosis patients. Mult Scler 22(8):1061–1070. doi:10.1177/1352458515608961
Kihara Y, Groves A, Rivera RR, Chun J (2015) Dimethyl fumarate inhibits integrin alpha4 expression in multiple sclerosis models. Ann Clin Transl Neurol 2(10):978–983. doi:10.1002/acn3.251
Fox RJ, Miller DH, Phillips JT, Hutchinson M, Havrdova E, Kita M et al (2012) Placebo-controlled phase 3 study of oral BG-12 or glatiramer in multiple sclerosis. N Engl J Med 367(12):1087–1097. doi:10.1056/NEJMoa1206328
Gold R, Kappos L, Arnold DL, Bar-Or A, Giovannoni G, Selmaj K et al (2012) Placebo-controlled phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J Med 367(12):1098–1107. doi:10.1056/NEJMoa1114287
Havrdova E, Hutchinson M, Kurukulasuriya NC, Raghupathi K, Sweetser MT, Dawson KT et al (2013) Oral BG-12 (dimethyl fumarate) for relapsing-remitting multiple sclerosis: a review of DEFINE and CONFIRM. Evaluation of: Gold R, Kappos L, Arnold D, et al. Placebo-controlled phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J Med 2012;367:1098-107; and Fox RJ, Miller DH, Phillips JT, et al. Placebo-controlled phase 3 study of oral BG-12 or glatiramer in multiple sclerosis. N Engl J Med 2012;367:1087-97. Expert Opin Pharmacother 14(15):2145–2156. doi:10.1517/14656566.2013.826190
Brennan MS, Matos MF, Li B, Hronowski X, Gao B, Juhasz P et al (2015) Dimethyl fumarate and monoethyl fumarate exhibit differential effects on KEAP1, NRF2 activation, and glutathione depletion in vitro. PLoS One 10(3):e0120254. doi:10.1371/journal.pone.0120254
Lareb (2016) Netherlands Pharmacovigilance Centre Lareb Databse. http://www.lareb.nl. Accessed 6 December 2016
Eudravigilance (2016) Eudravigilance Database. http://bi.eudra.org. Accessed 6 December 2016
Nieuwkamp DJ, Murk JL, van Oosten BW, Cremers CH, Killestein J, Viveen MC et al (2015) PML in a patient without severe lymphocytopenia receiving dimethyl fumarate. N Engl J Med 372(15):1474–1476. doi:10.1056/NEJMc1413724
Bartsch T, Rempe T, Wrede A, Leypoldt F, Bruck W, Adams O et al (2015) Progressive neurologic dysfunction in a psoriasis patient treated with dimethyl fumarate. Ann Neurol 78(4):501–514. doi:10.1002/ana.24471
Dammeier N, Schubert V, Hauser TK, Bornemann A, Bischof F (2015) Case report of a patient with progressive multifocal leukoencephalopathy under treatment with dimethyl fumarate. BMC Neurol 15:108. doi:10.1186/s12883-015-0363-8
Hoepner R, Faissner S, Klasing A, Schneider R, Metz I, Bellenberg B et al (2015) Progressive multifocal leukoencephalopathy during fumarate monotherapy of psoriasis. Neurol Neuroimmunol Neuroinflamm 2(3):e85. doi:10.1212/nxi.0000000000000085
Dubois E, Ruschil C, Bischof F (2015) Low frequencies of central memory CD4 T cells in progressive multifocal leukoencephalopathy. Neurol Neuroimmunol Neuroinflamm 2(6):e177. doi:10.1212/nxi.0000000000000177
Lehmann-Horn K, Penkert H, Grein P, Leppmeier U, Teuber-Hanselmann S, Hemmer B et al (2016) PML during dimethyl fumarate treatment of multiple sclerosis: how does lymphopenia matter? Neurology 87(4):440–441. doi:10.1212/wnl.0000000000002900
Baharnoori M, Lyons J, Dastagir A, Koralnik I, Stankiewicz JM (2016) Nonfatal PML in a patient with multiple sclerosis treated with dimethyl fumarate. Neurol Neuroimmunol Neuroinflamm 3(5):e274. doi:10.1212/nxi.0000000000000274
Hodel J, Darchis C, Outteryck O, Verclytte S, Deramecourt V, Lacour A et al (2016) Punctate pattern: a promising imaging marker for the diagnosis of natalizumab-associated PML. Neurology 86(16):1516–1523
Wijburg MT, Witte BI, Vennegoor A, Roosendaal SD, Sanchez E, Liu Y et al (2016) MRI criteria differentiating asymptomatic PML from new MS lesions during natalizumab pharmacovigilance. J Neurol Neurosurg Psychiatry 87(10):1138–1145. doi:10.1136/jnnp-2016-313772
Wattjes MP, Wijburg MT, Vennegoor A, Witte BI, de Vos M, Richert ND et al (2015) MRI characteristics of early PML–IRIS after natalizumab treatment in patients with MS. J Neurol Neurosurg Psychiatry 87(8):879–884. doi:10.1136/jnnp-2015-311411
Casado JL, Corral I, Garcia J, Martinez-San Millan J, Navas E, Moreno A et al (2014) Continued declining incidence and improved survival of progressive multifocal leukoencephalopathy in HIV/AIDS patients in the current era. Eur J Clin Microbiol Infect Dis 33(2):179–187. doi:10.1007/s10096-013-1941-6
Gheuens S, Bord E, Kesari S, Simpson DM, Gandhi RT, Clifford DB et al (2011) Role of CD4+ and CD8+ T-cell responses against JC virus in the outcome of patients with progressive multifocal leukoencephalopathy (PML) and PML with immune reconstitution inflammatory syndrome. J Virol 85(14):7256–7263. doi:10.1128/jvi.02506-10
Clifford DB, De Luca A, Simpson DM, Arendt G, Giovannoni G, Nath A (2010) Natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: lessons from 28 cases. Lancet Neurol 9(4):438–446. doi:10.1016/S1474-4422(10)70028-4
Longbrake EE, Naismith RT, Parks BJ, Wu GF, Cross AH (2015) Dimethyl fumarate-associated lymphopenia: risk factors and clinical significance. Mult Scler J Exp Transl Clin. doi:10.1177/2055217315596994
Wattjes MP, Rovira A, Miller D, Yousry TA, Sormani MP, De Stefano N et al (2015) Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis—establishing disease prognosis and monitoring patients. Nat Rev Neurol 11(10):597–606
McGuigan C, Craner M, Guadagno J, Kapoor R, Mazibrada G, Molyneux P et al (2016) Stratification and monitoring of natalizumab-associated progressive multifocal leukoencephalopathy risk: recommendations from an expert group. J Neurol Neurosurg Psychiatry 87(2):117–125. doi:10.1136/jnnp-2015-311100
Dong-Si T, Richman S, Wattjes MP, Wenten M, Gheuens S, Philip J et al (2014) Outcome and survival of asymptomatic PML in natalizumab-treated MS patients. Ann Clin Transl Neurol 1(10):755–764. doi:10.1002/acn3.114
Wattjes MP, Vennegoor A, Steenwijk MD, de Vos M, Killestein J, van Oosten BW et al (2015) MRI pattern in asymptomatic natalizumab-associated PML. J Neurol Neurosurg Psychiatry 86(7):793–798. doi:10.1136/jnnp-2014-308630
Wijburg MT, van Oosten BW, Murk J-L, Karimi O, Killestein J, Wattjes MP (2015) Heterogeneous imaging characteristics of JC virus granule cell neuronopathy (GCN): a case series and review of the literature. J Neurol 262(1):65–73. doi:10.1007/s00415-014-7530-5
Wijburg MT, Siepman D, van Eijk JJ, Killestein J, Wattjes MP (2016) Concomitant granule cell neuronopathy in patients with natalizumab-associated PML. J Neurol 263(4):649–656. doi:10.1007/s00415-015-8001-3
Calabrese LH, Molloy E, Berger J (2015) Sorting out the risks in progressive multifocal leukoencephalopathy. Nat Rev Rheumatol 11(2):119–123. doi:10.1038/nrrheum.2014.167
Hoefnagel JJ, Thio HB, Willemze R, Bouwes Bavinck JN (2003) Long-term safety aspects of systemic therapy with fumaric acid esters in severe psoriasis. Br J Dermatol 149(2):363–369
Reich K, Thaci D, Mrowietz U, Kamps A, Neureither M, Luger T (2009) Efficacy and safety of fumaric acid esters in the long-term treatment of psoriasis—a retrospective study (FUTURE). J Dtsch Dermatol Ges 7(7):603–611. doi:10.1111/j.1610-0387.2009.07120.x
Wattjes MP, Wijburg MT, Vennegoor A, Witte BI, Roosendaal SD, Sanchez E et al (2015) Diagnostic performance of brain MRI in pharmacovigilance of natalizumab-treated MS patients. Mult Scler 22(9):1174–1183. doi:10.1177/1352458515615225
European Medicines Agency (2015) Updated recommendations to minimise the risk of the rare brain infection PML with Tecfidera. http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2015/10/news_detail_002423.jsp&mid=WC0b01ac058004d5c1
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Funding
No funding was received.
Ethical standards
All procedures performed were in accordance with the ethical standards stated in the Declaration of Helsinki.
Additional information
The opinions and conclusions of the authors do not necessarily reflect those of the various National Pharmacovigilance Centers or the European Medicines Agency (EMA).
An erratum to this article is available at https://doi.org/10.1007/s00415-017-8557-1.
Rights and permissions
About this article
Cite this article
Gieselbach, RJ., Muller-Hansma, A.H., Wijburg, M.T. et al. Progressive multifocal leukoencephalopathy in patients treated with fumaric acid esters: a review of 19 cases. J Neurol 264, 1155–1164 (2017). https://doi.org/10.1007/s00415-017-8509-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-017-8509-9



