Abstract
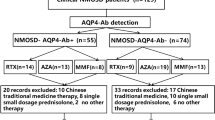
Neuromyelitis optica (NMO) and associated NMO spectrum disorders (NMOSDs) are neuroinflammatory diseases that frequently result in severe neurological disabilities. The aim of this study was to explore additional treatment options for NMO/NMOSD patients who are seropositive for anti-aquaporin 4 (AQP4) antibodies. We retrospectively evaluated the efficacy of immunosuppressants for NMO/NMOSDs by reviewing the clinical records of 52 patients confirmed as seropositive for anti-AQP4 antibodies. Of the 52 patients, 26 (23 women, three men) had received at least one kind of immunosuppressant other than corticosteroids. After eliminating ineligible cases, we evaluated the following 24 treatments in 22 patients (20 women, two men) that used azathioprine (AZA) (n = 9), cyclophosphamide (n = 1), cyclosporine A (CyA) (n = 9), tacrolimus (n = 2), methotrexate (n = 1), and mizoribine (n = 2). Both AZA and CyA treatments allowed us to decrease the median dose of the coadministered prednisone without affecting the expanded disability severity scale scores. In patients with relapsing-remitting courses, the annual relapse rate decreased from 1.7 (1.2–2.7) to 0.47 (0.36–0.59) after AZA treatments (n = 6, P = 0.028), and also showed a significant decrease from 2.7 (1.8–4.3) to 0.38 (0–0.97) after CyA treatment (n = 8, P = 0.012). These results indicate that CyA as well as AZA may help stabilize the disease activity in NMO/NMOSD patients seropositive for anti-AQP4 antibodies. This is the first case series study demonstrating the efficacy of CyA for the treatment of NMO/NMOSDs.

Similar content being viewed by others
References
Wingerchuk DM, Lennon VA, Lucchinetti CF, Pittock SJ, Weinshenker BG (2007) The spectrum of neuromyelitis optica. Lancet Neurol 6:805–815
Watanabe S, Misu T, Miyazawa I et al (2007) Low-dose corticosteroids reduce relapses in neuromyelitis optica: a retrospective analysis. Mult Scler 13:968–974
Costanzi C, Matiello M, Lucchinetti CF et al (2011) Azathioprine: tolerability, efficacy, and predictors of benefit in neuromyelitis optica. Neurology 77:659–666
Bichuetti DB, Lobato de Oliveira EM, Oliveira DM, Amorin de Souza N, Gabbai AA (2010) Neuromyelitis optica treatment: analysis of 36 patients. Arch Neurol 67:1131–1136
Mok CC, To CH, Mak A, Poon WL (2008) Immunoablative cyclophosphamide for refractory lupus-related neuromyelitis optica. J Rheumatol 35:172–174
Chia WC, Wang JN, Lai MC (2010) Neuromyelitis optica: a case report. Pediatr Neonatol 51:347–352
Weinstock-Guttman B, Ramanathan M, Lincoff N, Napoli SQ, Sharma J, Feichter J, Bakshi R (2006) Study of mitoxantrone for the treatment of recurrent neuromyelitis optica (Devic disease). Arch Neurol 63:957–963
Kim SH, Kim W, Park MS, Sohn EH, Li XF, Kim HJ (2011) Efficacy and safety of mitoxantrone in patients with highly relapsing neuromyelitis optica. Arch Neurol 68:473–479
Jacob A, Matiello M, Weinshenker BG, Wingerchuk DM, Lucchinetti C, Shuster E, Carter J, Keegan BM, Kantarci OH, Pittock SJ (2009) Treatment of neuromyelitis optica with mycophenolate mofetil: retrospective analysis of 24 patients. Arch Neurol 66:1128–1133
Jacob A, Weinshenker BG, Violich I et al (2008) Treatment of neuromyelitis optica with rituximab: retrospective analysis of 25 patients. Arch Neurol 65:1443–1448
Pellkofer HL, Krumbholz M, Berthele A et al (2011) Long-term follow-up of patients with neuromyelitis optica after repeated therapy with rituximab. Neurology 76:1310–1315
Weinshilboum RM, Sladek SL (1980) Mercaptopurine pharmacogenetics: monogenic inheritance of erythrocyte thiopurine methyltransferase activity. Am J Hum Genet 32:651–662
Lindsey JW, Meulmester KM, Brod SA, Nelson F, Wolinsky JS (2012) Variable results after rituximab in neuromyelitis optica. J Neurol Sci 317:103–105
Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG (2006) Revised diagnostic criteria for neuromyelitis optica. Neurology 66:1485–1489
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Rodrigo E, Ruiz JC, Angeles de Cos M et al (2009) Correlation of C0 and C2 levels with cyclosporine side effects in kidney transplantation. Transplant Proc 41:2328–2331
Hangler HB, Ruttmann E, Geltner C et al (2008) Single time point measurement by C2 or C3 is highly predictive in cyclosporine area under the curve estimation immediately after lung transplantation. Clin Transplant 22:35–40
Germano V, Picchianti Diamanti A, Ferlito C et al (2011) Cyclosporine A in the long-term management of systemic lupus erythematosus. J Biol Regul Homeost Agents 25:397–403
Kotani T, Takeuchi T, Makino S et al (2011) Combination with corticosteroids and cyclosporin-A improves pulmonary function test results and chest HRCT findings in dermatomyositis patients with acute/subacute interstitial pneumonia. Clin Rheumatol 30:1021–1028
Griveas I, Visvardis G, Papadopoulou D et al (2010) Effect of cyclosporine therapy with low doses of corticosteroids on idiopathic nephrotic syndrome. Artif Organs 34:234–237
Utsugisawa K, Nagane Y, Suzuki S, Suzuki N (2008) Monitoring treatment with cyclosporine microemulsion in myasthenia gravis. Eur J Neurol 15:598–604
Odaka M, Tatsumoto M, Susuki K, Hirata K, Yuki N (2005) Intractable chronic inflammatory demyelinating polyneuropathy treated successfully with ciclosporin. J Neurol Neurosurg Psychiatry 76:1115–1120
Takeuchi A, Shirai S, Horiuchi K et al (2012) Successful cyclosporine treatment in 2 patients with refractory CIDP, involving monitoring of both AUC(0–4 h) and trough levels. Rinsho Shinkeigaku 52:172–177
Arai M (2009) Relapsing transverse myelitis with anti-aquaporin 4 seropositivity: possible beneficial effects of ciclosporin. Rinsho Shinkeigaku 49:48–51
Heidt S, Roelen DL, Eijsink C et al (2009) Calcineurin inhibitors affect B cell antibody responses indirectly by interfering with T cell help. Clin Exp Immunol 159:199–207
Tsuda K, Yamanaka K, Kitagawa H et al (2012) Calcineurin inhibitors suppress cytokine production from memory t cells and differentiation of naive T cells into cytokine-producing mature T cells. PLoS One 7:e31465
Lucchinetti CF, Mandler RN, McGavern D et al (2002) A role for humoral mechanisms in the pathogenesis of Devic’s neuromyelitis optica. Brain 125:1450–1461
Lennon VA, Kryzer TJ, Pittock SJ, Verkman AS, Hinson SR (2005) IgG marker of optic-spinal multiple sclerosis binds to the aquaporin-4 water channel. J Exp Med 202:473–477
Kira J (2011) Autoimmunity in neuromyelitis optica and opticospinal multiple sclerosis: astrocytopathy as a common denominator in demyelinating disorders. J Neurol Sci 311:69–77
Matsuya N, Komori M, Nomura K et al (2011) Increased T-cell immunity against aquaporin-4 and proteolipid protein in neuromyelitis optica. Int Immunol 23:565–573
Uzawa A, Mori M, Ito M et al (2009) Markedly increased CSF interleukin-6 levels in neuromyelitis optica, but not in multiple sclerosis. J Neurol 256:2082–2084
Içöz S, Tüzün E, Kürtüncü M et al (2010) Enhanced IL-6 production in aquaporin-4 antibody positive neuromyelitis optica patients. Int J Neurosci 120:71–75
Araki M, Aranami T, Matsuoka T, Nakamura M, Miyake S, Yamamura T (2012) Clinical improvement in a patient with neuromyelitis optica following therapy with the anti-IL-6 receptor monoclonal antibody tocilizumab. Mod Rheumatol. doi:10.1007/s10165-012-0715-9
Bennett JL, Lam C, Kalluri SR et al (2009) Intrathecal pathogenic antiaquaporin-4 antibodies in early neuromyelitis optica. Ann Neurol 66:617–629
Bradl M, Misu T, Takahashi T et al (2009) Neuromyelitis optica: pathogenicity of patient immunoglobulin in vivo. Ann Neurol 66:630–643
Pohl M, Fischer MT, Mader S et al (2011) Pathogenic T cell responses against aquaporin 4. Acta Neuropathol 122:21–34
Acknowledgments
We would like to thank Dr Toshiyuki Takahashi, Department of Neurology, Tohoku University Graduate School of Medicine, Japan and Dr Takuya Matsushita, Department of Neurology, Neurological Institute, Graduate School of Medical Sciences, Kyushu University, Japan for their support for measuring anti-AQP4 antibodies. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standard
All human studies must state that they have been approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kageyama, T., Komori, M., Miyamoto, K. et al. Combination of cyclosporine A with corticosteroids is effective for the treatment of neuromyelitis optica. J Neurol 260, 627–634 (2013). https://doi.org/10.1007/s00415-012-6692-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-012-6692-2