Abstract
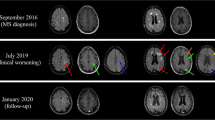
The diagnosis of natalizumab-associated progressive multifocal leukoencephalopathy (PML) in multiple sclerosis patients in an asymptomatic stage is crucial since it is associated with better clinical outcome measures. Current diagnostic criteria on PML diagnosis in asymptomatic patients require the detection of JC virus and corresponding imaging findings. Magnetic resonance imaging (MRI) is the most sensitive diagnostic method for these purposes. However, the diagnosis of asymptomatic natalizumab-associated PML based on MRI findings can be challenging particularly in case of inconclusive or negative results on JC virus detection in the cerebrospinal fluid. In this report, we present a case series demonstrating different diagnostic scenarios of asymptomatic PML diagnosis based on MRI findings in combination with inconclusive or negative results on JC virus detection in the cerebrospinal fluid. We discuss the challenges of applying current PML diagnostic criteria in asymptomatic natalizumab-associated PML patients and stress the need for specific diagnostic criteria and guidelines regarding managing these diagnostic dilemmas in order to facilitate an early and correct diagnosis of PML presumably leading to a better clinical outcome.

Similar content being viewed by others
References
Rudick R, Polman C, Clifford D, Miller D et al (2013) Natalizumab: bench to bedside and beyond. JAMA Neurol 70:172–182
Berger JR, Aksamit AJ, Clifford DB et al (2013) PML diagnostic criteria: consensus statement from the AAN neuroinfectious disease section. Neurology 80:1430–1438
Mentzer D, Prestel J, Adams O et al (2012) Case definition for progressive multifocal leukoencephalopathy following treatment with monoclonal antibodies. J Neurol Neurosurg Psychiatry 83:927–933
Yousry TA, Pelletier D, Cadavid D et al (2012) MRI pattern in natalizumab-associated progressive multifocal leukoencephalopathy. Ann Neurol 72:779–787
Wattjes MP, Richert ND, Killestein J et al (2013) The chameleon of neuroinflammation: magnetic resonance imaging characteristics of natalizumab-associated progressive multifocal leukoencephalopathy. Mult Scler 19:1826–1840
Blair NF, Brew BJ, Halpern JP (2012) Natalizumab-associated PML identified in the presymptomatic phase using MRI surveillance. Neurology 78:507–508
Havla J, Hohlfeld R, Kümpfel T (2014) Unusual natalizumab-associated progressive multifocal leukoencephalopathy starting in the brainstem. J Neurol 261:232–234
Phan-Ba R, Lommers E, Tshibanda L et al (2012) MRI preclinical detection and asymptomatic course of a progressive multifocal leukoencephalopathy (PML) under natalizumab therapy. J Neurol Neurosurg Psychiatry 83:224–226
Lindå H, von Heijne A (2013) Presymptomatic diagnosis with MRI and adequate treatment ameliorate the outcome after natalizumab-associated progressive multifocal leukoencephalopathy. Front Neurol 4:11
Dong-Si T, Richman S, Bloomgren G et al (2013) Survival and functional outcome in asymptomatic natalizumab-associated progressive multifocal leukoencephalopathy multiple sclerosis patients. Mult Scler 19: (S1) P879 Presented at ECTRIMS Copenhagen, 2013
Ryschkewitsch CF, Jensen PN, Monaco MC, Major EO (2010) JC virus persistence following progressive multifocal leukoencephalopathy in multiple sclerosis patients treated with natalizumab. Ann Neurol 68:384–391
Kuhle J, Gosert R, Bühler R et al (2011) Management and outcome of CSF-JC virus PCR-negative PML in a natalizumab-treated patient with MS. Neurology 77:2010–2016
Fine AJ, Sorbello A, Kortepeter C, Scarazzini L (2014) Progressive multifocal leukoencephalopathy after natalizumab discontinuation. Ann Neurol 75:108–115
Wattjes MP, Killestein J (2014) PML after natalizumab discontinuation: few and true? Ann Neurol 75:462
Warnke C, Markwerth P, Pawlita M et al (2013) The anti-JC virus antibody index in CSF for diagnosis of progressive multifocal leukoencephalopathy (PML) in patients with multiple sclerosis. Mult Scler (S1): 181. Presented at ECTRIMS Copenhagen, 2013
Conflicts of interest
Dr. Wattjes serves as a consultant for Biogen-Idec and Roche. He serves on the editorial board of European Radiology. Dr. Vennegoor reports no disclosures. Dr. Mostert reports no disclosures. Dr. van Oosten reports no disclosures. Dr. Barkhof serves on the editorial boards of Brain, European Radiology, Neuroradiology, Multiple Sclerosis and Radiology and serves as a consultant for Bayer-Schering Pharma, Sanofi-Aventis, Biogen-Idec, Teva, Novartis, Roche, Synthon BV, Genzyme, Jansen Research. Dr. Killestein has accepted consulting fees from Merck-Serono, TEVA, Biogen and Novartis. VU Medical Center has received financial support for research activities from Bayer Schering Pharma, Biogen-Idec, GlaxoSmithKline, Merck Serono, Novartis, and Teva.
Ethical standards
This case study has been approved by the appropriate ethics committee and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wattjes, M.P., Vennegoor, A., Mostert, J. et al. Diagnosis of asymptomatic natalizumab-associated PML: are we between a rock and a hard place?. J Neurol 261, 1139–1143 (2014). https://doi.org/10.1007/s00415-014-7336-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7336-5